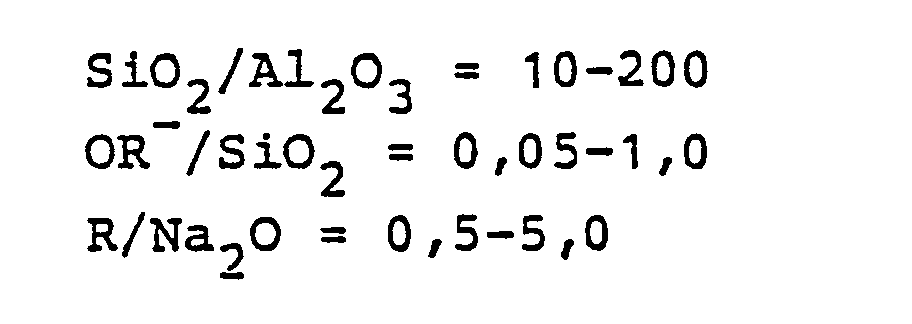EP0064205A1 - Crystalline aluminosilicate, process for producing it and its use in converting methanol and/or dimethyl ether into hydrocarbons - Google Patents
Crystalline aluminosilicate, process for producing it and its use in converting methanol and/or dimethyl ether into hydrocarbons Download PDFInfo
- Publication number
- EP0064205A1 EP0064205A1 EP82103256A EP82103256A EP0064205A1 EP 0064205 A1 EP0064205 A1 EP 0064205A1 EP 82103256 A EP82103256 A EP 82103256A EP 82103256 A EP82103256 A EP 82103256A EP 0064205 A1 EP0064205 A1 EP 0064205A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- crystalline aluminosilicate
- methanol
- catalyst
- zeolite
- hydrocarbons
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J29/00—Catalysts comprising molecular sieves
- B01J29/04—Catalysts comprising molecular sieves having base-exchange properties, e.g. crystalline zeolites
- B01J29/06—Crystalline aluminosilicate zeolites; Isomorphous compounds thereof
- B01J29/70—Crystalline aluminosilicate zeolites; Isomorphous compounds thereof of types characterised by their specific structure not provided for in groups B01J29/08 - B01J29/65
- B01J29/7038—MWW-type, e.g. MCM-22, ERB-1, ITQ-1, PSH-3 or SSZ-25
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B39/00—Compounds having molecular sieve and base-exchange properties, e.g. crystalline zeolites; Their preparation; After-treatment, e.g. ion-exchange or dealumination
- C01B39/02—Crystalline aluminosilicate zeolites; Isomorphous compounds thereof; Direct preparation thereof; Preparation thereof starting from a reaction mixture containing a crystalline zeolite of another type, or from preformed reactants; After-treatment thereof
- C01B39/46—Other types characterised by their X-ray diffraction pattern and their defined composition
- C01B39/48—Other types characterised by their X-ray diffraction pattern and their defined composition using at least one organic template directing agent
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C1/00—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon
- C07C1/02—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon from oxides of a carbon
- C07C1/04—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon from oxides of a carbon from carbon monoxide with hydrogen
- C07C1/0425—Catalysts; their physical properties
- C07C1/043—Catalysts; their physical properties characterised by the composition
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C1/00—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon
- C07C1/20—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon starting from organic compounds containing only oxygen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2529/00—Catalysts comprising molecular sieves
- C07C2529/04—Catalysts comprising molecular sieves having base-exchange properties, e.g. crystalline zeolites, pillared clays
- C07C2529/06—Crystalline aluminosilicate zeolites; Isomorphous compounds thereof
- C07C2529/70—Crystalline aluminosilicate zeolites; Isomorphous compounds thereof of types characterised by their specific structure not provided for in groups C07C2529/08 - C07C2529/65
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F02—COMBUSTION ENGINES; HOT-GAS OR COMBUSTION-PRODUCT ENGINE PLANTS
- F02B—INTERNAL-COMBUSTION PISTON ENGINES; COMBUSTION ENGINES IN GENERAL
- F02B1/00—Engines characterised by fuel-air mixture compression
- F02B1/02—Engines characterised by fuel-air mixture compression with positive ignition
- F02B1/04—Engines characterised by fuel-air mixture compression with positive ignition with fuel-air mixture admission into cylinder
Definitions
- the invention relates to a new zeolite-like material, hereinafter referred to as PSH-3, and a method for producing the same and its use as a catalyst in the production of hydrocarbons from methanol and / or dimethyl ether.
- Zeolites are water-containing framework silicates. They can be described as a rigid three-dimensional framework of SiO 4 - and A10 4 tetrahedra describe that are linked through common oxygen atoms. The charge of the AlO 4 tetrahedron is negative and is balanced, for example, by alkali or alkaline earth metalation. The cavities of the crystalline aluminosilicate are filled with water molecules that can be removed without the framework breaking down.
- the present invention relates to a new zeolite material, that is to say a crystalline aluminosilicate, with a composition corresponding to the formula (in molar ratios of the oxides): where M is a cation of valence n and with a powder X-ray diagram essentially as indicated in Table 1.
- the crystalline aluminosilicate according to the invention can be obtained from a synthesis mixture which contains a silica, an Al compound, alkali and hexamethyleneimine.
- the zeolite PSH-3 proves to be different from all known natural and synthetic zeolites.
- the zeolite PSH-3 according to the invention has the following oxidic composition and has a Polver X-ray diagram, as in the we substantial is listed in Table I.
- the SiO 2 / Al 2 O 3 ratio is preferably 30-100.
- the zeolite PSH-3 in the preferably synthesized form has the following composition: R is an organic nitrogen-containing cation derived from hexamethyleneimine.
- Hexamethyleneimine is a seven-membered heterocycle with one nitrogen atom.
- the synthesis mixture is preferably kept in the following ranges:
- the crystalline aluminosilicate zeolite PSH-3 has the following characteristic lines in the X-ray diagram, which are listed in Table I.
- the values were determined using the K ⁇ radiation of the copper.
- the original cations of the zeolite can already be replaced by ion exchange with other cations known techniques to be replaced.
- the exchange is preferably carried out after calcining the zeolite.
- the reaction time at a temperature of 80 to 180 ° C, about 12 to 144 hours, preferably 110 to 150 ° C for 24 to 96 hours.
- the crystalline product obtained is separated from the mother liquor, washed with water to a pH of 8-10 in the running water and dried at 120.degree.
- the Zeoltih PSH-3 is used as an adsorbent or preferably as a catalyst, the org. Connections and water from the cavities. This is done by heating to 300 to 600 ° C in an atmosphere such as air, nitrogen, ammonia, etc. within 1 to 24 hours.
- zeolites as a catalyst.
- a modification is necessary, as can be done by already known techniques of ion exchange.
- the H-form of the zeolite is obtained either by the action of dilute acids or by exchange with ammonium salts and subsequent calcination.
- the zeolite PSH-3 is preferably used in the dehydrated H form. It is also advantageous if a mixture of the H form with a metal-exchanged form, for example the Mg, Mn, Ba and / or rare earth form, is processed to form a catalyst.
- a metal-exchanged form for example the Mg, Mn, Ba and / or rare earth form
- Zeolite PSH-3 can be processed into catalysts in a variety of particle sizes.
- the particles can be used in the form of a powder, granules or as shaped bodies.
- zeolite PSH-3 described above is processed with inert materials and / or binders to industrially useful catalysts.
- the inert materials or binders, clays, clays, bentonites, smectites, naturally occurring or synthetically produced metal for example aluminum oxide, silicon dioxide, titanium dioxide, zirconium dioxide, thorium oxide, tin oxide, zinc oxide, magnesium oxide and / or beryllium oxide.
- the relative proportions of the zeolite component can be varied within wide limits with regard to the support material used; namely in the range from 1 to 99% and preferably 10 to 80% by weight, based on the dry catalyst mass.
- the PSH-3 zeolite described above is suitable as a catalyst for the conversion of lower aliphatic alcohols and / or ethers, preferably methanol and / or dimethyl ether, into predominantly ali-. phatic unsaturated hydrocarbons with low aromatic content.
- methanol and / or dimethyl ether on zeolites of the ZSM-5 or ZSM-11 type can be converted into water and an aromatic-rich hydrocarbon fraction.
- the catalyst activity can be reduced such that the reaction products are predominantly aliphatic in nature are.
- the preparation of the catalysts is complex and technically not harmless due to the toxicity of organic phosphines, phosphine oxides, phosphoric acid derivatives, etc. (US Pat. No. 3,911 Q41).
- the inventive use of the zeolite PSH-3 as a catalyst for the conversion of methanol and / or dimethyl ether can be carried out either batchwise, semi-continuously or continuously. It is irrelevant whether the catalyst is fixed or movable.
- the methanol insert can consist of pure methanol or of technical crude methanol containing water and oxygenate. An additional dilution of the insert with water can also be carried out.
- This regeneration is expediently carried out by supplying an oxygen-containing atmosphere (for example air or air-nitrogen mixtures) to the catalyst at elevated temperature using known techniques.
- the regeneration temperature should not exceed 600 ° C.
- synthesis gas can be used directly as the feedstock to be converted, provided that the zeolite PSH-3 is coated with metals which are catalytically suitable for the methanol or Fischer-Tropsch synthesis.
- the aluminosilicate according to the invention can thus be used in conjunction with catalysts which are suitable for the hydrogenation of carbon oxides. While the fraction of longer-chain hydrocarbons (C 5+ ) that occurs in the usual implementation of the carbon oxide hydrogenation (FISCHER- T ROPSCH synthesis with metal-containing catalysts) often shows up to over 20 to 25 C atoms in the molecule and is largely straight-chain , the application of the aluminosilicate according to the invention as a cocatalyst in the catalytic hydrogenation of carbon dioxide provides, among other product components, a Ce-hydrocarbon mixture which consists predominantly of molecules with 5 to 12 carbon atoms.
- This chain length-limiting effect of the aluminosilicate according to the invention is associated with the formation of branched hydrocarbons and possibly further reactions leading to aromatics if the reaction is carried out appropriately, so that, for example, a hydrocarbon mixture prepared in this way is much more suitable for further processing in petrol than is the case with the usual execution of FISCHER TROPSCH synthesis resulting hydrocarbon mixture.
- a mixture of 217 ml water glass (27% SiO 2 and 8% Na 2 O) and 316 ml water is mixed with 49.5 g hexamethyleneimine and stirred vigorously in a 2 liter steel autoclave with a stirring insert.
- a solution of 9.22 g of Al sulfate, 23.4 g of H 2 SO 4 (conc.) And 460 ml of water is slowly poured into this mixture with stirring
- the solid is separated from the liquid phase, washed out until the pH was 7-9 and dried at 120.degree.
- the zeolite had the specified table in the X-ray diffraction data and after calcination at 500 ° C fol.
- zeolite PSH-3 in the calcined form 100 g are stirred with 1000 ml of 0.5N HCl at 80 ° C. for 5 hours. The solid is separated off and washed. The exchange is repeated two more times.
- the H form of the zeolite can be used as a catalyst for the production of hydrocarbons from methanol.
- Example 1 The batch as in Example 1 was crystallized at 130 ° C. within 144 hours.
- the crystalline product obtained had the same X-ray data as the zeolite PSH-3 from Example 1.
- a mixture of 650 g of silica sol, 790 ml of water and 78 g of NaOH is mixed with 124 g of hexamethyleneimine in a 5 liter steel autoclave with a stirrer insert. This mixture slowly becomes a solution. 23 g of Al-sulfonic fa t, 58.5 g H 2 SO 4 (conc.) And 1150 ml H 2 0 woks.
- the reaction mixture is allowed to crystallize at 150 ° C. for 5 days with stirring.
- the crystalline solid is separated off, washed out until the pH in the effluent water was 8-9. After calcination, the zeolite had the X-ray diffraction data in Table 1.
- the crystalline end product mainly had the X-ray data in Table 1, in addition to small amounts of quartz.
- the crystalline end product had the following oxidic composition: 0.2 Na 2 0, A1 2 0 3 , 41 Si0 2 .
- Examples 7 to 8 below relate to the use of the zeolite PSH-3 prepared according to Examples 1-6 as a catalyst for the conversion of methanol and / or dimethyl ether into hydrocarbons.
- the catalyst was brought to a reaction temperature of 370 ° C. under nitrogen and then charged with gaseous methanol in an amount of 790 g per liter of catalyst per hour.
- This example relates to the conversion of a methanol / dimethyl ether / water equilibrium mixture into predominantly aliphatic-olefinic hydrocarbons.
- a stream of 50 ml of methanol per hour was passed in vaporized form without applying pressure at a temperature of 280 ° C. over a bed of catalyst consisting of 100 ml of precipitated and granulated aluminum oxide.
- aluminum oxide catalyzes the conversion of methanol to dimethyl ether (see, for example, JB Senderens, Ann. Chim. Phys. 25, 509 (1912).
- the product mixture leaving the reactor was passed through a mixture according to Examples 1 and 2 without further separation prepared and granulated with an addition of 20% Montigel according to known techniques based on zeolite PSH-3.
- the contact load was 800 g insert per liter catalyst and hour; the transition temperature was 370 ° C.
- the product stream leaving the reactor was composed as follows (without taking the water of reaction into account):
- C -Share can also contain small amounts of oxygen-containing compounds.
- D can be a the resultant dimethyl ether recycled, is calculated for the aliphatic hydrocarbons in the B enzinsiede Suite (C , P + O) a carbon selectivity of approx. 47%. This value increases to approx. 83% if C 3 and C 4 hydrocarbons are also added.
- Carburetor fuels can also be obtained from the latter using known techniques (alkylation processes).
- the precipitate was filtered off, washed neutral with water and dried for 4 h at 150 ° C. in vacuo (20 torr). After grinding, the mass was pressed with graphite and bentonite into cylindrical 5 x 5 mm tablets.
- the catalyst was first filled for 14 h at 300 ° C, normal pressure and a gas load of 1000 Nl / (hx 1 catalyst) and then with carbon monoxide for 8 h at 300 ° C, normal pressure and a gas load of-1000 Nl / (hx 1 catalyst ) treated.
- the chain length-limiting effect of the aluminosilicate according to the invention can be seen from the information in the two examples, according to which 95% by weight of the C 5+ fraction has 12 12 C atoms in the molecule.
Abstract
Die vorliegende Erfindung betrifft ein neues Zeolithmaterial mit einer Zusammensetzung (Molverhältnisse der Oxide) etwa der Formel M2/n O . Al2O3 . (20 - 150) SiO2 (M = Kation der Wertigkeit n), ein Verfahren zu dessen Herstellung unter Verwendung von Hexamethylenimin sowie die Verwendung des Zeolithmaterials als Katalysator für die Umwandlung von Methanol und/oder Dimethylether in Kohlenwasserstoffe.The present invention relates to a new zeolite material with a composition (molar ratio of oxides) approximately of the formula M2 / n O. Al2O3. (20 - 150) SiO2 (M = cation of valence n), a process for its production using hexamethyleneimine and the use of the zeolite material as a catalyst for the conversion of methanol and / or dimethyl ether into hydrocarbons.
Description
Gegenstand der Erfindung ist ein neues zeolithartiges Material, das nachfolgend als PSH-3 bezeichnet wird, sowie ein Verfahren zur Herstellung desselben und dessen Verwendung als Katalysator bei der Herstellung von Kohlen-' wasserstoffen aus Methanol und/oder Dimethylether.The invention relates to a new zeolite-like material, hereinafter referred to as PSH-3, and a method for producing the same and its use as a catalyst in the production of hydrocarbons from methanol and / or dimethyl ether.
