US6583255B1 - Polyester oligomer - Google Patents
Polyester oligomer Download PDFInfo
- Publication number
- US6583255B1 US6583255B1 US08/463,437 US46343795A US6583255B1 US 6583255 B1 US6583255 B1 US 6583255B1 US 46343795 A US46343795 A US 46343795A US 6583255 B1 US6583255 B1 US 6583255B1
- Authority
- US
- United States
- Prior art keywords
- oligomer
- general formula
- radical
- group
- mixtures
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 C.C.C.C.C.C.C.C.C.C.C.C.CC(=O)OBOC(=O)*C(=O)OBOC(C)=O.COC(=O)*C(=O)OBOC(=O)*C(=O)OC Chemical compound C.C.C.C.C.C.C.C.C.C.C.C.CC(=O)OBOC(=O)*C(=O)OBOC(C)=O.COC(=O)*C(=O)OBOC(=O)*C(=O)OC 0.000 description 28
- JBRFOKDRXFPODV-UHFFFAOYSA-N Cc1ccc([W]c2ccc(C)cc2)cc1 Chemical compound Cc1ccc([W]c2ccc(C)cc2)cc1 JBRFOKDRXFPODV-UHFFFAOYSA-N 0.000 description 8
- ZLVVTPOIPOXOMK-IEAYMUCESA-N C#Cc1ccc2c(c1)C(=O)N(C)C2=O.CC.CC.CC.CC.CC.CC.CC1C2C=CC1C1C(=O)N(C)C(=O)C21.CN1C(=O)C2C(C1=O)C1(C)C=CC2(C)O1.CN1C(=O)C2C3C=CC(C3)C2C1=O.CN1C(=O)C2C3C=CC(C3)C2C1=O.CN1C(=O)C=CC1=O.[3H]C Chemical compound C#Cc1ccc2c(c1)C(=O)N(C)C2=O.CC.CC.CC.CC.CC.CC.CC1C2C=CC1C1C(=O)N(C)C(=O)C21.CN1C(=O)C2C(C1=O)C1(C)C=CC2(C)O1.CN1C(=O)C2C3C=CC(C3)C2C1=O.CN1C(=O)C2C3C=CC(C3)C2C1=O.CN1C(=O)C=CC1=O.[3H]C ZLVVTPOIPOXOMK-IEAYMUCESA-N 0.000 description 5
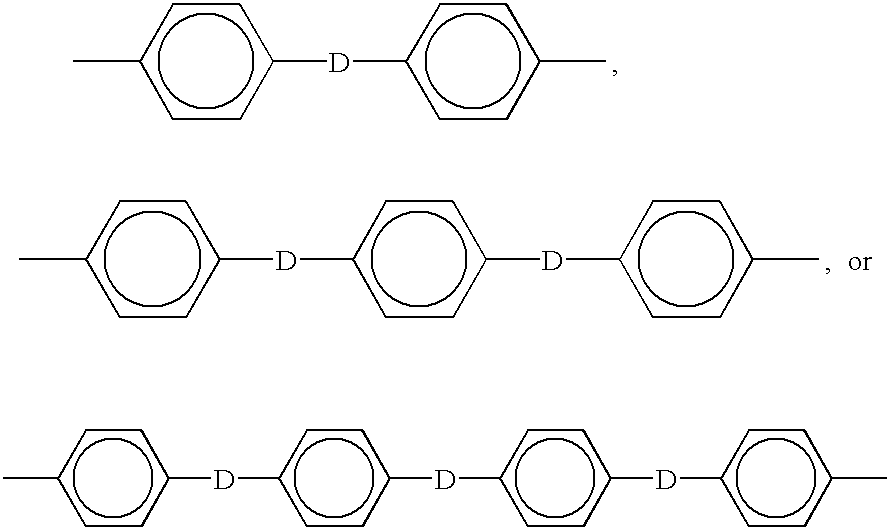
- GULLCTYMGYZGIG-JJTJTZLASA-N Cc1ccc([2H]c2ccc(C)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc(C)cc3)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 Chemical compound Cc1ccc([2H]c2ccc(C)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc(C)cc3)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 GULLCTYMGYZGIG-JJTJTZLASA-N 0.000 description 3
- MEKDPHXPVMKCON-UHFFFAOYSA-N C.CC Chemical compound C.CC MEKDPHXPVMKCON-UHFFFAOYSA-N 0.000 description 2
- KJVJIESCQKZPJX-JUCBKUTKSA-N Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 Chemical compound Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 KJVJIESCQKZPJX-JUCBKUTKSA-N 0.000 description 2
- IRSVBDBNYGOVQL-HNSMEKFASA-N C.C.C.Cc1ccc([2H]c2ccc(C)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc(C)cc3)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 Chemical compound C.C.C.Cc1ccc([2H]c2ccc(C)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc(C)cc3)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 IRSVBDBNYGOVQL-HNSMEKFASA-N 0.000 description 1
- KWGXTFJJJBRFKK-OWAABQRYSA-N C.C.Cc1ccc([2H]c2ccc(C)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc(C)cc3)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 Chemical compound C.C.Cc1ccc([2H]c2ccc(C)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc(C)cc3)cc2)cc1.Cc1ccc([2H]c2ccc([2H]c3ccc([2H]c4ccc(C)cc4)cc3)cc2)cc1 KWGXTFJJJBRFKK-OWAABQRYSA-N 0.000 description 1
- RCQXUUKLFUVGMO-UHFFFAOYSA-N C.CF1C(=O)N(FOF)C1=O Chemical compound C.CF1C(=O)N(FOF)C1=O RCQXUUKLFUVGMO-UHFFFAOYSA-N 0.000 description 1
- WEQXVGNWXCDEGR-UHFFFAOYSA-N COC.Nc1ccccc1.c1ccccc1 Chemical compound COC.Nc1ccccc1.c1ccccc1 WEQXVGNWXCDEGR-UHFFFAOYSA-N 0.000 description 1
- POQFGMORYOIZJZ-UHFFFAOYSA-N Cc1ccc(Cc2ccc(Oc3ccc(C)cc3)cc2)cc1 Chemical compound Cc1ccc(Cc2ccc(Oc3ccc(C)cc3)cc2)cc1 POQFGMORYOIZJZ-UHFFFAOYSA-N 0.000 description 1
- GHPZVITXGGLOBG-UHFFFAOYSA-N Cc1ccc(Oc2ccc(Cc3ccc(Oc4ccc(C)cc4)cc3)cc2)cc1 Chemical compound Cc1ccc(Oc2ccc(Cc3ccc(Oc4ccc(C)cc4)cc3)cc2)cc1 GHPZVITXGGLOBG-UHFFFAOYSA-N 0.000 description 1