Unter Zeolithe versteht man wasserhaltige Gerüstsilikate. Sie lassen sich als starres dreidimensionales Gitter von SiO4 - und A104-Tetraedern beschreiben, die durch gemeinsame Sauerstoffatome verknüpft sind. Die Ladung des AlO4-Tetraeders ist negativ und wird beispielsweise durch ein Alkali- bzw. Erdalkalikation ausgeglichen. Die Hohlräume des kristallinen Alumosilicate sind mit Wassermolekülen besetzt, die entfernt werden können, ohne daß das Gerüst zusammenbricht.Zeolites are water-containing framework silicates. They can be described as a rigid three-dimensional framework of SiO 4 - and A10 4 tetrahedra describe that are linked through common oxygen atoms. The charge of the AlO 4 tetrahedron is negative and is balanced, for example, by alkali or alkaline earth metalation. The cavities of the crystalline aluminosilicate are filled with water molecules that can be removed without the framework breaking down.
Es ist bekannt, organische Stickstoffverbindungen für Zeolith-Synthesen einzusetzen. Hierbei werden vor allem SiO2-reiche Zeolith-Typen erhalten. Am bekanntesten sind in letzter Zeit Zeolithe mit der Bezeichnung ZSM 5 bzw. ZSM 11 geworden, die besondere katalytische Eigenschaften aufweisen. Einen überblick der Zeolithe, die mit Hilfe organischer Kationen erhalten wurden, findet man in "Zeolite Molekular Sieves" von D.W. Breck (Wiley, Interscience 1974), Seiten 304 bis 312 und 348 bis 378.It is known to use organic nitrogen compounds for zeolite syntheses. In particular, zeolite types rich in SiO 2 are obtained. Zeolites with the designations ZSM 5 and ZSM 11 have become best known in recent times, the special catalytic properties have ten. An overview of the zeolites obtained with the aid of organic cations can be found in "Zeolite Molecular Sieves" by DW Breck (Wiley, Interscience 1974), pages 304 to 312 and 348 to 378.
Gegenstand der vorliegenden Erfindung ist ein neues Zeolithmaterial, also ein kristallines Alumosilicat, mit einer Zusammensetzung entsprechend der Formel (in Molverhältnissen der Oxide):
Das erfindungsgemäße kristalline Alumonosilicat kann aus einer Synthesemischung erhalten werden, die eine Kieselsäure, eine Al-Verbindung, Alkali und Hexamethylenimin enthält.The crystalline aluminosilicate according to the invention can be obtained from a synthesis mixture which contains a silica, an Al compound, alkali and hexamethyleneimine.
Der Zeolith PSH-3 erweist sich anhand von Röntgenbeugungsdaten und anderen Charakteristiken als unterschiedlich von allen bisher bekannten natürlichen und synthetischen Zeolithen.On the basis of X-ray diffraction data and other characteristics, the zeolite PSH-3 proves to be different from all known natural and synthetic zeolites.
Der erfindungsgemäße Zeolith PSH-3 hat folgende oxidische Zusammensetzung
Vorzugsweise beträgt das SiO2/Al2O3-Verhältnis 30-100.. Der Zeolith PSH-3 hat in der bevorzugt synthetisierten Form folgende Zusammensetzung:
Entsprechend dem Herstellungsverfahren wird ein Gemisch aus einer Mineralsäure, Na20, SiO2, Al2O3, Hexamethylenimin und Wasser gebildet, das die Zusammensetzung in Molverhältnissen von Oxiden in den folgenden Bereichen aufweist:
Hexamethylenimin ist ein siebengliedriger Heterocyclus mit einem Stickstoffatom.Hexamethyleneimine is a seven-membered heterocycle with one nitrogen atom.
Vorzugsweise wird die Synthesemischung in folgenden Bereichen gehalten:
Der kristalline Alumosilicat-Zeolith PSH-3 hat folgende charakteristische Linien im Röntgendiagramm, die in der Tabelle I anfgeführt sind.The crystalline aluminosilicate zeolite PSH-3 has the following characteristic lines in the X-ray diagram, which are listed in Table I.
Die Werte wurden mit der K α-Strahlung des Kupfers ermittelt. Die relativen Intensitäten 100 I/I0, wobei I0 die Intensität der stärksten Linie und d der Netzebenenabstand in Å ist, wurden aus dem Schreiberdiagramm ermittelt.The values were determined using the K α radiation of the copper. The relative intensities 100 I / I 0 , where I 0 is the intensity of the strongest line and d the network plane spacing in Å, were determined from the plotter diagram.
In der Tabelle I werden die relativen Intensitäten wie folgt beschrieben.
Die Gleichgewichtsadsorptionen des kristallinen Alumosilicats PSH-3 für einige Verbindungen sind in der folgenden Tabelle II aufgeführt.
Die ursprünglichen Kationen des Zeoliths können durch Ionenaustausch mit anderen Kationen nach bereits bekannten Techniken ersetzt werden. Bevorzugt erfolgt der Austausch nach dem Calcinieren des Zeoliths.The original cations of the zeolite can already be replaced by ion exchange with other cations known techniques to be replaced. The exchange is preferably carried out after calcining the zeolite.
Der krist. Zeolith wird durch Kristallisieren eines Reaktionsgemisches einer Zusammensetzung in den folgenden oxidischen Molverhältnissen erhalten:
Die Reaktionsdauer beträgt bei einer Temperatur von 80 bis 180°C, ca. 12 bis 144 Stunden, bevorzugt 110 bis 150°C 24 bis 96 Stunden.The reaction time at a temperature of 80 to 180 ° C, about 12 to 144 hours, preferably 110 to 150 ° C for 24 to 96 hours.
Das erhaltene kristalline Produkt wird von der Mutterlauge abgetrennt, mit Wasser auf einen pH-Wert im ablaufenden Wasser von 8 - 10 gewaschen und bei 120°C getrocknet.The crystalline product obtained is separated from the mother liquor, washed with water to a pH of 8-10 in the running water and dried at 120.degree.
Wird der Zeoltih PSH-3 als Adsorbens oder bevorzugt als Katalysator eingesetzt, so erfolgt unter Anwendung üblicher Techniken eine Entfernung der org. Verbindungen und des Wassers aus den Hohlräumen. Dies geschieht durch Erhitzen auf 300 bis 600°C in einer Atmosphäre wie Luft, Stickstoff, Ammoniak usw. innerhalb von 1 bis 24 Stunden.If the Zeoltih PSH-3 is used as an adsorbent or preferably as a catalyst, the org. Connections and water from the cavities. This is done by heating to 300 to 600 ° C in an atmosphere such as air, nitrogen, ammonia, etc. within 1 to 24 hours.
Die Ausgangsstoffe für die Herstellung des erfindungsmäßen Zeoliths sind Materialien, die die geeigneten oxidischen Verbindungen liefern. Hierfür kommen beispielsweise in Frage Na-Aluminat, Al-Sulfat, Al (OH)3, A1203, Kieselsol, SiO2-Füllstoffe, Na-Silikate, NaOH sowie Mineralsäuren wie z.B. H2SO4 und als org. Verbindung Hexamethylenimin.The starting materials for the manufacture of the Invention Moderate zeolites are materials that provide the appropriate oxidic compounds. For example, Na aluminate, Al sulfate, Al (OH) 3 , A1 2 03, silica sol, SiO 2 fillers, Na silicates, NaOH and mineral acids such as H 2 SO 4 and as org. Compound hexamethyleneimine.
Für eine weitere Verwendung der krist. Zeolithe als Katalysator ist. im allgemeinen eine Modifizierung notwendig, wie sie durch bereits bekannte Techniken des Ionenaustausches erfolgen kann. So erhält man die H-Form des Zeoliths entweder durch Einwirken von verdünnten Säuren bzw. durch Austausch mit Ammoniumsalzen und anschließender Calcinierung.For further use of the krist. Zeolites as a catalyst. generally a modification is necessary, as can be done by already known techniques of ion exchange. The H-form of the zeolite is obtained either by the action of dilute acids or by exchange with ammonium salts and subsequent calcination.
Weiterhin sind Austausche mit Ubergangsmetallionen, z.B. der Gruppen VIIa, VIIIa, Ib und IIb möglich. Zusätzliche Austausche bzw. Modifikationen mit Hauptgruppenelementen sind durchführbar.Furthermore, exchanges with transition metal ions, e.g. of groups VIIa, VIIIa, Ib and IIb possible. Additional exchanges or modifications with main group elements can be carried out.
Für die erfindungsgemäße Anwendung wird der Zeolith PSH-3 vorzugsweise in der dehydratisierten H-Form eingesetzt. Ebenso ist es vorteilhaft, wenn ein Gemisch der H-Form mit einer Metall-ausgetauschten Form, beispielsweise der Mg-, Mn-, Ba- und/oder Seltenerdform zu einem Katalysator verarbeitet wird.For the application according to the invention, the zeolite PSH-3 is preferably used in the dehydrated H form. It is also advantageous if a mixture of the H form with a metal-exchanged form, for example the Mg, Mn, Ba and / or rare earth form, is processed to form a catalyst.
Der Zeolith PSH-3 kann in einer Vielzahl von Teilchengrößenzu Katalysatoren verarbeitet werden.Zeolite PSH-3 can be processed into catalysts in a variety of particle sizes.
Je nach verfahrenstechnischer Durchführung (Flugstaubverfahren, Wirbelschichtverfahren, Wanderbettverfahren, Festbettverfahren) können die Teilchen in Form eines Pulvers, eines Granulats oder als Formkörper eingesetzt werden.Depending on the procedural implementation (fly dust process, fluidized bed process, moving bed process, Fixed bed process) the particles can be used in the form of a powder, granules or as shaped bodies.
Zur Herstellung der verschiedenen Anwendungsformen, die nach bekannten Techniken geschehen kann,.ist es u.U. erforderlich, daß man den vorstehend beschriebenen Zeolith PSH-3 mit Inertmaterialien und/oder Bindemitteln zu technisch brauchbaren Katalysatoren verarbeitet.It may be necessary to produce the various forms of use, which can be done using known techniques. required that the zeolite PSH-3 described above is processed with inert materials and / or binders to industrially useful catalysts.
Beispielsweise sind für die Herstellung technischer Katalysatoren auf Basis Zeolith PSH-3 die Inertmaterialien bzw. Bindemittel, Tone, Tonerden, Bentonite, Smectite, natürlich vorkommende oder synthetisch hergestellte Metall- . oxide, beispielsweise Aluminiumoxid, Siliciumdioxid, Titandioxid, Zirkoniumdioxid, Thoriumoxid, Zinnoxid, Zinkoxid, Magnesiumoxid und/oder Berylliumoxid geeignet.For example, for the manufacture of technical catalysts based on zeolite PSH-3, the inert materials or binders, clays, clays, bentonites, smectites, naturally occurring or synthetically produced metal. oxides, for example aluminum oxide, silicon dioxide, titanium dioxide, zirconium dioxide, thorium oxide, tin oxide, zinc oxide, magnesium oxide and / or beryllium oxide.
Die relativen Anteile der Zeolithkomponente können im Hinblick auf das verwendete Trägermaterial in weiten Bereichen variiert werden; nämlich im Bereich von 1 bis 99 % und bevorzugt 10 bis 80 Gew.-%, bezogen auf die trockene Katalysatormasse.The relative proportions of the zeolite component can be varied within wide limits with regard to the support material used; namely in the range from 1 to 99% and preferably 10 to 80% by weight, based on the dry catalyst mass.
Der vorsthend beschriebene Zeolith PSH-3 eignet sich als Katalysator für die Umwandlung von niederen aliphatischen Alkoholen und/oder Ethern, vorzugsweise Methanol und/oder Dimethylether, in überwiegend ali- . phatisch ungesättigte Kohlenwasserstoffe mit niedrigen Aromatengehalt.The PSH-3 zeolite described above is suitable as a catalyst for the conversion of lower aliphatic alcohols and / or ethers, preferably methanol and / or dimethyl ether, into predominantly ali-. phatic unsaturated hydrocarbons with low aromatic content.
Beispielsweise ist bekannt, daß sich Methanol und/oder Dimethylether an Zeolithen vom Typ ZSM-5 oder ZSM-11 .in Wasser und eine aromatenreiche KohlenwasserstoffFraktion umwandeln läßt.For example, it is known that methanol and / or dimethyl ether on zeolites of the ZSM-5 or ZSM-11 type can be converted into water and an aromatic-rich hydrocarbon fraction.
Beispielsweise ist in dpr US-PS 3 894 107 beschrieben, daß das durch Zeolith-Katalyse aus Methanol enthaltene Kohlenwasserstoffgemisch zu 40 % und mehr aus Aromaten bestehen kann. Weiterhin lehrt die US-PS 3 894 105, daß die aus Methanol erhaltenen Aromaten einen beträchtlichen Anteil an 1,2,4,5-Tetramethylbenzol(Durol) aufweisen. Bei Verwendung des aus Methanol erhaltenen Kohlenwasserstoffgemisches als Vergaserkraftstoff ist der Duolanteil in der Aromatenfraktion nachteilig:
- Durol ist in Kohlenwasserstoffen bei Raumtemperatur schwerlöslich, und es neigt aufgrund seines hohen Schmelzpunktes (79,2°C) zum Auskristallisieren. Es ist offentsichtlich, daß dieses Auskristallisieren Störungen beim Betrieb von Verbrennungsmotoren verursachen kann, die mit aus Methanol hergestelltem Kraftstoff betrieben werden. Es hat zahlreiche Versuche gegeben, die Umwandlung von Methanol an Zeolithen so zu steuern, daß die flüssigen Reaktionsprodukte nur wenig Durol bzw. allgemein wenig Aromaten enthalten.
- Durol is sparingly soluble in hydrocarbons at room temperature and due to its high melting point (79.2 ° C) it tends to crystallize. It is obvious that this crystallization can cause disturbances in the operation of internal combustion engines which are operated with fuel made from methanol. There have been numerous attempts to control the conversion of methanol to zeolites in such a way that the liquid reaction products contain little durol or generally little aromatics.
Beispielsweise läßt sich durch Modifizieren eines ZSM-5 Zeolithkatalysators mit organischen Verbindungen des Phosphors die Katalysatoraktivität derartig herabsetzen, daß die Reaktionsprodukte vorwiegend aliphatischer Natur sind. Allerdings ist die Herstellung der Katalysatoren aufwendig und in technischer Hinsicht aufgrund der Toxizität von organischen Phosphinen, Phosphinoxiden, Phosphorsäurederivaten etc. nicht unbedenklich (US-PS 3 911 Q41).For example, by modifying a ZSM-5 zeolite catalyst with organic compounds of phosphorus, the catalyst activity can be reduced such that the reaction products are predominantly aliphatic in nature are. However, the preparation of the catalysts is complex and technically not harmless due to the toxicity of organic phosphines, phosphine oxides, phosphoric acid derivatives, etc. (US Pat. No. 3,911 Q41).
Weiterhin wurde versucht, die Bildung alkylierter Aromaten durch Einstellung bestimmter verfahrenstechnischer Parameter, z.B. Herabsetzung des Partialdruckes des Einsatz-Methanols, zu verhindern. Wie beispielsweise aus der US-PS 4 025 575 hervorgeht, läßt sich durch Verdünnung des als Einsatz dienenden Methanols mit inerten Gasen die Aromatenbildung unterdrücken und die niederen Olefinen, erhöhen. Es ist offenkundig, daß eine solche Verfahrensweise mit technischen Problemen hinsichtlich Raum-Zeit-Ausbeute, Gasrückführung und Trennung der Reaktionsprodukte von den gasförmigen Inertstoffen belastet ist.Attempts have also been made to prevent the formation of alkylated aromatics by setting certain process parameters, e.g. To prevent the partial pressure of the feed methanol from being reduced. As can be seen, for example, from US Pat. No. 4,025,575, dilution of the methanol used as an insert with inert gases can suppress the formation of aromatics and increase the lower olefins. It is evident that such a procedure is burdened with technical problems with regard to space-time yield, gas recirculation and separation of the reaction products from the gaseous inert substances.
überraschenderweise wurde nun gefunden, daß die katalytische Umwandlung von Methanol und/oder Dimethylether in Gegenwart des Zeolithen PSH-3 ein Kohlenwasserstoffgemisch liefert, daß einen niedrigen Aromatengehalt aufweist.Surprisingly, it has now been found that the catalytic conversion of methanol and / or dimethyl ether in the presence of the zeolite PSH-3 provides a hydrocarbon mixture which has a low aromatic content.
Die erfindungsgemäße Anwendung des Zeoliths PSH-3 als Katalysator, zur Umwandlung von Methanol und/oder Dimethylether kann entweder diskontinuierlich, halbkontinuierlich oder kontinuierlich durchgeführt werden. Dabei ist es unerheblich, ob der Katalysator fest oder beweglich angeordnet ist.The inventive use of the zeolite PSH-3 as a catalyst for the conversion of methanol and / or dimethyl ether can be carried out either batchwise, semi-continuously or continuously. It is irrelevant whether the catalyst is fixed or movable.
Die nachfolgende Erläuterung bezieht sich auf Methanol als Einsatzmaterial. Es ist jedoch zu berücksichtigen, daß mit gleichem Erfolg aus Dimethylether oder Dimethylether-Wasser-Mischungen eingesetzt werden können.The following explanation refers to methanol as the feed. However, it must be taken into account that dimethyl ether or dimethyl ether / water mixtures can be used with equal success.
Der Methanol-Einsatz kann aus reinem Methanol oder aus Wasser- und oxygenathaltigem technichem Rohmethanol bestehen. Ebenso kann eine zusätzliche Verdünnung des Einsatzes mit Wasser vorgenommen werden.The methanol insert can consist of pure methanol or of technical crude methanol containing water and oxygenate. An additional dilution of the insert with water can also be carried out.
Das Verfahren ist durch folgende Raktionsbedingungen gekennzeichnet:
Es kann notwendig sein, den verwendeten Katalysator auf Basis Zeolith PSH-3 nach einer gewissen Betiebsdauer von desaktivierenden Ablagerungen zu befreien. Diese Regenerierung wird zweckmäßigerweise durch .Zufuhr einer sauerstoffhaltigen Atmosphäre, (z.B. Luft oder Luft-Stickstoff-Gemische) zum Katalysator bei erhöhter Temperatur nach bekannten Techniken durchgeführt. Die Regeneriertemperatur sollte 600°C nicht übersteigen.It may be necessary to remove the deactivating deposits from the zeolite-based PSH-3 catalyst after a certain operating time. This regeneration is expediently carried out by supplying an oxygen-containing atmosphere (for example air or air-nitrogen mixtures) to the catalyst at elevated temperature using known techniques. The regeneration temperature should not exceed 600 ° C.
Ebenso kann anstelle von Methanol und/oder Dimethylether direkt Synthesegas als umzuwandelndes Einsatzmaterial verwendet werden, sofern der Zeolith PSH-3 mit für die Methanol- bzw. Fischer-Tropsch-Synthese katalytisch geeigneten Metallen belegt ist.Similarly, instead of methanol and / or dimethyl ether, synthesis gas can be used directly as the feedstock to be converted, provided that the zeolite PSH-3 is coated with metals which are catalytically suitable for the methanol or Fischer-Tropsch synthesis.
Das erfindungsgemäße Alumosilicat kann so in Verbindung mit Katalysatoren eingesetzt werden, die für die Kohlenoxidhydrierung geeignet sind. Während die bei der üblichen-Ausführung der.Kohlenoxidhydrierung (FISCHER- TROPSCH-Synthese mit metallhaltigen Katalysatoren) entstehende Fraktion der längerkettigen Kohlenwasserstoffe (C5+) oft bis über 20 bis 25 C-Atome im Molekül aufzeigt und zum großen Teil geradkettig aufgebaut ist, liefert die Anwendung des erfindungsgemäßen Alumosilicats als Cokatalysator bei der katalytischen Kohlenoxidhydrierung neben anderen Produktbestandteilen ein Ce-Kohlenwasserstoffgemisch, das überwiegend aus Molekülen mit 5 bis 12 C-Atomen besteht. Dieser kettenlängenbegrenzende Effekt des erfindungsgemäßen Alumosilicats ist bei entsprechender Führung der Reaktion mit einer Bildung verzweigter Kohlenwasserstoffe und gegebenenfalls weiteren, zu Aromaten führenden Reaktionen verbunden, wodurch z.B. ein so hergestellte Kohlenwasserstoffgemisch zur Weiterverarbeitung in Ottokraftstoff wesentlich besser geeignet ist als das bei der üblichen Ausführung der FISCHER-TROPSCH-Synthese entstehende Kohlenwasserstoffgemisch.The aluminosilicate according to the invention can thus be used in conjunction with catalysts which are suitable for the hydrogenation of carbon oxides. While the fraction of longer-chain hydrocarbons (C 5+ ) that occurs in the usual implementation of the carbon oxide hydrogenation (FISCHER- T ROPSCH synthesis with metal-containing catalysts) often shows up to over 20 to 25 C atoms in the molecule and is largely straight-chain , the application of the aluminosilicate according to the invention as a cocatalyst in the catalytic hydrogenation of carbon dioxide provides, among other product components, a Ce-hydrocarbon mixture which consists predominantly of molecules with 5 to 12 carbon atoms. This chain length-limiting effect of the aluminosilicate according to the invention is associated with the formation of branched hydrocarbons and possibly further reactions leading to aromatics if the reaction is carried out appropriately, so that, for example, a hydrocarbon mixture prepared in this way is much more suitable for further processing in petrol than is the case with the usual execution of FISCHER TROPSCH synthesis resulting hydrocarbon mixture.
Anhand der folgenden Beispiele soll die Erfindung noch häher erläutert werden. %-Angaben beziehen sich - soweit nicht anders vermerkt ist - auf Gew.-%.The following examples are intended to explain the invention in greater detail. Unless otherwise stated,% figures relate to% by weight.
In einem 2 Liter Stahl-Autoklay mit Rühreinsatz wird eine Mischung aus 217 ml Wasserglas (27 % SiO2 und 8 % Na2O) und 316 ml Wasser mit 49,5 g Hexamethylenimin versetzt und intensiv gerührt. In diese Mischung läßt man langsam unter Rühren eine Lösung aus 9,22 g Al-Sulfat, 23,4 g H2SO4 (konz.) und 460 ml Wasser einfließenA mixture of 217 ml water glass (27% SiO 2 and 8% Na 2 O) and 316 ml water is mixed with 49.5 g hexamethyleneimine and stirred vigorously in a 2 liter steel autoclave with a stirring insert. A solution of 9.22 g of Al sulfate, 23.4 g of H 2 SO 4 (conc.) And 460 ml of water is slowly poured into this mixture with stirring
Nachdem das entstandene Reaktionsgel homogenisiert worden ist, läßt man bei 150°C innerhalb von 70 Stunden kristallisieren.After the resulting reaction gel has been homogenized, it is left to crystallize at 150 ° C. within 70 hours.
Nach Ende der Reaktionszeit wird der Feststoff von der flüssigen Phase abgetrennt, ausgewaschen, bis der pH-Wert bei 7-9 lag und bei 120°C getrocknet.At the end of the reaction time, the solid is separated from the liquid phase, washed out until the pH was 7-9 and dried at 120.degree.
Der Zeolith hatte die in der Tabelle angegebenen Röntgenbeugungsdaten und nach dem Calcinieren bei 500°C fol .gende Zusammensetzung: 0,4 Na2O · Al2O3 · 55 SiO2.
100 g Zeolith PSH-3 in der calcinierten Form werden mit 1000 ml 0,5 n HCl bei 80°C 5 Stunden gerührt. Der Feststoff wird abgetrennt und gewaschen. Der Austausch wird noch zweimal wiederholt. Die H-Form des Zeoliths kann als Katalysator für die Herstellung von Kohlenwasserstoffen aus Methanol eingesetzt werden.100 g of zeolite PSH-3 in the calcined form are stirred with 1000 ml of 0.5N HCl at 80 ° C. for 5 hours. The solid is separated off and washed. The exchange is repeated two more times. The H form of the zeolite can be used as a catalyst for the production of hydrocarbons from methanol.
Der Ansatz wie in Beispiel 1 wurde.bei 130°C innerhalb von 144 Stunden kristallisiert. Das erhaltene kristalline Produkt wies die identischen Röntgendaten wie der Zeolith PSH-3 aus Beispiel 1 auf.The batch as in Example 1 was crystallized at 130 ° C. within 144 hours. The crystalline product obtained had the same X-ray data as the zeolite PSH-3 from Example 1.
In einem 5 Liter Stahl-Autoklav mit Rührereinsatz wird eine Mischung aus 650 g Kieselsol, 790 ml Wasser und 78 g NaOH mit 124 g Hexamethylenimin versetzt. diese Mischung wird langsam eine Lösung aus. 23 g Al-Sul- fat, 58,5 g H2SO4 (konz.) und 1150 ml H20 gegegeben.A mixture of 650 g of silica sol, 790 ml of water and 78 g of NaOH is mixed with 124 g of hexamethyleneimine in a 5 liter steel autoclave with a stirrer insert. this mixture slowly becomes a solution. 23 g of Al-sulfonic fa t, 58.5 g H 2 SO 4 (conc.) And 1150 ml H 2 0 woks.
Das Reaktionsgemisch läßt man bei 150°C 5 Tage unter Rühren kristallisieren.The reaction mixture is allowed to crystallize at 150 ° C. for 5 days with stirring.
Der kristalline Feststoff wird abgetrennt, ausgewaschen, bis der pH-Wert im ablaufenden Wasser 8-9 betrug. Der Zeolith hatte nach dem Calcinieren die Röntgenbeugungsdaten der Tabelle 1.The crystalline solid is separated off, washed out until the pH in the effluent water was 8-9. After calcination, the zeolite had the X-ray diffraction data in Table 1.
Ansatz wie in Beispiel 1, nur wurde hier die Menge des Hexamethylenimins um 50 % reduziert. Die Kristallisation erfolgte bei 140°C innerhalb von 96 Stunden.Approach as in Example 1, except that the amount of hexamethyleneimine was reduced by 50%. The crystallization took place at 140 ° C. within 96 hours.
Das kristalline Endprodukt wies hauptsächlich die Röntgendaten der Tabelle 1 auf, neben geringen Mengen an Quarz.The crystalline end product mainly had the X-ray data in Table 1, in addition to small amounts of quartz.
Ansatz wie in Beispiel 1, nur wurde hier die Menge des Al-Sulfats auf 15 g erhöht. Das kristalline Endprodukt wies nach der Calcinierung folgende oxidische Zusammensetzung auf: 0,2 Na20, A1203, 41 Si02.Approach as in Example 1, except that the amount of Al sulfate was increased to 15 g. After the calcination, the crystalline end product had the following oxidic composition: 0.2 Na 2 0, A1 2 0 3 , 41 Si0 2 .
Die Röntgendaten stimmten im wesentlichen mit denen der Tabelle 1 überein.The X-ray data were essentially the same as those in Table 1.
Die nachfolgenden Beispiele 7 bis 8 betreffen die Verwendung des nach den Beispielen 1-6 hergestellten Zeoliths PSH-3 als Katalysator für die Umwandlung von Methanol und/oder Dimethylether in Kohlenwasserstoffe.Examples 7 to 8 below relate to the use of the zeolite PSH-3 prepared according to Examples 1-6 as a catalyst for the conversion of methanol and / or dimethyl ether into hydrocarbons.
Ein nach Beispiel 1 hergestellter und nach Beispiel 2, dekationisierter Zeolith PSH-3 wurde mit 15 % Bentonit versetzt und mit Wasser zu einer extrudierbaren Masse verarbeitet. Nach Extrusion, Trocknung bei 120°C und Calcinierung bei 500°C wurden Katalsator-Formkörper erhalten. Die Ausprüfung dieses Katalysatormaterials erfolgte ohne Anwendung von Druck in einem elektrisch beheiztem Rohrreaktor (d = 20 mm, Katalysator-Füllhöhe 160 mm), der mit einer Vorheizzone sowie Einlässen für Stickstoff und Methanol versehen war.A zeolite PSH-3 prepared according to Example 1 and decationized according to Example 2 was mixed with 15% bentonite and processed with water to an extrudable mass. After extrusion, drying at 120 ° C. and calcination at 500 ° C., shaped catalyst bodies were obtained. This catalyst material was tested without applying pressure in an electrically heated tubular reactor (d = 20 mm, catalyst filling height 160 mm), which was provided with a preheating zone and inlets for nitrogen and methanol.
Der Katalysator wurde unter Stickstoff auf eine Reaktionstemperatur von 370°C gebracht und dann mit gasförmigem Methanol in einer Menge von 790 g pro Liter Katalysator und Stunde beaufschlagt.The catalyst was brought to a reaction temperature of 370 ° C. under nitrogen and then charged with gaseous methanol in an amount of 790 g per liter of catalyst per hour.
Der Methanolumsatz betrug bei diesem Versuch 94,5 Gew.-%. Neben Wasser und nichtumgesetztem Methanol wurde ein organisches Reaktionsprodukt der folgenden Zusammensetzung erhalten (Angaben in Gewichtsprozent):
Da der bei der Reaktion entstehende Dimethylether rezirkulierbar ist, errechnet sich für die Summe der C- Kohlenwasserstoffe eine Kohlenstoff-Selektivität von ca. 46 %. Unter Einbeziehung der C-Kohlenwassarstoffe erhöht sich dieser Wert auf ca. 84 %. Hierdurch wird offenbar, daß sich Zeolith PSH-3 in besonderer Weise für die Herstellung eines olefinreichen, aliphatischen Kohlenwasserstoffgemisches aus Methanol eignet.Since the dimethyl ether formed during the reaction can be recirculated, the sum of the C - Hydrocarbons have a carbon selectivity of approx. 46%. Including the C - Hydrocarbons this value increases to approx. 84%. This shows that zeolite PSH-3 is particularly suitable for the production of an olefin-rich, aliphatic hydrocarbon mixture from methanol.