- WEHFQAFKIFYONA-UHFFFAOYSA-N Nc1ccc(C(=O)O)cc1.O=C(Cl)c1ccc(N=Cc2cccc(C=Nc3ccc(C(=O)Cl)cc3)c2)cc1.O=C(O)c1ccc(N=Cc2cccc(C=Nc3ccc(C(=O)O)cc3)c2)cc1.O=Cc1cccc(C=O)c1.O=[SH](=O)Cl Chemical compound Nc1ccc(C(=O)O)cc1.O=C(Cl)c1ccc(N=Cc2cccc(C=Nc3ccc(C(=O)Cl)cc3)c2)cc1.O=C(O)c1ccc(N=Cc2cccc(C=Nc3ccc(C(=O)O)cc3)c2)cc1.O=Cc1cccc(C=O)c1.O=[SH](=O)Cl WEHFQAFKIFYONA-UHFFFAOYSA-N 0.000 description 1
- ZLUDDFQVFAZDED-UHFFFAOYSA-O Nc1ccc(C(=O)[OH2+])cc1.O=C(O)c1ccc(N=Cc2cccc(C=Nc3ccc(C(=O)O)cc3)c2)cc1.O=Cc1cccc(C=O)c1.O=[SH](=O)Cl Chemical compound Nc1ccc(C(=O)[OH2+])cc1.O=C(O)c1ccc(N=Cc2cccc(C=Nc3ccc(C(=O)O)cc3)c2)cc1.O=Cc1cccc(C=O)c1.O=[SH](=O)Cl ZLUDDFQVFAZDED-UHFFFAOYSA-O 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/02—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds
- C08G63/12—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/16—Dicarboxylic acids and dihydroxy compounds
- C08G63/18—Dicarboxylic acids and dihydroxy compounds the acids or hydroxy compounds containing carbocyclic rings
- C08G63/181—Acids containing aromatic rings
- C08G63/185—Acids containing aromatic rings containing two or more aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/02—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds
- C08G63/12—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/16—Dicarboxylic acids and dihydroxy compounds
- C08G63/20—Polyesters having been prepared in the presence of compounds having one reactive group or more than two reactive groups
- C08G63/21—Polyesters having been prepared in the presence of compounds having one reactive group or more than two reactive groups in the presence of unsaturated monocarboxylic acids or unsaturated monohydric alcohols or reactive derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/02—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds
- C08G63/12—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/16—Dicarboxylic acids and dihydroxy compounds
- C08G63/18—Dicarboxylic acids and dihydroxy compounds the acids or hydroxy compounds containing carbocyclic rings
- C08G63/19—Hydroxy compounds containing aromatic rings
- C08G63/193—Hydroxy compounds containing aromatic rings containing two or more aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/68—Polyesters containing atoms other than carbon, hydrogen and oxygen
- C08G63/682—Polyesters containing atoms other than carbon, hydrogen and oxygen containing halogens
- C08G63/6824—Polyesters containing atoms other than carbon, hydrogen and oxygen containing halogens derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/6826—Dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/68—Polyesters containing atoms other than carbon, hydrogen and oxygen
- C08G63/685—Polyesters containing atoms other than carbon, hydrogen and oxygen containing nitrogen
- C08G63/6854—Polyesters containing atoms other than carbon, hydrogen and oxygen containing nitrogen derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/6856—Dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/68—Polyesters containing atoms other than carbon, hydrogen and oxygen
- C08G63/688—Polyesters containing atoms other than carbon, hydrogen and oxygen containing sulfur
- C08G63/6884—Polyesters containing atoms other than carbon, hydrogen and oxygen containing sulfur derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/6886—Dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L81/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing sulfur with or without nitrogen, oxygen or carbon only; Compositions of polysulfones; Compositions of derivatives of such polymers