Dieses Beispiel bezieht sich auf die Umwandlung einer Methanol/Dimethylether/Wasser-Gleichgewichtsmis-hung in überwiegend aliphatisch-olefinische Kohlenwasserstoffe. Ein Strom von 50 ml Methanol pro Stunde wurde in ver- dampfter Form ohne Anwendung von Druck bei einer Temperatur von 280°C über eine Katalysatorschüttung bestehend aus 100 ml gefälltem und granuliertem Aluminiumoxid geleitet. Aus der Literatur ist bekannt, daß Aluminiumoxid die Umsetzung von Methanol zu Dimethylether katalysiert (siehe beispielsweise J.B. Senderens, Ann. Chim. Phys. 25, 509 (1912). Das den Reaktor verlassende Produktgemisch wurde ohne weitere Trennung über einen nach Beispiel 1 und 2 hergestellten und mit einem Zusatz von 20 % Montigel nach bekannten Techniken granulierten Katalysator auf Basis Zeolith PSH-3 geleitet.This example relates to the conversion of a methanol / dimethyl ether / water equilibrium mixture into predominantly aliphatic-olefinic hydrocarbons. A stream of 50 ml of methanol per hour was passed in vaporized form without applying pressure at a temperature of 280 ° C. over a bed of catalyst consisting of 100 ml of precipitated and granulated aluminum oxide. It is known from the literature that aluminum oxide catalyzes the conversion of methanol to dimethyl ether (see, for example, JB Senderens, Ann. Chim. Phys. 25, 509 (1912). The product mixture leaving the reactor was passed through a mixture according to Examples 1 and 2 without further separation prepared and granulated with an addition of 20% Montigel according to known techniques based on zeolite PSH-3.
Die Kontaktbelastung betrug 800 g Einsatz pro Liter Katalysator und Stunde; die Umwandlungstemperatur betrug 370°C.The contact load was 800 g insert per liter catalyst and hour; the transition temperature was 370 ° C.
Der den Reaktor verlassende Produktstrom setzte sich wie folgt zusammen (ohne Berücksichtigung des Reaktionswassers):
Im C-Anteil können auch kleine Anteile sauerstoffhaltiger Verbindungen enthalten sein.
Zur Herstellung eines Katalysators zur Kohlenoxidhydrierung wurde eine Suspension aus 18,4 g des erfindungsgemäßen Alumosilicats in 250 ml einer wäßrigen Lösung mit 18,4 g Kaliwasserglas und 70,1 g Fe(N03)3 x 9 H20 in der Siedehitze mit 160 ml 11 %-iger Ammoniaklösung versetzt. Der Niederschlag wurde abfiltriert, mit Wasser neutral gewaschen und 4 h bei 150°C im Vakuum (20 Torr) getrocknet. Nach Mahlung wurde die Masse mit Graphit und Bentonit zu zylindrischen 5 x 5 mm-Tabletten gepreßt. Unter der Annahme vollständiger Ausfällung des Eisens und quantitativer Sorption des Kaliwasserglases durch den Niederschlag berechnet man folgende Katalysatorzusammensetzung:
Claims (9)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3117135 | 1981-04-30 | ||
| DE19813117135 DE3117135A1 (en) | 1981-04-30 | 1981-04-30 | CRYSTALLINE ALUMOSILICATE, METHOD FOR THE PRODUCTION THEREOF AND THE USE THEREOF FOR CATALYTICALLY CONVERTING METHANOL AND / OR DIMETHYL ETHER IN HYDROCARBONS |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0064205A1 true EP0064205A1 (en) | 1982-11-10 |
| EP0064205B1 EP0064205B1 (en) | 1984-12-05 |
Family
ID=6131162
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP82103256A Expired EP0064205B1 (en) | 1981-04-30 | 1982-04-19 | Crystalline aluminosilicate, process for producing it and its use in converting methanol and/or dimethyl ether into hydrocarbons |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US4439409A (en) |
| EP (1) | EP0064205B1 (en) |
| JP (1) | JPS57183321A (en) |
| AU (1) | AU8306482A (en) |
| CA (1) | CA1174219A (en) |
| DE (2) | DE3117135A1 (en) |
| ZA (1) | ZA822922B (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1987004368A1 (en) * | 1986-01-24 | 1987-07-30 | Haldor Topso^/E A/S | Crystalline aluminosilicate and a method for its preparation |
| WO1991014753A1 (en) * | 1990-03-19 | 1991-10-03 | Mobil Oil Corporation | Catalytic hydroisomerization process |
| WO1992006921A1 (en) * | 1990-10-17 | 1992-04-30 | Mobil Oil Corporation | Synthetic porous crystalline material, its synthesis and use |
| AU639503B2 (en) * | 1990-10-17 | 1993-07-29 | Mobil Oil Corporation | Synthetic porous crystalline material, its synthesis and use |
| WO2000063117A1 (en) * | 1999-04-16 | 2000-10-26 | Centre National De La Recherche Scientifique | Irc1 zeolite and methods for preparing same |
Families Citing this family (410)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4481177A (en) * | 1982-12-09 | 1984-11-06 | Mobil Oil Corporation | Synthesis of zeolite ZSM-22 with a heterocyclic organic compound |
| NZ206332A (en) * | 1982-12-15 | 1986-07-11 | Mobil Oil Corp | Catalytic conversion of methanol and/or methyl ethyl ether to light olefins using zsm-45 |
| US4496786A (en) * | 1983-09-30 | 1985-01-29 | Chevron Research Company | Selective conversion of methanol to low molecular weight olefins over high silica SSZ-13 zeolite |
| US5202014A (en) * | 1986-01-29 | 1993-04-13 | Chevron Research And Technology Company | Zeolite SSZ-25 |
| US5421992A (en) * | 1987-02-17 | 1995-06-06 | Chevron U.S.A. Inc. | Hydrocarbon conversion process using zeolite SSZ-25 |
| US5019664A (en) * | 1988-10-06 | 1991-05-28 | Mobil Oil Corp. | Process for the conversion of paraffins to olefins and/or aromatics and low acidity zeolite catalyst therefor |
| US5039640A (en) * | 1986-07-29 | 1991-08-13 | Mobil Oil Corp. | Catalyst composition for octane improvement in catalytic cracking |
| US4954325A (en) * | 1986-07-29 | 1990-09-04 | Mobil Oil Corp. | Composition of synthetic porous crystalline material, its synthesis and use |
| US5072054A (en) * | 1986-07-29 | 1991-12-10 | Mobil Oil Corp. | Process for preparing short chain alkyl phenols |
| US5019670A (en) * | 1986-07-29 | 1991-05-28 | Mobil Oil Corporation | Process for producing alkylaromatic lubricant fluids |
| US4962256A (en) * | 1988-10-06 | 1990-10-09 | Mobil Oil Corp. | Process for preparing long chain alkyl aromatic compounds |
| US5107049A (en) * | 1986-07-29 | 1992-04-21 | Mobil Oil Corporation | Stabilization of polyalpha-olefins |
| US4954663A (en) * | 1988-10-06 | 1990-09-04 | Mobil Oil Corp. | Process for preparing long chain alkyl phenols |
| US4956514A (en) * | 1988-10-06 | 1990-09-11 | Mobil Oil Corp. | Process for converting olefins to higher hydrocarbons |
| US5013422A (en) * | 1986-07-29 | 1991-05-07 | Mobil Oil Corp. | Catalytic hydrocracking process |
| US4983276A (en) * | 1988-10-06 | 1991-01-08 | Mobil Oil Corp. | Octane improvement in catalytic cracking and cracking catalyst composition therefor |
| US5105023A (en) * | 1987-12-30 | 1992-04-14 | Mobil Oil Corp. | Process for the hydration of olefins cross reference to related applications |
| US4992606A (en) * | 1988-10-06 | 1991-02-12 | Mobil Oil Corp. | Process for preparing short chain alkyl aromatic compounds |
| US4973784A (en) * | 1988-10-06 | 1990-11-27 | Mobil Oil Corp. | Process for reducing the durene content of effluent resulting from the catalytic conversion of C1 -C4 oxygenates to gasoline |
| US4982033A (en) * | 1988-10-06 | 1991-01-01 | Mobil Oil Corp. | Process for converting light aliphatics to aromatics |
| US5001283A (en) * | 1988-10-06 | 1991-03-19 | Mobil Oil Corp. | Process for preparing aromatic alkanols |
| US4962239A (en) * | 1988-10-06 | 1990-10-09 | Mobil Oil Corp. | Process for preparing ethers |
| US5012033A (en) * | 1988-10-06 | 1991-04-30 | Mobil Oil Corp. | Isoparaffin-olefin alkylation process and catalyst composition thereof |
| US5001295A (en) * | 1988-10-06 | 1991-03-19 | Mobil Oil Corp. | Process for preparing dialkylnaphthalene |
| US4962257A (en) * | 1988-10-06 | 1990-10-09 | Mobil Oil Corp. | Process for the catalytic disproportionation of toluene |
| US4982040A (en) * | 1988-10-06 | 1991-01-01 | Mobil Oil Corp. | Process for the catalytic disproportionation of methylnaphthalenes |
| US5073655A (en) * | 1988-10-06 | 1991-12-17 | Mobil Oil Corp. | Method for preparing diarylalkanes |
| US5030785A (en) * | 1988-10-06 | 1991-07-09 | Mobil Oil Corp. | Process for preparing long chain alkyl aromatic compounds employing Lewis acid-promoted zeolite catalysts |
| JP2754063B2 (en) * | 1988-11-28 | 1998-05-20 | モービル オイル コーポレーシヨン | Synthetic porous crystalline material, its synthesis and use |
| US4927977A (en) * | 1989-01-12 | 1990-05-22 | Mobil Oil Corporation | Process and apparatus for the uniform distribution of a two-phase feed in a chemical reaction zone |
| US4935552A (en) * | 1989-01-12 | 1990-06-19 | Mobil Oil Corporation | Dual stage process for the production of ethers |
| US5144084A (en) * | 1989-01-12 | 1992-09-01 | Mobil Oil Corporation | Process for the conversion of olefins to alcohols and/or ethers |
| US4925548A (en) * | 1989-07-13 | 1990-05-15 | Mobil Oil Corp. | Synthesis of crystalline ZSM-35 structure |
| US4992611A (en) * | 1989-12-13 | 1991-02-12 | Mobil Oil Corp. | Direct conversion of C1 -C4 oxygenates to low aromatic distillate range hydrocarbons |
| US5030787A (en) * | 1990-01-24 | 1991-07-09 | Mobil Oil Corp. | Catalytic disproportionation/transalkylation utilizing a C9+ aromatics feed |
| US4962250A (en) * | 1990-01-24 | 1990-10-09 | Mobile Oil Corp. | Process for the conversion of paraffins to olefins and/or aromatics and non-acidic zeolite catalyst therefor |
| WO1991011412A1 (en) * | 1990-01-25 | 1991-08-08 | Mobil Oil Corporation | Isoparaffin-olefin alkylation process |
| US5220101A (en) * | 1990-01-25 | 1993-06-15 | Mobil Oil Corp. | Sorption separation over modified synthetic mesoporous crystalline material |
| US5145816A (en) * | 1990-12-10 | 1992-09-08 | Mobil Oil Corporation | Method for functionalizing synthetic mesoporous crystalline material |
| US5378440A (en) * | 1990-01-25 | 1995-01-03 | Mobil Oil Corp. | Method for separation of substances |
| US4992615A (en) * | 1990-01-25 | 1991-02-12 | Mobil Oil Corp. | Isoparaffin-olefin alkylation process |
| US4968402A (en) * | 1990-02-14 | 1990-11-06 | Mobil Oil Corp. | Process for upgrading hydrocarbons |
| US5001296A (en) * | 1990-03-07 | 1991-03-19 | Mobil Oil Corp. | Catalytic hydrodealkylation of aromatics |
| US5043513A (en) * | 1990-03-07 | 1991-08-27 | Mobil Oil Corp. | Catalytic hydrodealkylation of aromatics |
| US5043501A (en) * | 1990-03-15 | 1991-08-27 | Mobil Oil Corp. | Process for preparing dimethylnaphthalene |
| US5334795A (en) * | 1990-06-28 | 1994-08-02 | Mobil Oil Corp. | Production of ethylbenzene |
| US5085762A (en) * | 1990-07-05 | 1992-02-04 | Mobil Oil Corporation | Catalytic cracking of hydrocarbons |
| US5073665A (en) * | 1990-07-31 | 1991-12-17 | Mobil Oil Corp. | Process for alkylating olefins and isoparaffins in a fixed bed reactor |
| US5107054A (en) * | 1990-08-23 | 1992-04-21 | Mobil Oil Corporation | Zeolite MCM-22 based catalyst for paraffin isomerization |
| US5107047A (en) * | 1990-08-23 | 1992-04-21 | Mobil Oil Corporation | Zeolite MCM-22 catalysts for olefin isomerization |
| US5021141A (en) * | 1990-09-14 | 1991-06-04 | Mobil Oil Corp. | Synthesis of crystalline ZSM-12 type structure |
| US5364999A (en) * | 1991-01-11 | 1994-11-15 | Mobil Oil Corp. | Organic conversion with a catalyst comprising a crystalline pillared oxide material |
| US5250277A (en) * | 1991-01-11 | 1993-10-05 | Mobil Oil Corp. | Crystalline oxide material |
| US5229341A (en) * | 1991-01-11 | 1993-07-20 | Mobil Oil Corp. | Crystalline oxide material |
| US5296428A (en) * | 1991-01-11 | 1994-03-22 | Mobil Oil Corp. | Catalyst comprising MCM-36 and a hydrogenation/dehydrogenation component |
| US5310715A (en) * | 1991-01-11 | 1994-05-10 | Mobil Oil Corp. | Method for preparing a pillared layered oxide material |
| US5137705A (en) * | 1991-04-08 | 1992-08-11 | Mobil Oil Corp. | Synthesis of crystalline silicate ZSM-12 |
| US5068096A (en) * | 1991-04-08 | 1991-11-26 | Mobil Oil Corporation | Synthesis of crystalline silicate MCM-47 |
| US5236575A (en) * | 1991-06-19 | 1993-08-17 | Mobil Oil Corp. | Synthetic porous crystalline mcm-49, its synthesis and use |
| US5220089A (en) * | 1991-06-21 | 1993-06-15 | Mobil Oil Corporation | Olefin upgrading by selective catalysis |
| US5174980A (en) * | 1991-10-04 | 1992-12-29 | Mobil Oil Corp. | Synthesis of crystalline ZSM-35 |
| US5174977A (en) * | 1991-10-04 | 1992-12-29 | Mobil Oil Corp. | Synthesis of crystalline ZSM-5-type material |
| US5174981A (en) * | 1991-10-04 | 1992-12-29 | Mobil Oil Corp. | Synthesis of crystalline ZSM-5-type material |
| US5209918A (en) * | 1991-10-04 | 1993-05-11 | Mobil Oil Corp. | Synthesis of crystalline ZSM-5-type material |
| US5174978A (en) * | 1991-10-04 | 1992-12-29 | Mobil Oil Corp. | Synthesis of crystalline ZSM-5-type material |
| US5173281A (en) * | 1991-10-07 | 1992-12-22 | Mobil Oil Corp. | Synthesis of a synthetic porous crystalline material |
| US5254792A (en) * | 1991-10-10 | 1993-10-19 | Mobil Oil Corporation | Isoparaffin:olefin alkylation in the presence of synthetic porous MCM-49 |
| US5192521A (en) * | 1991-11-19 | 1993-03-09 | Mobil Oil Corp. | Synthesis of crystalline silicate ZSM-12 |
| US5213786A (en) * | 1991-12-20 | 1993-05-25 | Mobil Oil Corp. | Synthesis of crystalline silicate ZSM-11 |
| US5258565A (en) * | 1992-05-04 | 1993-11-02 | Mobil Oil Corp. | Process for preparing short chain alkylaromatic compounds |
| US5278115A (en) * | 1992-05-04 | 1994-01-11 | Mobil Oil Corp. | Method for preparing a pillared layered oxide material |
| US5278116A (en) * | 1992-05-04 | 1994-01-11 | Mobil Oil Corp. | Method for preparing a pillared layered oxide material |
| US5258566A (en) * | 1992-05-04 | 1993-11-02 | Mobil Oil Corp. | Process for preparing long chain alkylaromatic compounds |
| US5231233A (en) * | 1992-05-04 | 1993-07-27 | Mobil Oil Corp. | Process for the hydration of olefins |
| US5231235A (en) * | 1992-06-01 | 1993-07-27 | Mobil Oil Corp. | Process for preparing ethers |
| US5294330A (en) * | 1992-08-12 | 1994-03-15 | Mobil Oil Corp. | Hydrocracking process with a catalyst comprising MCM-36 |
| US5326922A (en) * | 1992-08-13 | 1994-07-05 | Mobil Oil Corp. | Hydrogen transfer process |
| US5258569A (en) * | 1992-08-13 | 1993-11-02 | Mobil Oil Corp. | Isoparaffin-olefin alkylation process with zeolite MCM-36 |
| US5264643A (en) * | 1992-12-09 | 1993-11-23 | Mobil Oil Corp. | Process for converting olefins to higher hydrocarbons |
| US5284643A (en) * | 1992-12-10 | 1994-02-08 | Mobil Oil Corp. | Gallium-containing zeolite MCM-22 |
| US5827491A (en) * | 1993-04-26 | 1998-10-27 | Mobil Oil Corporation | Process for preparing the synthetic porous crystalline material MCM-56 |
| US5362697A (en) * | 1993-04-26 | 1994-11-08 | Mobil Oil Corp. | Synthetic layered MCM-56, its synthesis and use |
| CA2165427A1 (en) * | 1993-06-16 | 1994-12-17 | Jane Chi-Ya Cheng | Liquid phase ethylbenzene synthesis |
| US5329059A (en) * | 1993-07-06 | 1994-07-12 | Mobil Oil Corp. | Alkylaromatic disproportionation |
| US5488194A (en) * | 1994-05-16 | 1996-01-30 | Mobil Oil Corp. | Selective production of para-dialkyl substituted benzenes and catalyst therefor |
| US5900520A (en) * | 1995-01-23 | 1999-05-04 | Mobil Oil Corporation | Aromatics alkylation |
| ES2105982B1 (en) * | 1995-11-23 | 1998-07-01 | Consejo Superior Investigacion | ZEOLITE ITQ-1 |
| US5942651A (en) * | 1997-06-13 | 1999-08-24 | Mobile Oil Corporation | Process for converting C9 + aromatic hydrocarbons to lighter aromatic products by transalkylation in the prescence of two zeolite-containing catalysts |
| US6037513A (en) * | 1998-07-09 | 2000-03-14 | Mobil Oil Corporation | Hydroalkylation of aromatic hydrocarbons |
| DE60011814T2 (en) * | 1999-09-20 | 2005-07-07 | Consejo Superior de Investigaciónes Científicas | ALKYLATION OF AROMATES |
| US6797155B1 (en) | 1999-12-21 | 2004-09-28 | Exxonmobil Research & Engineering Co. | Catalytic cracking process using a modified mesoporous aluminophosphate material |
| US6489529B1 (en) | 2000-01-24 | 2002-12-03 | Exxonmobil Oil Corporation | Production of monocycloalkyl aromatic compounds |
| US6281406B1 (en) | 2000-03-31 | 2001-08-28 | Chevron Corporation | Adsorption process for paraxylene purifacation using Cs SSZ-25 adsorbent with benzene desorbent |
| US6548718B2 (en) | 2000-04-27 | 2003-04-15 | Shell Oil Company | Process for catalytic hydroxylation of, saturated or unsaturated, aliphatic compounds |
| US6437197B1 (en) | 2000-04-27 | 2002-08-20 | Shell Oil Company | Process for catalytic hydroxylation of aromatic hydrocarbons |
| US6936744B1 (en) | 2000-07-19 | 2005-08-30 | Exxonmobil Chemical Patents, Inc. | Alkylaromatics production |
| US6641714B2 (en) | 2000-07-21 | 2003-11-04 | Exxonmobil Research And Engineering Company | Hydrocarbon upgrading process |
| US6617482B1 (en) * | 2000-08-16 | 2003-09-09 | Exxonmobil Chemical Patents Inc. | Removable of polar contaminants from aromatic feedstocks |
| US6670517B1 (en) | 2000-08-24 | 2003-12-30 | Exxon Mobil Chemical Patents Inc. | Process for alkylating aromatics |
| ATE429412T1 (en) | 2000-09-25 | 2009-05-15 | Exxonmobil Chem Patents Inc | HYDROGENATION OF DECISION PRODUCTS IN PHENOL PRODUCTION |
| US6525234B1 (en) * | 2000-11-21 | 2003-02-25 | Exxonmobil Oil Corporation | Process for liquid phase aromatics alkylation comprising in-situ catalyst reactivation with polar compounds |
| EP1358141B8 (en) * | 2001-02-07 | 2008-02-13 | ExxonMobil Chemical Patents Inc. | Production of alkylaromatic compounds |
| EP1894909B1 (en) | 2001-02-07 | 2017-04-05 | Badger Licensing LLC | Production of alkylaromatic compounds |
| US6933419B2 (en) * | 2001-04-27 | 2005-08-23 | Exxonmobil Oil Corporation | Production of diisopropylbenzene |
| US7038100B2 (en) * | 2001-04-30 | 2006-05-02 | Exxonmobil Chemical Patents, Inc. | Aromatics alkylation |
| US7838708B2 (en) | 2001-06-20 | 2010-11-23 | Grt, Inc. | Hydrocarbon conversion process improvements |
| US6518471B1 (en) * | 2001-06-25 | 2003-02-11 | Exxonmobil Chemical Patents Inc. | Selective production of meta-diisopropylbenzene |
| US20020042548A1 (en) * | 2001-07-11 | 2002-04-11 | Dandekar Ajit B. | Process for producing cumene |
| US6781025B2 (en) | 2001-07-11 | 2004-08-24 | Exxonmobil Chemical Patents Inc. | Reactivation of aromatics alkylation catalysts |
| US6878654B2 (en) * | 2001-07-11 | 2005-04-12 | Exxonmobil Chemical Patents Inc. | Reactivation of aromatics alkylation catalysts |
| JP3891827B2 (en) * | 2001-11-07 | 2007-03-14 | 本田技研工業株式会社 | Exhaust gas purification catalyst |
| WO2003082781A1 (en) * | 2002-03-29 | 2003-10-09 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization process |
| US7622621B2 (en) * | 2002-03-29 | 2009-11-24 | Exxonmobil Chemical Patents Inc. | Preparation of alkylaromatic hydrocarbons and alkylaryl sulfonates |
| US7148388B2 (en) * | 2002-03-29 | 2006-12-12 | Exxonmobil Chemical Patents Inc. | Process for preparing and olefinic hydrocarbon mixture |
| US6995295B2 (en) * | 2002-09-23 | 2006-02-07 | Exxonmobil Chemical Patents Inc. | Alkylaromatics production |
| US7148391B1 (en) * | 2002-11-14 | 2006-12-12 | Exxonmobil Chemical Patents Inc. | Heavy aromatics processing |
| US6753453B2 (en) | 2002-11-19 | 2004-06-22 | Exxonmobil Chemical Patents Inc. | Production of meta-diisopropylbenzene |
| US7019185B2 (en) * | 2002-12-06 | 2006-03-28 | Exxonmobil Chemical Patents Inc. | Aromatic alkylation process |
| WO2005021468A1 (en) | 2003-07-15 | 2005-03-10 | Grt, Inc. | Hydrocarbon synthesis |
| US20050171393A1 (en) | 2003-07-15 | 2005-08-04 | Lorkovic Ivan M. | Hydrocarbon synthesis |
| US8642822B2 (en) | 2004-04-16 | 2014-02-04 | Marathon Gtf Technology, Ltd. | Processes for converting gaseous alkanes to liquid hydrocarbons using microchannel reactor |
| US20080275284A1 (en) | 2004-04-16 | 2008-11-06 | Marathon Oil Company | Process for converting gaseous alkanes to liquid hydrocarbons |
| US8173851B2 (en) | 2004-04-16 | 2012-05-08 | Marathon Gtf Technology, Ltd. | Processes for converting gaseous alkanes to liquid hydrocarbons |
| US7674941B2 (en) | 2004-04-16 | 2010-03-09 | Marathon Gtf Technology, Ltd. | Processes for converting gaseous alkanes to liquid hydrocarbons |
| US20060100469A1 (en) | 2004-04-16 | 2006-05-11 | Waycuilis John J | Process for converting gaseous alkanes to olefins and liquid hydrocarbons |
| US7244867B2 (en) | 2004-04-16 | 2007-07-17 | Marathon Oil Company | Process for converting gaseous alkanes to liquid hydrocarbons |
| ES2246704B1 (en) * | 2004-05-28 | 2007-06-16 | Universidad Politecnica De Valencia | ZEOLITA ITQ-30. |
| KR101015754B1 (en) | 2005-03-31 | 2011-02-22 | 바져 라이센싱 엘엘씨 | Multiphase alkylaromatics production |
| US8057664B2 (en) * | 2005-11-17 | 2011-11-15 | Exxonmobil Chemical Patents, Inc. | Process for reducing bromine index of hydrocarbon feedstocks |
| US7744750B2 (en) * | 2005-11-17 | 2010-06-29 | Exxonmobil Chemical Patents Inc. | Process for reducing Bromine Index of hydrocarbon feedstocks |
| US7517824B2 (en) * | 2005-12-06 | 2009-04-14 | Exxonmobil Chemical Company | Process for steam stripping hydrocarbons from a bromine index reduction catalyst |
| US7425659B2 (en) * | 2006-01-31 | 2008-09-16 | Exxonmobil Chemical Patents Inc. | Alkylaromatics production |
| MY153701A (en) | 2006-02-03 | 2015-03-13 | Grt Inc | Separation of light gases from halogens |
| EA020442B1 (en) | 2006-02-03 | 2014-11-28 | ДжиАрТи, ИНК. | Process for converting hydrocarbon feedstock (embodiments) and system therefor |
| EP1996327A1 (en) * | 2006-02-14 | 2008-12-03 | ExxonMobil Chemical Patents Inc. | A molecular sieve composition |
| US8557219B2 (en) | 2006-02-14 | 2013-10-15 | Exxonmobil Chemical Patents Inc. | High throughput process for manufacturing molecular sieves |
| CN102527428B (en) * | 2006-02-14 | 2014-11-26 | 埃克森美孚化学专利公司 | Method of preparing a molecular sieve composition |
| CN101384367B (en) * | 2006-02-14 | 2012-05-30 | 埃克森美孚化学专利公司 | A process for manufacturing mcm-22 family molecular sieves |
| WO2007094938A1 (en) * | 2006-02-14 | 2007-08-23 | Exxonmobil Chemical Patents Inc. | An mcm-22 family molecular sieve composition |
| CN101384361A (en) * | 2006-02-14 | 2009-03-11 | 埃克森美孚化学专利公司 | Process for manufacturing molecular sieve of mfs framework type and its use |
| US8859836B2 (en) | 2006-02-14 | 2014-10-14 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion process using molecular sieve of MFS framework type |
| CN101437611B (en) | 2006-05-08 | 2015-04-22 | 埃克森美孚化学专利公司 | Improved catalyst composition |
| CN101437783B (en) * | 2006-05-08 | 2012-12-12 | 埃克森美孚化学专利公司 | Translation method for organic compound |
| US7501547B2 (en) * | 2006-05-10 | 2009-03-10 | Exxonmobil Chemical Patents Inc. | Alkylaromatics production |
| WO2007139629A1 (en) | 2006-05-24 | 2007-12-06 | Exxonmobil Chemical Patents Inc. | Monoalkylated aromatic compound production |
| US7842277B2 (en) * | 2006-07-28 | 2010-11-30 | Exxonmobil Chemical Patents Inc. | Molecular sieve composition, a method of making and a process of using the same |
| US7910785B2 (en) * | 2006-07-28 | 2011-03-22 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion process using EMM-10 family molecular sieve |
| KR101120880B1 (en) | 2006-07-28 | 2012-02-27 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Molecular sieve compositionemm-10-p, its method of making, and use for hydrocarbon conversions |
| US7959899B2 (en) | 2006-07-28 | 2011-06-14 | Exxonmobil Chemical Patents Inc. | Molecular sieve composition (EMM-10-P), its method of making, and use for hydrocarbon conversions |
| JP5571950B2 (en) | 2006-07-28 | 2014-08-13 | エクソンモービル・ケミカル・パテンツ・インク | Molecular sieve composition (EMM-10), production method thereof, and hydrocarbon conversion method using the composition |
| CN101489677B (en) | 2006-07-28 | 2012-12-26 | 埃克森美孚化学专利公司 | A mcm-22 family molecular sieve composition, its method of making, and use for hydrocarbon conversions |
| CN100554156C (en) | 2006-08-11 | 2009-10-28 | 中国石油化工股份有限公司 | Organic silicon micro-pore zeolite, synthetic method and application thereof |
| ES2418955T3 (en) | 2007-02-09 | 2013-08-19 | Exxonmobil Chemical Patents Inc. A Corporation Of The State Of Delaware | Improved alkylaromatic production procedure |
| US7737314B2 (en) | 2007-02-12 | 2010-06-15 | Exxonmobil Chemical Patents Inc. | Production of high purity ethylbenzene from non-extracted feed and non-extracted reformate useful therein |
| US20080253959A1 (en) * | 2007-03-30 | 2008-10-16 | Johnson Ivy D | Synthesis crystalline molecular sieves |
| NZ581216A (en) * | 2007-05-24 | 2011-06-30 | Grt Inc | Zone reactor incorporating reversible hydrogen halide capture and release |
| US8816145B2 (en) | 2007-06-21 | 2014-08-26 | Exxonmobil Chemical Patents Inc. | Liquid phase alkylation process |
| US7790940B2 (en) | 2007-06-21 | 2010-09-07 | Exxonmobil Chemical Patents Inc. | Liquid phase alkylation process |
| US7910778B2 (en) * | 2007-08-25 | 2011-03-22 | Exxonmobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| EP2080746A1 (en) | 2008-01-21 | 2009-07-22 | ExxonMobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| KR101175849B1 (en) * | 2007-09-21 | 2012-08-24 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Process for producing cyclohexylbenzene |
| KR101151978B1 (en) * | 2007-09-21 | 2012-06-01 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Process for producing cyclohexylbenzene |
| EP2098498A1 (en) | 2008-03-04 | 2009-09-09 | ExxonMobil Chemical Patents Inc. | Selective oligomerization of isobutene |
| US8398955B2 (en) * | 2007-10-26 | 2013-03-19 | Exxonmobil Chemical Patents Inc. | Method of preparing a molecular sieve composition |
| US7786338B2 (en) * | 2007-10-26 | 2010-08-31 | Exxonmobil Research And Engineering Company | Selective oligomerization of isobutene |
| WO2011031374A2 (en) | 2009-08-28 | 2011-03-17 | Exxonmobil Chemical Patents Inc. | Oxidation of hydrocarbons |