- C08L81/06—Polysulfones; Polyethersulfones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2367/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2367/06—Unsaturated polyesters
- C08J2367/07—Unsaturated polyesters having terminal carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L67/00—Compositions of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Compositions of derivatives of such polymers
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31786—Of polyester [e.g., alkyd, etc.]
- Y10T428/31794—Of cross-linked polyester
Definitions
- the present invention relates to linear or multidimensional polyester oligomers and blends that include unsaturated hydrocarbon end caps to provide solvent resistance through crosslinking upon curing.
- the backbone includes electronegative (“sulfone”) segments that alternate with the ester linkages.
- oligomers for high performance advanced composites suitable for aerospace applications. These composites should exhibit solvent resistance, be tough, impact resistant, and strength, be easy to process, and be thermoplastic. Oligomers and composites that have thermo-oxidative stability, and, accordingly can be used at elevated temperatures, are particularly desirable.
- Linear polysulfone, polyether sulfone, polyester, and polyamide systems are also known, but each of these systems fails to provide as high thermal stability as is required in some aerospace applications.
- polyimide sulfone compounds synthesized to provide unique properties or combinations of properties.
- Kwiatkowski and Brode synthesized maleic-capped, linear polyarylimides as disclosed in U.S. Pat. No. 3,839,287.
- Holub and Evans synthesized maleic- or nadic-capped, imido-substituted polyester compositions as disclosed in U.S. Pat. No. 3,729,446.
- We synthesized thermally stable polysulfone oligomers as disclosed in U.S. Pat. No. 4,476,184 or U.S. Pat. No.
- Multidimensional oligomers such as disclosed in our U.S. Pat. No. 5,210,213; are easier to process than many other advanced composite oligomers since they can be handled at lower temperatures.
- the unsaturated phenylimide end caps crosslink so that the thermal resistance of the resulting composite is markedly increased with only a minor loss of stiffness, matrix stress transfer (impact resistance), toughness, elasticity, and other mechanical properties. Glass transition temperatures above 950° F. are achievable.
- polyesters when combined-with well-known diluents, such as styrene, do not exhibit satisfactory thermal and oxidative resistance to be useful for aircraft or aerospace applications.
- Polyarylesters are unsatisfactory, also, since the resins often are semicrystalline which makes them insoluble in laminating solvents, intractable in fusion, and subject to shrinking or warping during composite fabrication. Those polyarylesters that are soluble in conventional laminating solvents remain so in composite form, thereby limiting their usefulness in structural composites.
- the high concentration of ester groups contributes to resin strength and tenacity, but also makes the resin susceptible to the damaging effects of water absorption. High moisture absorption by commercial polyesters can lead to distortion of the composite when it is loaded at elevated temperature.