| ES2915260T3 (en) * | 2007-11-02 | 2022-06-21 | Badger Licensing Llc | A process for rejuvenating a catalyst composition |
| EP2214819A2 (en) * | 2007-11-16 | 2010-08-11 | ExxonMobil Chemical Patents Inc. | Catalyst regeneration process |
| ES2392015T3 (en) * | 2008-02-12 | 2012-12-03 | Exxonmobil Chemical Patents Inc. | Procedure to produce cyclohexylbenzene |
| EP2098504A1 (en) | 2008-03-06 | 2009-09-09 | ExxonMobil Chemical Patents Inc. | Oxidation of hydrocarbons |
| EP2098505A1 (en) | 2008-03-06 | 2009-09-09 | ExxonMobil Chemical Patents Inc. | Oxidation of hydrocarbons |
| EP2103585A1 (en) | 2008-03-21 | 2009-09-23 | ExxonMobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| WO2009128984A1 (en) | 2008-04-14 | 2009-10-22 | Exxonmobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| US8106243B2 (en) * | 2008-05-01 | 2012-01-31 | Exxonmobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| US8282810B2 (en) | 2008-06-13 | 2012-10-09 | Marathon Gtf Technology, Ltd. | Bromine-based method and system for converting gaseous alkanes to liquid hydrocarbons using electrolysis for bromine recovery |
| AU2009270801B2 (en) | 2008-07-18 | 2014-04-24 | Reaction 35, Llc | Continuous process for converting natural gas to liquid hydrocarbons |
| WO2010011451A1 (en) * | 2008-07-22 | 2010-01-28 | Exxonmobil Chemical Patents Inc. | Preparation of molecular sieve containing catalyst and its use in the production of alkylaromatic hydrocarbons |
| CN102216215B (en) * | 2008-07-28 | 2014-03-26 | 埃克森美孚化学专利公司 | Process of making alkylaromatics using emm-12 |
| US8704023B2 (en) * | 2008-07-28 | 2014-04-22 | Exxonmobil Chemical Patents Inc. | Molecular sieve composition EMM-13, a method of making and a process of using the same |
| US8466333B2 (en) | 2008-07-28 | 2013-06-18 | Exxonmobil Chemical Patents Inc. | Process of making alkylaromatics using EMM-12 |
| KR101273039B1 (en) * | 2008-07-28 | 2013-06-10 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | A novel molecular sieve composition emm-12, a method of making and a process of using the same |
| US8212096B2 (en) | 2008-07-28 | 2012-07-03 | Exxonmobil Chemical Patents Inc. | Hydroalkylation of aromatic compounds using EMM-13 |
| RU2509054C2 (en) * | 2008-07-28 | 2014-03-10 | Эксонмобил Кемикэл Пейтентс Инк. | Method of producing alkyl aromatic compounds using emm-13 |
| TWI458695B (en) * | 2008-10-06 | 2014-11-01 | Badger Licensing Llc | Process for producing cumene |
| BRPI0920691A2 (en) * | 2008-10-06 | 2015-12-29 | Badger Licensing Llc | process for cumene production |
| KR101343332B1 (en) * | 2008-10-10 | 2013-12-20 | 바져 라이센싱 엘엘씨 | Process for producing alkylaromatic compounds |
| JP5599812B2 (en) * | 2008-11-18 | 2014-10-01 | インヴィスタ テクノロジーズ エスアエルエル | 1,2-diaminocyclohexane and chemical methods |
| US8519194B2 (en) | 2009-02-26 | 2013-08-27 | Exxonmobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| US8921603B2 (en) | 2009-02-26 | 2014-12-30 | Exxonmobil Chemical Patents Inc. | Cyclohexanone compositions |
| US8884067B2 (en) | 2009-02-26 | 2014-11-11 | Exxonmobil Chemical Patents Inc. | Phenol and cyclohexanone mixtures |
| US8372946B2 (en) * | 2009-04-15 | 2013-02-12 | Invista North America S.A R.L. | Copolyether glycol manufacturing process |
| US8551192B2 (en) * | 2009-04-15 | 2013-10-08 | Invista North America S.A.R.L. | Miscibility of otherwise immiscible compounds |
| JP5486083B2 (en) | 2009-05-26 | 2014-05-07 | エクソンモービル ケミカル パテンツ インコーポレイテッド | Transalkylation of polycyclohexylbenzene. |
| US9169181B2 (en) | 2009-09-17 | 2015-10-27 | Exxonmobil Chemical Patents Inc. | Production of cyclohexylbenzene hydroperoxide |
| WO2011061204A1 (en) * | 2009-11-18 | 2011-05-26 | Shell Internationale Research Maatschappij B.V. | Catalyst and isomerisation process |
| US8921609B2 (en) | 2009-11-25 | 2014-12-30 | Exxonmobil Chemical Patents Inc. | Oxidation of hydrocarbons |
| CN102652132B (en) | 2009-12-11 | 2015-03-25 | 因温斯特技术公司 | Depolymerization of oligomeric cyclic ethers |
| US9029612B2 (en) | 2010-01-25 | 2015-05-12 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| WO2012145031A2 (en) | 2011-04-19 | 2012-10-26 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US9321710B2 (en) | 2010-01-25 | 2016-04-26 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| CN102741201A (en) | 2010-02-05 | 2012-10-17 | 埃克森美孚化学专利公司 | Dehydrogenation process |
| US9242227B2 (en) | 2010-02-05 | 2016-01-26 | Exxonmobil Chemical Patents Inc. | Dehydrogenation catalyst and process |
| CN102711988B (en) | 2010-02-05 | 2015-11-25 | 埃克森美孚化学专利公司 | Containing iridium catalyst, their preparation and purposes |
| US20120271078A1 (en) | 2010-02-05 | 2012-10-25 | Exxonmobil Chemical Patents Inc. | Dehydrogenation Process |
| CN102791658B (en) | 2010-02-05 | 2016-03-02 | 埃克森美孚化学专利公司 | Method of dehydrogenating |
| SG10201501677SA (en) | 2010-02-05 | 2015-05-28 | Exxonmobil Chem Patents Inc | Dehydrogenation of cyclohexanone to produce phenol |
| WO2011096989A1 (en) | 2010-02-05 | 2011-08-11 | Exxonmobil Chemical Patents Inc. | Dehydrogenation of cyclohexanone to produce phenol |
| US8969639B2 (en) | 2010-02-05 | 2015-03-03 | Exxonmobil Chemical Patents Inc. | Dehydrogenation process |
| US8658834B2 (en) | 2010-02-12 | 2014-02-25 | Exxonmobil Chemical Patents Inc. | Hydrogenation process |
| US8367884B2 (en) | 2010-03-02 | 2013-02-05 | Marathon Gtf Technology, Ltd. | Processes and systems for the staged synthesis of alkyl bromides |
| US8198495B2 (en) | 2010-03-02 | 2012-06-12 | Marathon Gtf Technology, Ltd. | Processes and systems for the staged synthesis of alkyl bromides |
| US8629311B2 (en) | 2010-03-10 | 2014-01-14 | Stone & Webster, Inc. | Alkylated aromatics production |
| US8853482B2 (en) | 2010-03-19 | 2014-10-07 | Exxonmobil Chemical Patents Inc. | Dealkylation process |
| SG184865A1 (en) | 2010-05-20 | 2012-11-29 | Exxonmobil Chem Patents Inc | Hydroalkylation processes |
| WO2012030440A1 (en) | 2010-08-30 | 2012-03-08 | Exxonmobil Chemical Patents Inc. | Improved alkylation process |
| KR101748068B1 (en) | 2010-09-14 | 2017-06-15 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Processes for producing phenol |
| SG10201509483QA (en) | 2010-09-14 | 2015-12-30 | Exxonmobil Chem Patents Inc | Oxidation of cyclohexylbenzene |
| SG187647A1 (en) | 2010-09-14 | 2013-03-28 | Exxonmobil Chem Patents Inc | Phenol compositions |
| EP2616414A1 (en) | 2010-09-14 | 2013-07-24 | ExxonMobil Chemical Patents Inc. | Cyclohexylbenzene compositions |
| US9365474B2 (en) | 2010-09-14 | 2016-06-14 | Exxonmobil Chemical Patents Inc. | Processes for producing phenol |
| CN103052618B (en) | 2010-09-14 | 2015-08-05 | 埃克森美孚化学专利公司 | Produce the method for phenol |
| WO2012036820A1 (en) | 2010-09-14 | 2012-03-22 | Exxonmobil Chemical Patents Inc. | Cyclohexylbenzene hydroperoxide compositions |
| US8921610B2 (en) | 2010-09-14 | 2014-12-30 | Exxonmobil Chemical Patents Inc. | Oxidation of alkylbenzenes |
| US9242918B2 (en) | 2010-09-14 | 2016-01-26 | Exxonmobil Chemical Patents Inc. | Dehydrogenation processes and phenol compositions |
| US9073840B2 (en) | 2010-09-14 | 2015-07-07 | Exxonmobil Chemical Patents Inc. | Oxidation of hydrocarbons |
| CN103153461B (en) | 2010-10-11 | 2015-11-25 | 埃克森美孚化学专利公司 | The activation of hydroalkylation catalyst and use |
| CN103153456A (en) | 2010-10-11 | 2013-06-12 | 埃克森美孚化学专利公司 | Process for producing phenol |
| WO2012067711A1 (en) | 2010-11-16 | 2012-05-24 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| WO2012082229A1 (en) | 2010-12-17 | 2012-06-21 | Exxonmobil Chemical Patents Inc. | Composition comprising cyclohexylbenzene and 1 - phenyl - 1 -methylcyclopentane and use thereof in oxidation processes |
| US9365467B2 (en) | 2010-12-17 | 2016-06-14 | Exxonmobil Chemical Patents Inc. | Process of producing cyclohexylbenzene |
| WO2012082407A1 (en) | 2010-12-17 | 2012-06-21 | Exxonmobil Chemical Patents Inc. | Dehydrogenation catalyst and process |
| WO2012082232A1 (en) | 2010-12-17 | 2012-06-21 | Exxonmobil Chemical Patenst Inc. | Process for producing cycloalkylaromatic compounds |
| WO2012134552A1 (en) | 2011-03-28 | 2012-10-04 | Exxonmobil Chemical Patents Inc. | Dehydrogenation process |
| CN103261125B (en) | 2010-12-21 | 2016-03-30 | 埃克森美孚化学专利公司 | The method of the aromatic substance of manufacture order cycloalkyl substituted |
| ES2552069T3 (en) | 2011-02-18 | 2015-11-25 | Exxonmobil Chemical Patents Inc. | Procedure to produce cyclohexylbenzene |
| US9017641B2 (en) | 2011-02-21 | 2015-04-28 | Exxonmobil Chemical Patents Inc. | Hydrogen purification process |
| US9061975B2 (en) | 2011-02-28 | 2015-06-23 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US8815050B2 (en) | 2011-03-22 | 2014-08-26 | Marathon Gtf Technology, Ltd. | Processes and systems for drying liquid bromine |
| US9044741B2 (en) | 2011-04-13 | 2015-06-02 | Kior, Inc. | Process for converting biomass to a fuel |
| KR101579359B1 (en) | 2011-04-19 | 2015-12-21 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Method for producing phenol and/or cyclohexanone |
| WO2012145028A2 (en) | 2011-04-19 | 2012-10-26 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US9388102B2 (en) | 2011-04-19 | 2016-07-12 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US8436220B2 (en) | 2011-06-10 | 2013-05-07 | Marathon Gtf Technology, Ltd. | Processes and systems for demethanization of brominated hydrocarbons |
| WO2014209557A1 (en) | 2013-06-28 | 2014-12-31 | Exxonmobil Chemical Patents Inc. | Process for producing phenol and/or cyclohexanone |
| US8829256B2 (en) | 2011-06-30 | 2014-09-09 | Gtc Technology Us, Llc | Processes and systems for fractionation of brominated hydrocarbons in the conversion of natural gas to liquid hydrocarbons |
| WO2013039673A1 (en) | 2011-09-16 | 2013-03-21 | Exxonmobil Chemical Patents Inc. | Improved liquid phase alkylation process |
| US9446961B2 (en) | 2011-09-16 | 2016-09-20 | Exxonmobil Chemical Patents Inc. | MCM-56 manufacture |
| WO2013043272A1 (en) | 2011-09-23 | 2013-03-28 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| WO2013043271A1 (en) | 2011-09-23 | 2013-03-28 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US9115105B2 (en) | 2011-09-30 | 2015-08-25 | Exxonmobil Chemical Patents Inc. | Production of propylene oxide |
| US9115060B2 (en) | 2011-10-07 | 2015-08-25 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US9108893B2 (en) | 2011-10-17 | 2015-08-18 | Exxonmobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| CA2852780A1 (en) | 2011-10-17 | 2013-04-25 | Exxonmobil Research And Engineering Company | Selective dehydration of alcohols to dialkylethers and integrated alcohol-to-gasoline processes |
| US8802908B2 (en) | 2011-10-21 | 2014-08-12 | Marathon Gtf Technology, Ltd. | Processes and systems for separate, parallel methane and higher alkanes' bromination |
| US20130144097A1 (en) | 2011-12-06 | 2013-06-06 | Exxonmobil Chemical Patents Inc. | Aromatics Production Process and Apparatus |
| US9193641B2 (en) | 2011-12-16 | 2015-11-24 | Gtc Technology Us, Llc | Processes and systems for conversion of alkyl bromides to higher molecular weight hydrocarbons in circulating catalyst reactor-regenerator systems |
| US8981158B2 (en) | 2011-12-19 | 2015-03-17 | Exxonmobil Chemical Patents Inc. | Oxidation of cyclohexylbenzene |
| US9096509B2 (en) | 2012-01-18 | 2015-08-04 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| SG11201404420SA (en) | 2012-02-08 | 2014-08-28 | Exxonmobil Chem Patents Inc | Process for producing cyclohexylbenzene |
| US9902664B2 (en) | 2012-02-08 | 2018-02-27 | Exxonmobil Chemical Patents Inc. | Production of monoalkyl aromatic compounds |
| CN104136402B (en) | 2012-02-27 | 2015-12-09 | 埃克森美孚化学专利公司 | Hydroalkylation method |
| US9458074B2 (en) | 2012-02-29 | 2016-10-04 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| EP2819977A1 (en) | 2012-03-01 | 2015-01-07 | ExxonMobil Chemical Patents Inc. | Process of producing cyclohexylbenzene |
| US9464020B2 (en) | 2012-05-02 | 2016-10-11 | Exxonmobil Chemical Patents Inc. | Process for producing phenol and cyclohexanone |
| CN104284876B (en) | 2012-05-02 | 2017-03-08 | 埃克森美孚化学专利公司 | Method for separating the mixture comprising phenol and Ketohexamethylene |
| US9169172B2 (en) | 2012-05-03 | 2015-10-27 | Exxonmobil Chemical Patents Inc. | Hydrogenation process |
| EP2867191B1 (en) | 2012-06-27 | 2019-09-11 | Badger Licensing, LLC | Process for producing cumene |
| CN104428277B (en) | 2012-07-05 | 2017-03-15 | 巴杰许可有限责任公司 | The method of production isopropylbenzene |
| WO2014011359A1 (en) | 2012-07-13 | 2014-01-16 | Badger Licensing Llc | Process for producing phenol |
| WO2014014708A1 (en) | 2012-07-20 | 2014-01-23 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US9555403B2 (en) | 2012-07-24 | 2017-01-31 | Exxonmobil Chemical Patents Inc. | Activation and use of hydroalkylation catalysts |