- High performance, aerospace, polyester advanced composites can be prepared using crosslinkable, end-capped polyester imide ether sulfone oligomers that have an acceptable combination of solvent resistance, toughness, impact resistance, strength, ease of processing, formability, and thermal resistance.
- Schiff base —CH ⁇ N—
- imidazole, thiazole, or oxazole linkages in the oligomer chain
- the linear, advanced composites formed with polyester oligomers of our copending application U.S. Ser. No. 726, 259 now abandoned can have semiconductive or conductive properties when appropriately doped.
- Conductive and semiconductive plastics have been extensively studied (see, e.g., U.S. Pat. Nos. 4,375,427; 4,338,222; 3,966,987; 4,344,869; and 4,344,870), but these polymers do not possess the blend of properties which are essential for aerospace applications. That is, the conductive polymers do not possess the blend of (1) toughness, (2) stiffness, (3) elasticity, (4) ease of processing, (5) impact resistance (and other matrix stress transfer capabilities), (6) retention of properties (over a broad range of temperatures), and (7) high temperature resistance that is desirable on aerospace advanced composites. These prior art composites are often too brittle.
- Thermally stable multidimensional oligomers having semiconductive or conductive properties when doped with suitable dopants are also known and are described in our copending applications (including U.S. Ser. No. 773,381 now U.S. Pat. No. 5,968.640 to Lubowitz, Sheppard, and Torre).
- the linear arms of the oligomers contain conductive linkages, such as Schiff base (—N ⁇ CH—) linkages, between aromatic groups. Sulfone and ether linkages are interspersed in the arms.
- Each arm is terminated with a mono- or difunctional end cap (i.e., a radical having one or two crosslinking sites) to allow controlled crosslinking upon heat-induced or chemically-induced curing.
- Polyamides of this same general type are described in U.S. Pat. No. 4,876,328; polyetherimides, in U.S Pat. No. 5,104,965 and polyamideimides, in U.S. Pat. No. 5,164,967.
- High performance, aerospace advanced composites can be prepared using crosslinkable, end-capped polyester sulfone oligomers of the present invention that have a desired blend of solvent resistance, toughness, impact resistance, strength, ease of processing, formability, and thermal resistance.
- the linear polyester oligomers include compounds selected from the group consisting of:
- E a crosslinkable (unsaturated) end cap to improve the solvent resistance of the cured oligomer in the advanced composite
- A a residue of a dicarboxylic acid halide
- a and B are each linear aromatic moieties that include electronegative (“sulfone”) linkages between aromatic radicals. Suitable electronegative linkages are selected from —SO 2 —, —S—, —O—, —CO—, —(CH 3 ) 2 C—, and —(CF 3 ) 2 C—.
- Schiff base (—CH ⁇ N—), oxazole, thiazole, and imidazole linkages can also be used in the backbone where semiconductive or conductive advanced composites are desired, since these linkages are conductive when suitably doped.
- Preferred Schiff base oligomers have the general formula:
- the crosslinkable end caps (E) are usually imidophenols or phenylimide acid halides that include a radical selected from the group consisting of:
- R 1 lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
- j 0, 1, or 2;
- G —CH 2 —, —O—, —S—, or —SO 2 —;
- T methallyl or allyl
- Multidimensional oligomers include an aromatic hub and three or more arms wherein each arm includes an ester linkage and an end cap.
- Blends of the linear or multidimensional oligomers comprise mixtures of the oligomers and one or more compatible, noncrosslinking polymers, and may be interpenetrating networks (IPNs).
- IPNs interpenetrating networks
- the oligomers and blends can be processed with suitable reinforcing additives in continuous or chopped form to make prepregs. Upon curing, the composites are relatively thermally stable, tough, and solvent resistant.
- the oligomers are prepared by simultaneously condensing the end caps, dicarboxylic acid halides, and dialcohols, (and hubs, if making multidimensional oligomers) in a suitable solvent under an inert atmosphere.
- Linear polyester oligomers of the present invention have the general formula:
- E a crosslinkable end cap radical preferably selected from the group consisting of:
- R 1 lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
- j 0, 1, or 2;
- G —CH 2 —, —O—, —S—, or —SO 2 —;
- T methallyl or allyl
- A the residue of a dicarboxylic acid halide (i.e. diacid halide);
- n an integer, usually between 0 and 50.