| US9259722B2 (en) | 2012-07-26 | 2016-02-16 | Badger Licensing Llc | Process for producing cumene |
| WO2014028139A1 (en) | 2012-08-14 | 2014-02-20 | Exxonmobil Chemical Patents Inc. | Process for producing cyclohexylbenzene |
| EP2885264B1 (en) | 2012-08-14 | 2018-12-19 | Badger Licensing LLC | Integrated process for producing cumene and purifying isopropanol |
| WO2014039653A2 (en) | 2012-09-07 | 2014-03-13 | Exxonmobil Chemical Patents Inc. | Oxidation of hydrocarbons |
| WO2014043371A1 (en) * | 2012-09-17 | 2014-03-20 | Exxonmobil Chemical Patents Inc. | Process for producing phenol and/or cyclohexanone from cyclohexylbenzene |
| US9272974B2 (en) * | 2012-09-17 | 2016-03-01 | Exxonmobil Chemical Patents Inc. | Process for producing phenol and/or cyclohexanone from cyclohexylbenzene |
| EP2906346A1 (en) | 2012-10-12 | 2015-08-19 | ExxonMobil Chemical Patents Inc. | Activation and use of hydroalkylation catalysts for the preparation of cycloalkylaromatic compounds, phenol and cyclohexanone |
| CN104936929B (en) | 2012-11-27 | 2018-11-02 | 巴杰许可有限责任公司 | The production of styrene |
| KR101745220B1 (en) | 2012-11-30 | 2017-06-08 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Dehydrogenation process |
| KR101735227B1 (en) | 2012-12-05 | 2017-05-12 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Cyclohexane dehydrogenation |
| US8940941B2 (en) | 2012-12-06 | 2015-01-27 | Exxonmobil Chemical Patents Inc. | Process for producing phenol and method for regenerating catalyst deactivated in the process |
| SG11201503421WA (en) | 2012-12-06 | 2015-05-28 | Exxonmobil Chem Patents Inc | Process for producing phenol |
| US9260387B2 (en) * | 2012-12-06 | 2016-02-16 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| US9334218B2 (en) | 2012-12-20 | 2016-05-10 | Exxonmobil Chemical Patents Inc. | Process for making phenol and/or cyclohexanone |
| WO2014109766A1 (en) | 2013-01-14 | 2014-07-17 | Badger Licensing Llc | Process for balancing gasoline and distillate production in a refinery |
| WO2014159106A1 (en) | 2013-03-14 | 2014-10-02 | Exxonmobil Chemical Patents Inc. | Methyl-substituted biphenyl compounds, their production and their use in the manufacture of plasticizers |
| WO2014159104A1 (en) | 2013-03-14 | 2014-10-02 | Exxonmobil Chemical Patents Inc. | Methyl-substituted biphenyl compounds, their production and their use in the manufacture of plasticizers |
| EP2978529A1 (en) | 2013-03-25 | 2016-02-03 | ExxonMobil Chemical Patents Inc. | Process for making alkylated aromatic compound |
| EP2978526A1 (en) | 2013-03-25 | 2016-02-03 | ExxonMobil Chemical Patents Inc. | Process for making alkylated aromatic compound |
| US9409841B2 (en) | 2013-04-02 | 2016-08-09 | Exxonmobil Chemical Patents Inc. | Process and apparatus for making phenol and/or cyclohexanone |
| WO2014182294A1 (en) | 2013-05-08 | 2014-11-13 | Badger Licensing Llc | Aromatics alkylation process |
| WO2014182442A1 (en) | 2013-05-09 | 2014-11-13 | Exxonmobil Chemical Patents Inc. | Regeneration of aromatic alkylation catalysts using aromatic solvents |
| WO2014182440A1 (en) | 2013-05-09 | 2014-11-13 | Exxonmobil Chemical Patetns Inc. | Regeneration of aromatic alkylation catalysts using hydrogen-containing gases |
| US9382178B2 (en) | 2013-05-22 | 2016-07-05 | Exxonmobil Chemical Patents Inc. | Process for producing phenol |
| EP3327003B1 (en) | 2013-06-28 | 2020-09-16 | ExxonMobil Chemical Patents Inc. | Process for concentrating a mixture containing organic hydroperoxide |
| US20160368844A1 (en) | 2013-07-24 | 2016-12-22 | Exxonmobil Chemical Patents Inc. | Process for Producing Mixtures of Cyclohexanone and Cyclohexanol |
| US9388094B2 (en) | 2013-08-30 | 2016-07-12 | Exxonmobil Chemical Patents Inc. | Catalytic alkane conversion |
| US9169170B2 (en) | 2013-10-01 | 2015-10-27 | Exxonmobil Chemical Patents Inc. | Hydroalkylating process |
| SG11201602275SA (en) | 2013-10-18 | 2016-04-28 | Exxonmobil Chem Patents Inc | Hydroalkylating process comprising an activation of the hydroalkylation catalyst and μετηod of making phenol and cyclohexanone. |
| US9335285B2 (en) | 2013-10-18 | 2016-05-10 | Exxonmobil Chemical Patents Inc. | Method for measuring acid strength in reaction medium using trimethylphosphine oxide and 31P NMR |
| US9144792B2 (en) | 2013-10-23 | 2015-09-29 | Exxonmobil Chemical Patents Inc. | Hydroalkylating process |
| SG11201603200VA (en) | 2013-11-22 | 2016-05-30 | Exxonmobil Chem Patents Inc | Process for making phenol and/or cyclohexanone |
| US9682900B2 (en) | 2013-12-06 | 2017-06-20 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion |
| WO2015084575A2 (en) | 2013-12-06 | 2015-06-11 | Exxonmobil Chemical Patents Inc. | Production of c2+ olefins |
| US10167361B2 (en) | 2014-03-25 | 2019-01-01 | Exxonmobil Chemical Patents Inc. | Production of aromatics and C2+olefins |
| US9682899B2 (en) | 2013-12-06 | 2017-06-20 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion |
| US10131588B2 (en) | 2013-12-06 | 2018-11-20 | Exxonmobil Chemical Patents Inc. | Production of C2+ olefins |
| US20170057895A1 (en) | 2013-12-20 | 2017-03-02 | Exxonmobil Chemical Patents Inc. | Phenol Composition |
| WO2015094528A1 (en) | 2013-12-20 | 2015-06-25 | Exxonmobil Chemical Patents Inc. | Cyclohexylbenzene composition |
| EP3083538A2 (en) | 2013-12-20 | 2016-10-26 | ExxonMobil Chemical Patents Inc. | Process for making phenol and/or cyclohexanone |
| WO2015094500A2 (en) | 2013-12-20 | 2015-06-25 | Exxonmobil Chemical Patents Inc. | Production of para-xylene |
| TWI646125B (en) | 2014-01-06 | 2019-01-01 | 盧森堡商英威達技術有限公司 | Copolyether ester polyol process |
| EP3099634B1 (en) | 2014-01-27 | 2021-04-14 | ExxonMobil Chemical Patents Inc. | Synthesis of molecular sieve mcm-56 |
| US9950971B2 (en) | 2014-07-23 | 2018-04-24 | Exxonmobil Chemical Patents Inc. | Process and catalyst for methane conversion to aromatics |
| US9714386B2 (en) | 2014-07-24 | 2017-07-25 | Exxonmobil Chemical Patents Inc. | Production of xylenes from syngas |
| US9809758B2 (en) | 2014-07-24 | 2017-11-07 | Exxonmobil Chemical Patents Inc. | Production of xylenes from syngas |
| WO2016025214A1 (en) | 2014-08-15 | 2016-02-18 | Exxonmobil Chemical Patents Inc. | Process and system for making cyclohexanone |
| KR20170027808A (en) | 2014-08-15 | 2017-03-10 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Process and system for making cyclohexanone |
| EP3180304B1 (en) | 2014-08-15 | 2019-10-30 | ExxonMobil Chemical Patents Inc. | Process for producing cyclohexanone |
| US9868687B2 (en) | 2014-09-30 | 2018-01-16 | Exxonmobil Chemical Patents Inc. | Process for making cyclohexanone |
| WO2016085908A1 (en) | 2014-11-25 | 2016-06-02 | Badger Licensing Llc | Process for reducing the benzene content of gasoline |
| US10781149B2 (en) | 2014-12-19 | 2020-09-22 | Exxonmobil Chemical Patents Inc. | Transalkylation process |
| US10053403B2 (en) | 2015-02-04 | 2018-08-21 | Exxonmobil Chemical Patents Inc. | Catalyst compositions and their use in transalkylation of heavy aromatics to xylenes |
| US10118165B2 (en) | 2015-02-04 | 2018-11-06 | Exxonmobil Chemical Patents Inc. | Catalyst compositions and use in heavy aromatics conversion processes |
| WO2016148755A1 (en) | 2015-03-19 | 2016-09-22 | Exxonmobil Chemical Patents Inc. | Process and apparatus for the production of para-xylene |
| JP6526228B2 (en) | 2015-03-25 | 2019-06-05 | エクソンモービル ケミカル パテンツ インコーポレイテッド | Production of biphenyl compounds |
| WO2016160092A1 (en) | 2015-03-31 | 2016-10-06 | Exxonmobil Chemical Patents Inc. | Process for making cyclohexanone and/or phenol |
| US10017433B2 (en) | 2015-03-31 | 2018-07-10 | Exxonmobil Chemical Patents Inc. | Transalkylated cyclohexylbenzyl and biphenyl compounds |
| CN107531590B (en) | 2015-04-30 | 2020-11-06 | 埃克森美孚化学专利公司 | Process and apparatus for producing paraxylene |
| WO2017023429A1 (en) | 2015-07-31 | 2017-02-09 | Exxonmobil Chemical Patents Inc. | Process for making cyclohexanone |
| EP3328823A1 (en) | 2015-07-31 | 2018-06-06 | ExxonMobil Chemical Patents Inc. | Process for making cyclohexanone |
| US10202318B2 (en) | 2015-09-25 | 2019-02-12 | Exxonmobil Chemical Patents Inc. | Catalyst and its use in hydrocarbon conversion process |
| US9988325B2 (en) | 2015-09-25 | 2018-06-05 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion |
| US9796643B2 (en) | 2015-09-25 | 2017-10-24 | Exxonmobil Chemical Patents Inc. | Hydrocarbon dehydrocyclization in the presence of carbon dioxide |
| WO2017052856A1 (en) | 2015-09-25 | 2017-03-30 | Exxonmobil Chemical Patents Inc. | Catalyst and its use in dehydrocyclization processes |
| US9963406B2 (en) | 2015-09-25 | 2018-05-08 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion |
| WO2017052858A1 (en) | 2015-09-25 | 2017-03-30 | Exxonmobile Chemical Patents Inc. | Conversion of non-aromatic hydrocarbon |
| US9845272B2 (en) | 2015-09-25 | 2017-12-19 | Exxonmobil Chemical Patents Inc. | Hydrocarbon conversion |
| KR102452149B1 (en) | 2015-10-15 | 2022-10-06 | 바져 라이센싱 엘엘씨 | Method for preparing an alkylaromatic compound |
| CA3004322C (en) | 2015-11-04 | 2020-06-30 | Exxonmobil Chemical Patents Inc. | Production of cyclic c5 compounds |
| CA3004332C (en) | 2015-11-04 | 2019-12-31 | Exxonmobil Chemical Patents Inc. | Processes and systems for converting hydrocarbons to cyclopentadiene |
| CN108349834B (en) | 2015-11-04 | 2019-04-23 | 埃克森美孚化学专利公司 | It is used to prepare the method and system of cyclopentadiene and/or dicyclopentadiene |
| EP3371137B1 (en) | 2015-11-04 | 2021-08-04 | ExxonMobil Chemical Patents Inc. | Fired tube conversion system and process |
| KR102570207B1 (en) | 2016-02-17 | 2023-08-25 | 바져 라이센싱 엘엘씨 | Method for producing ethylbenzene |
| US10906851B2 (en) | 2016-02-26 | 2021-02-02 | Exxonmobil Chemical Patents Inc. | Process for recovering para-xylene |
| US20200325096A1 (en) | 2016-04-08 | 2020-10-15 | Exxonmobil Chemical Patents Inc. | Oxidation of Methyl-substituted Biphenyl Compounds |
| CN109310999A (en) | 2016-04-25 | 2019-02-05 | 埃克森美孚化学专利公司 | Catalytic aromatization |
| AU2016404249B2 (en) | 2016-04-26 | 2022-04-07 | Badger Licensing Llc | Process for reducing the benzene content of gasoline |
| KR102176960B1 (en) | 2016-06-09 | 2020-11-10 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | Method for producing mono-alkylated aromatic compounds |
| WO2017222767A1 (en) | 2016-06-23 | 2017-12-28 | Exxonmobil Research And Engineering Company | Isoparaffin-olefin aklylation |
| WO2017222768A1 (en) | 2016-06-23 | 2017-12-28 | Exxonmobil Research And Engineering Company | Isoparaffin-olefin alkylation |
| US20170369393A1 (en) | 2016-06-23 | 2017-12-28 | Exxonmobil Research And Engineering Company | Isoparaffin-olefin alkylation |
| WO2017222765A1 (en) | 2016-06-23 | 2017-12-28 | Exxonmobil Research And Engineering Company | Isoparaffin-olefin alkylation |
| US10300404B2 (en) | 2016-06-30 | 2019-05-28 | Exxonmobil Chemical Patents Inc. | Process for the recovering of paraxylene |
| US10351489B2 (en) | 2016-06-30 | 2019-07-16 | Exxonmobil Chemical Patents Inc. | Processes for recovering paraxylene |
| WO2018071184A1 (en) | 2016-10-10 | 2018-04-19 | Exxonmobil Chemical Patents Inc. | Heavy aromatics to btx conversion process and catalyst compositions used |
| WO2018071185A1 (en) | 2016-10-10 | 2018-04-19 | Exxonmobil Chemical Patents Inc. | Heavy aromatics to btx conversion process and dual bed catalyst systems used |
| CA3044543A1 (en) | 2016-12-14 | 2018-06-21 | Exxonmobil Research And Engineering Company | Production of alkylate from isoparaffin |
| WO2018118440A1 (en) | 2016-12-20 | 2018-06-28 | Exxonmobil Research And Engineering Company | Upgrading ethane-containing light paraffins streams |
| WO2018118439A1 (en) | 2016-12-20 | 2018-06-28 | Exxonmobil Research And Engineering Company | Upgrading ethane-containing light paraffins streams |
| US10450242B2 (en) | 2016-12-20 | 2019-10-22 | Exxonmobil Research And Engineering Company | Upgrading ethane-containing light paraffins streams |
| WO2018140149A1 (en) | 2017-01-25 | 2018-08-02 | Exxonmobil Chemical Patents Inc. | Transalkylation process and catalyst composition used therein |
| KR102524765B1 (en) | 2017-02-16 | 2023-04-25 | 엑손모빌 테크놀로지 앤드 엔지니어링 컴퍼니 | Fixed bed radial flow reactor for hard paraffin conversion |
| WO2018160327A1 (en) | 2017-02-28 | 2018-09-07 | Exxonmobil Chemical Patents Inc. | Catalyst compositions and their use in aromatic alkylation processes |
| WO2018183009A1 (en) | 2017-03-29 | 2018-10-04 | Exxonmobil Chemical Patents Inc. | Catalyst compositions and their use in aromatic alkylation processes |
| WO2018183012A1 (en) | 2017-03-29 | 2018-10-04 | Exxonmobil Chemical Patents Inc. | Methods for removing impurities from a hydrocarbon stream and their use in aromatic alkylation processes |
| WO2018217337A1 (en) | 2017-05-22 | 2018-11-29 | Exxonmobil Chemical Patents Inc. | Integrated aromatics formation and methylation |
| PT3640207T (en) | 2017-06-13 | 2022-04-14 | China Petroleum & Chem Corp | Molecular sieve scm-15, synthesis method therefor and use thereof |
| PT3640208T (en) | 2017-06-13 | 2022-04-11 | China Petroleum & Chem Corp | Molecular sieve scm-14, synthesis method therefor and use thereof |
| US10752570B2 (en) | 2017-06-28 | 2020-08-25 | Exxonmobil Chemical Patents Inc. | Processes for making cyclohexanone |
| US10941099B2 (en) | 2017-06-28 | 2021-03-09 | Exxonmobil Chemical Patents Inc. | Cyclohexanone-containing products and processes for making the same |
| US10745336B2 (en) | 2017-06-28 | 2020-08-18 | Exxonmobil Chemical Patents Inc. | Cyclohexanone-containing products and processes for making the same |
| CN110740810A (en) | 2017-08-31 | 2020-01-31 | 优美科股份公司及两合公司 | Use of palladium/platinum/zeolite-based catalysts as passive nitrogen oxide adsorbents for the purification of exhaust gases |
| WO2019042883A1 (en) | 2017-08-31 | 2019-03-07 | Umicore Ag & Co. Kg | Palladium/zeolite-based passive nitrogen oxide adsorber catalyst for purifying exhaust gas |
| EP3724155A1 (en) | 2017-12-14 | 2020-10-21 | ExxonMobil Chemical Patents Inc. | Processes for isomerizing alpha olefins |
| JP2021506909A (en) | 2017-12-22 | 2021-02-22 | エクソンモービル・ケミカル・パテンツ・インク | Catalyst for the production of para-xylene by methylation of benzene and / or toluene |
| WO2019134958A1 (en) | 2018-01-05 | 2019-07-11 | Umicore Ag & Co. Kg | Passive nitrogen oxide adsorber |
| WO2019143495A1 (en) | 2018-01-22 | 2019-07-25 | Exxonmobil Chemical Patents Inc. | Production and use of 3,4' and 4,4'-dimethylbiphenyl isomers |
| WO2019182744A1 (en) | 2018-03-23 | 2019-09-26 | Exxonmobil Chemical Patents Inc. | Processes and systems for the conversion of hydrocarbons |
| WO2019199459A1 (en) | 2018-04-12 | 2019-10-17 | Exxonmobil Chemical Patents Inc. | Preparation and use of biphenyl carboxylic acids, alcohols, and esters |
| WO2019212784A1 (en) | 2018-05-03 | 2019-11-07 | Exxonmobil Chemical Patents Inc | Preparation of an hydroalkylation catalyst |
| JP7341247B2 (en) | 2019-02-27 | 2023-09-08 | シェブロン ユー.エス.エー. インコーポレイテッド | Molecular sieve SSZ-115, its synthesis and use |
| CN113574038A (en) | 2019-03-28 | 2021-10-29 | 埃克森美孚化学专利公司 | Method for converting benzene and/or toluene via methylation |
| US11691933B2 (en) | 2019-03-28 | 2023-07-04 | Exxonmobil Chemical Patents Inc. | Processes and systems for converting benzene and/or toluene via methylation |
| WO2020197890A1 (en) | 2019-03-28 | 2020-10-01 | Exxonmobil Chemical Patents Inc. | Processes for converting benzene and/or toluene via methylation |
| WO2021025834A1 (en) | 2019-08-06 | 2021-02-11 | Exxonmobil Research And Engineering Company | Multistage alkylation at improved isoparaffin to olefin ratios |
| WO2021025836A1 (en) | 2019-08-06 | 2021-02-11 | Exxonmobil Research And Engineering Company | Catalysts and multistage alkylation of isoparaffin |
| WO2021025835A1 (en) | 2019-08-06 | 2021-02-11 | Exxonmobil Research And Engineering Company | Catalyst rejuvenation in multistage alkylation of isoparaffin |
| WO2021025833A1 (en) | 2019-08-06 | 2021-02-11 | Exxonmobil Research And Engineering Company | Multistage alkylation using interstage hydrogenation |
| WO2021076260A1 (en) | 2019-10-17 | 2021-04-22 | Exxonmobil Chemical Patents Inc. | Production of alkylaromatic compounds |
| US11820723B2 (en) | 2019-10-17 | 2023-11-21 | Exxonmobil Chemicals Patents Inc. | Production of alkylaromatic compounds |
| US20240051898A1 (en) | 2019-10-25 | 2024-02-15 | Exxonmobil Chemical Patents Inc. | Production of Alkylaromatic Compounds |
| EP3824988A1 (en) | 2019-11-20 | 2021-05-26 | UMICORE AG & Co. KG | Catalyst for reducing nitrogen oxides |
| WO2021154440A1 (en) | 2020-01-29 | 2021-08-05 | Exxonmobil Research And Engineering Company | Oligomerization of isobutanol in the presence of mww zeolite solid acid catalysts |
| BR112022013640A2 (en) | 2020-02-18 | 2022-09-06 | Symrise Ag | PROCESS FOR PREPARATION OF LIMONENOS AND COMPOSITION CONTAINING LIMONENOS |
| CN115175875A (en) | 2020-02-18 | 2022-10-11 | 西姆莱斯股份公司 | Process for preparing limonene and compositions containing limonene |
| EP4182078A1 (en) | 2020-07-16 | 2023-05-24 | ExxonMobil Chemical Patents Inc. | Method of synthesizing a molecular sieve of mww framework type |
| WO2022060352A1 (en) | 2020-09-16 | 2022-03-24 | Exxonmobil Research And Engineering Company | System and method for isoparaffin alkylation |
| WO2022060353A1 (en) | 2020-09-16 | 2022-03-24 | Exxonmobil Research And Engineering Company | High activity mww type zeolite catalyst for alkylation of light olefins with isoparaffin |
| US20230365479A1 (en) | 2020-09-30 | 2023-11-16 | Exxonmobil Chemical Patents Inc. | Processes for Converting C8 Aromatic Hydrocarbons |
| JP2023548551A (en) | 2020-11-06 | 2023-11-17 | エクソンモービル ケミカル パテンツ インコーポレイテッド | Production of alkyl aromatic compounds |
| EP4247920A1 (en) | 2020-11-17 | 2023-09-27 | ExxonMobil Chemical Patents Inc. | Concurrent isomerization/hydrogenation of unsaturated polyalphaolefin in the presence of a high activity catalyst |
| WO2022184759A1 (en) | 2021-03-03 | 2022-09-09 | Exxonmobil Chemical Patents Inc. | Method of synthesizing a molecular sieve of mww framework type |
| WO2023044278A1 (en) | 2021-09-16 | 2023-03-23 | Exxonmobil Chemical Patents Inc. | Xylene isomer separation processes |
| TW202315841A (en) | 2021-10-11 | 2023-04-16 | 大陸商中國石油化工科技開發有限公司 | Silicon-aluminum molecular sieve scm-36, manufacturing method therefor and application thereof |
| WO2023064684A1 (en) | 2021-10-12 | 2023-04-20 | Exxonmobil Chemical Patents Inc. | Staged alkylation for producing xylene products |
| WO2023204947A1 (en) | 2022-04-19 | 2023-10-26 | Exxonmobil Chemical Patents Inc. | Processes for oxidizing p-xylene or p-xylene-containing mixtures |
| WO2023244389A1 (en) | 2022-06-14 | 2023-12-21 | Exxonmobil Chemical Patents Inc. | Production of p-xylene by liquid-phase isomerization in the presence of c9+ aromatic hydrocarbons and separation thereof |
| WO2023244394A1 (en) | 2022-06-14 | 2023-12-21 | Exxonmobil Chemical Patents Inc. | Production of p-xylene by liquid-phase isomerization and separation thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3894107A (en) * | 1973-08-09 | 1975-07-08 | Mobil Oil Corp | Conversion of alcohols, mercaptans, sulfides, halides and/or amines |
| US3894105A (en) * | 1973-08-09 | 1975-07-08 | Mobil Oil Corp | Production of durene |
| US3911041A (en) * | 1974-09-23 | 1975-10-07 | Mobil Oil Corp | Conversion of methanol and dimethyl ether |
| US4025575A (en) * | 1975-04-08 | 1977-05-24 | Mobil Oil Corporation | Process for manufacturing olefins |
| US4104151A (en) * | 1976-11-08 | 1978-08-01 | Mobil Oil Corporation | Organic compound conversion over ZSM-23 |
| DE2909927A1 (en) * | 1979-03-14 | 1980-09-25 | Basf Ag | METHOD FOR PRODUCING OLEFINES FROM RAW METHANOL |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3994105A (en) * | 1972-03-20 | 1976-11-30 | Hughes Aircraft Company | Shelter construction |
-
1981
- 1981-04-30 DE DE19813117135 patent/DE3117135A1/en not_active Withdrawn
-
1982
- 1982-04-13 US US06/368,055 patent/US4439409A/en not_active Expired - Lifetime
- 1982-04-19 EP EP82103256A patent/EP0064205B1/en not_active Expired
- 1982-04-19 DE DE8282103256T patent/DE3261429D1/en not_active Expired
- 1982-04-23 JP JP57067520A patent/JPS57183321A/en active Granted
- 1982-04-28 AU AU83064/82A patent/AU8306482A/en not_active Withdrawn
- 1982-04-28 CA CA000401848A patent/CA1174219A/en not_active Expired
- 1982-04-29 ZA ZA822922A patent/ZA822922B/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3894107A (en) * | 1973-08-09 | 1975-07-08 | Mobil Oil Corp | Conversion of alcohols, mercaptans, sulfides, halides and/or amines |
| US3894105A (en) * | 1973-08-09 | 1975-07-08 | Mobil Oil Corp | Production of durene |
| US3911041A (en) * | 1974-09-23 | 1975-10-07 | Mobil Oil Corp | Conversion of methanol and dimethyl ether |
| US4025575A (en) * | 1975-04-08 | 1977-05-24 | Mobil Oil Corporation | Process for manufacturing olefins |
| US4104151A (en) * | 1976-11-08 | 1978-08-01 | Mobil Oil Corporation | Organic compound conversion over ZSM-23 |
| DE2909927A1 (en) * | 1979-03-14 | 1980-09-25 | Basf Ag | METHOD FOR PRODUCING OLEFINES FROM RAW METHANOL |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1987004368A1 (en) * | 1986-01-24 | 1987-07-30 | Haldor Topso^/E A/S | Crystalline aluminosilicate and a method for its preparation |
| WO1991014753A1 (en) * | 1990-03-19 | 1991-10-03 | Mobil Oil Corporation | Catalytic hydroisomerization process |
| WO1992006921A1 (en) * | 1990-10-17 | 1992-04-30 | Mobil Oil Corporation | Synthetic porous crystalline material, its synthesis and use |
| AU639503B2 (en) * | 1990-10-17 | 1993-07-29 | Mobil Oil Corporation | Synthetic porous crystalline material, its synthesis and use |
| WO2000063117A1 (en) * | 1999-04-16 | 2000-10-26 | Centre National De La Recherche Scientifique | Irc1 zeolite and methods for preparing same |
| FR2793236A1 (en) * | 1999-04-16 | 2000-11-10 | Centre Nat Rech Scient | NEW ZEOLITE AND METHODS FOR PREPARING THE SAME |
Also Published As
| Publication number | Publication date |
|---|---|
| ZA822922B (en) | 1983-03-30 |
| AU8306482A (en) | 1982-11-04 |
| US4439409A (en) | 1984-03-27 |
| DE3117135A1 (en) | 1982-11-18 |
| DE3261429D1 (en) | 1985-01-17 |
| JPH0210761B2 (en) | 1990-03-09 |
| CA1174219A (en) | 1984-09-11 |
| EP0064205B1 (en) | 1984-12-05 |
| JPS57183321A (en) | 1982-11-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0064205B1 (en) | Crystalline aluminosilicate, process for producing it and its use in converting methanol and/or dimethyl ether into hydrocarbons | |
| EP0369364B1 (en) | Catalyst based on a crystalline aluminosilicate | |
| DE69819989T3 (en) | Composition containing pentasil-type molecular sieves, and their preparation and use | |
| DE3218787A1 (en) | CATALYST, METHOD FOR THE PRODUCTION THEREOF AND ITS USE FOR THE PRODUCTION OF HYDROCARBONS FROM SYNTHESIS GAS | |
| DE2830787A1 (en) | METHOD FOR PRODUCING NITROGEN-CONTAINING CRYSTALLINE METAL SILICATES WITH ZEOLITE STRUCTURE | |
| DE2755901A1 (en) | ZEOLITE MATERIAL AND THEIR USE IN A METHOD FOR THE PRODUCTION OF AROMATIC HYDROCARBONS | |
| DE2826865C2 (en) | ||
| DE2321399C2 (en) | Process for the alkylation of aromatic hydrocarbons | |
| DE3149979C2 (en) | Process for the preparation of an ethyl or isopropyl ester of a carboxylic acid | |
| DD151877A5 (en) | METHOD FOR ALKYLATING HYDROCARBONS | |
| DE3341902C2 (en) | Crystalline aluminosilicate, process for its production and its use as a catalyst in the pyrolysis treatment of methanol and / or dimethyl ether for the production of olefins | |
| DE2913552A1 (en) | METHOD OF MANUFACTURING ZEOLITHES AND THEIR USE | |
| DE3524890A1 (en) | Process for the preparation of low-molecular-weight olefins | |
| DE3027358A1 (en) | METHOD FOR PRODUCING AN AROMATIC HYDROCARBON MIXTURE | |
| EP0111808A2 (en) | Catalyst, process for preparing said catalyst and isomerization process in the presence of said catalyst | |
| DE3316320C2 (en) | ||
| EP0090283B1 (en) | Process for the preparation of olefines from methanol and/or dimethyl ether | |
| DE3903622A1 (en) | METHOD FOR PRODUCING 1,4-DIAZABICYCLO-2,2,2-OCTANE | |
| DE2322710A1 (en) | HYDROCARBON CONVERSION CATALYST WITH CONTENT IN A FAUJASITE | |
| EP0266691B1 (en) | Method for the production of alkene carboxylic acid esters | |
| EP0423526B1 (en) | Process for the preparation of triethylenediamine and piperazine | |
| EP0206143B1 (en) | Process for the preparation of 4-pentenoic-acid esters | |
| DE3518094C2 (en) | ||
| DE3419379A1 (en) | METHOD FOR PRODUCING SERVES BY DEHYDRATING ALDEHYDES | |
| DE3542171A1 (en) | METHOD FOR PRODUCING DIISOBUTEN FROM ISOBUTEN |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19820419 |
|
| AK | Designated contracting states |
Designated state(s): BE DE FR GB IT NL |
|
| ITF | It: translation for a ep patent filed |
Owner name: ING. C. GREGORJ S.P.A. |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): BE DE FR GB IT NL |
|
| REF | Corresponds to: |
Ref document number: 3261429 Country of ref document: DE Date of ref document: 19850117 |
|
| ET | Fr: translation filed | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Effective date: 19850430 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| BERE | Be: lapsed |
Owner name: BAYER A.G. Effective date: 19850419 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19851101 |
|
| 26N | No opposition filed | ||
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19881121 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20010317 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20010322 Year of fee payment: 20 |