- the condensation can occur between alcohol (phenol) and acid halide functionalities or the corresponding acid or phenate groups.
- linear oligomers are usually prepared by reacting:
- the diacid halide (or dicarboxylic acid, if desired) generally includes an aromatic radical selected from the group consisting of:
- D —S—, —O—, —CO—, —SO 2 —, —(CH 3 ) 2 C—, —(CF 3 ) 2 C—, or mixtures thereof throughout the chain;
- R is selected from the group consisting of:
- R 1 a C 2 to C 12 divalent aliphatic alicyclic, or aromatic radical, and, preferably, phenyl (as described in U.S. Pat. No. 4,556,697).
- the preferred diacid halides include:
- Schiff base acid halides can be prepared by the condensation of aldehydes and amines in the general reaction scheme:
- Polyaryl or aryl diacid halides are preferred to achieve the highest thermal stabilities in the resulting oligomers and composites insofar as aliphatic bonds are not as thermally stable as aromatic bonds.
- Particularly preferred compounds include intermediate electronegative (“sulfone”) linkages to improve toughness of the resulting oligomers.
- “sulfone” linkages include —SO 2 —, —S—, —CO—, and —(CF 3 ) 2 C—, unless clearly limited to only —SO 2 —.
- dialcohol i.e. diol, dihydric phenol, or bisphenol
- dialcohol is generally selected from the group consisting of:
- L —CH 2 —, —(CH 3 ) 2 C—, —(CF 3 ) 2 C—, —O—, —S—, —SO 2 —, or —CO—;
- T and T 1 lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
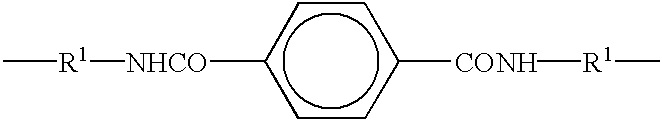
- the dialcohols also include hydroquinone; bisphenol-A; p,p′-biphenol; 4,4′-dihydroxydiphenylsulfide; 4,4′-dihydroxydiphenylether; 4,4′-dihydroxydiphenylisopropane; 4,4′-dihydroxydiphenylhexafluoropropane; a dialcohol having a Schiff base segment, the radical being selected from the group consisting of:
- R is selected from the group consisting of:
- n an integer, generally less than 5, and preferably 0 or 1;
- D any of —CO—, —SO 2 —, or —(CF 3 ) 2 C—.
- the other dialcohols can be used to add rigidity to the oligomer without significantly increasing the average formula weight, and, therefore, can increase the solvent resistance. Random or a block copolymers are possible.
- dialcohols may also be selected from the those described in U.S. Pat. Nos. 4,584,364; 3,262,914; or 4,611,048.
- Dialcohols of this nature are commercially available. Some may be easily synthesized by reacting halide intermediates with bis-phenates, such as by the reaction of 4,4′-dichlorodiphenylsulfone with bis(disodium biphenolate).
- Polyester Schiff base compounds are then prepared by condensing the Schiff base diols with end cap acid halides (or carboxylic acids), and diacid halide chains.
- polyesters are made by condensing the acid halides with suitable diols.
- Dopants for creating semiconductive or conductive composites are generally selected from two of the three categories commonly used to dope other polymers, namely (I) dispersions of alkali metals (for high activity) or (II) strong chemical oxidizers, particularly alkali perchlorates (for lower activity). Arsenic compounds and elemental halogens, while active dopants, are too dangerous for general usage, and are not recommended.
- N-type semiconductors result from doping with alkali metal dispersions.
- P-type semiconductive result from doping with elemental iodine or perchlorates.
- the polymers of the present invention exhibit greater oxidative stability and greater dimensional stability at elevated temperatures, greater impact strengths and greater dimensional stiffness over a wider range of temperatures, and toughness over those oligomers of the prior art.
- the polymers include mechanical swivels interspersed along the backbone coupling the rigid, conductive ribbons. Thermally stable swivels are afforded through “sulfone” linkages, particularly in phenoxyphenylsulfone or phenoxyphenylketone moieties.
- the conductive, semiconductive, and nonconductive polymers are usually made by mixing the appropriate proportions of the reactants in a polar organic solvent at temperatures between about ⁇ 30 to 100° C. Stepwise synthesis may also be used, if desired, as outlined for the Schiff base oligomers.
- the aromatic polyester sulfone resins synthesized in accordance with this invention have appreciable molecular weight between the reactive groups, the oligomers will retain sufficient plasticity to be processible during fabrication prior to crosslinking of the end caps to thermoset composites. If possible, thermoplastic formulations with even higher molecular weights should be synthesized.
- the polyesters preferably have MWs between about 5000-40,000, and, more preferably, between about 15,000-25,000.
- Blends can improve impact resistance of composites prepared from the crosslinkable oligomers without causing a significant loss of solvent resistance.
- the blends comprise mixtures of one or more crosslinkable oligomers and one or more polymers that are incapable of crosslinking, and generally comprise substantially equimolar amounts of one polymer and an oligomer having substantially identical backbones.
- the cross-linkable oligomer and comparable polymer can be blended together by mixing mutually soluble solutions of each. While the blend is preferably equimolar in the oligomer and polymer, the ratio of the oligomer and polymer can be adjusted to achieve the desired physical properties.
- the polymer in the blend usually has the same type of backbone (i.e. polyester sulfone) and same length backbone as the oligomer the properties of the composite formed from the blend can be adjusted by altering the ratio of formula weights for the polymer and oligomer.
- the oligomer and polymer generally have substantially identical repeating units, but the oligomer and polymer merely need be compatible in the solution prior to sweeping out as a prepreg. Of course, if the polymer and oligomer have identical backbones, compatibility in the blend is likely to occur.
- Quenching compounds can be employed, if desired to regulate the polymerization of the comparable polymer, so that it has an average formula weight substantially identical with the crosslinkable oligomer.
- an aromatic quenching compound such as phenol or benzoic acid chloride, is preferred.
- the noncrosslinking polymer can be made by the same synthetic method as the oligomer with the substitution of a quenching cap for the crosslinking end cap.
- Solvent resistance may decrease markedly if the comparable polymer is provided in large excess to the crosslinkable oligomer in the blend.
- the blends will generally comprise a mixture of one oligomer and a polymer of the same type.
- the polymer may, however, be from another family, such as an ester, imide, ether, or amide.
- the mixture may include multiple oligomers or multiple polymers, such as a three component mixture of oligomers and polymers. Other combinations will be recognized by those of ordinary skill in the art.
- the blends may be semi-interpenetrating networks of the general type described by Egli et al., “Semi-Interpenetrating Networks of LARC-TPI” available from NASA-Langley Research Center.
- the multidimensional oligomers then, have three or more chains radiating from the hub like rays or spokes. Each chain includes a crosslinking end-cap radical at its distal end to provide one or two crosslinking sites. Accordingly, the multidimensional oligomers have a greater number (i.e. higher density) of crosslinking bonds when cured than the linear oligomers.
- the hub will have three radiating chains to form a “Y” pattern. In some cases, four chains may be used. Including more chains leads to steric hindrance as the hub is too small to accommodate the radiating chains.
- a trisubstituted phenyl hub (such as phloroglucinol) is highly preferred with the chains being symmetrically placed about the hub.
- Biphenyl, naphthyl, or azaline (e.g., melamine) may also be used as the hub radical along the other aromatic moieties, if desired.
- Triazine derivatives described in U.S. Pat. No. 4,617,390 can be used as the hub.
- Other suitable triazine derivatives described in U.S. Pat. No. 4,574,154
- suitable hubs can be made by reacting a compound of the general formula:
- R 2 is a divalent hydrocarbon residue containing 1-12 carbon atoms (and, preferably, ethylene) with phthalic acid anhydride to form arms that include imide linkages and terminal acid functionalities (that can be converted to acid halides, if desired).
- acid halide (or carboxylic acid) hubs can be made by reacting polyol aromatic hubs, such as phloroglucinol, with nitrobenzoic acid or nitrophthalic acid to form ether linkages and active, terminal carboxylic acid functionalities.
- polyol aromatic hubs such as phloroglucinol
- nitrobenzoic acid or nitrophthalic acid to form ether linkages and active, terminal carboxylic acid functionalities.
- the nitrobenzoic acid products would have three active sites while the nitrophthalic acid products would have six; each having the respective formula:
- Hubs can also be formed by reacting the corresponding halo-hub (such a tribromobenzene) with aminophenol to form triamine compounds represented by the formula:
- Phenoxyphenyl sulfone arms radiating from a hub with a terminal amine, carboxylic acid, or hydroxyl group are also precursors for making multidimensional polyester oligomers of the present invention.
- the multidimensional oligomers are generally prepared by simultaneously reacting the hub with dialcohols; diacid halides dialcohols and diacid halides; and suitable end-cap monomers unreaction conditions similar to those for the linear oligomers. Care should be taken, however, to reduce the likelihood of undesirable side reactions. For example, a mixture of about 1 mole of the hub, w moles of dialcohol, w moles of diacid halide, and w moles of end-cap monomer is likely to form a complex mixture of multidimensional and linear oligomers and polymers. Accordingly, staged or stepwise reaction of the several reactants is preferred in this synthesis.
- the oligomers can be synthesized in a homogeneous reaction scheme wherein all the reactants are mixed at one time, or in a stepwise reaction scheme wherein the radiating chains are affixed to the hub and the product of the first reaction is subsequently reacted with the end cap groups.
- the hub may be reacted with end-capped arms that include one reactive, terminal functionality for linking the arm to the hub.
- Homogeneous reaction is preferred, resulting undoubtedly in a mixture of oligomers because of the complexity of the reactions.
- the products of the processes are preferred oligomer mixtures which can be used without further separation to form advanced composites.
- Blends of the multidimensional oligomers comparable to the blends of linear oligomers, can also be prepared, as will be understood.
- the preferred aromatic moieties in the linear or multidimensional oligomers are aryl groups, such as phenyl, biphenyl, and naphthyl.
- Other aromatic groups can be used, if desired, since their stabilized aromatic bonds should provide the desired thermal stability.
- azaline groups may be used.
- the aryl groups may include substituents, if desired, such as halogen, lower alkyl up to about 4 carbon atoms, lower alkoxy up to about 4 carbon atoms, or aryl side chains. Steric hindrance may arise in synthesizing the oligomers or in cross-linking the oligomers into cured composites, if the side chains are too large.
- the substituents may also effect the thermal stability of the resulting oligomers and composites.
- Unsubstituted phenyl groups are preferred for cost, convenience, and performance.
- the m-isomer may be preferred, since it tends to create a folded ribbon structure in the backbone.
- Prepregs of the oligomers or blends can be prepared by conventional techniques. While woven fabrics are the typical reinforcement, the fibers can be continuous or discontinuous (in chopped or whisker form) and may be ceramic, organic, carbon (graphite), or glass, as suited for the desired application.
- Composites can be formed by curing the oligomers or prepregs in conventional vacuum bag techniques.
- the oligomers can also be used as adhesives, varnishes, films, or coatings.
Abstract
High performance polyester sulfone oligomers are prepared for aerospace applications by condensing mono- or difunctional crosslinkable end caps (i.e. unsaturated hydrocarbons having one or two crosslinking sites) with dicarboxylic acid halides and dialcohols (i.e. diols). Multidimensional oligomers have an aromatic hub from which the polyester chains radiate. Blends of the linear and multidimensional oligomers can be made using compatible, non-crosslinking polymers. Prepregs and composites are formed from the oligomers or blends.
Description
This application is a divisional application based upon U.S. patent application Ser. No. 07/131,493, filed Dec. 23, 1987, now U.S. Pat. No. 5,705,598, which was a continuation-in-part application based upon U.S. patent application Ser. No. 06/726,259, filed Apr. 23, 1985, now abandoned.
The present invention relates to linear or multidimensional polyester oligomers and blends that include unsaturated hydrocarbon end caps to provide solvent resistance through crosslinking upon curing. Preferably the backbone includes electronegative (“sulfone”) segments that alternate with the ester linkages.
Recently, chemists have sought to synthesize oligomers for high performance advanced composites suitable for aerospace applications. These composites should exhibit solvent resistance, be tough, impact resistant, and strength, be easy to process, and be thermoplastic. Oligomers and composites that have thermo-oxidative stability, and, accordingly can be used at elevated temperatures, are particularly desirable.
While epoxy-based composites are suitable for many applications, their brittle nature and susceptibility to degradation make them inadequate for many aerospace applications, especially those applications which require thermally stable, tough composites. Accordingly, research has recently focused upon polyimide composites to achieve an acceptable balance between thermal stability, solvent resistance, and toughness. The maximum use temperatures of conventional polyimide composites, such as PMR-15, are still only about 600-625° F., since they have glass transition temperatures of about 690° F.
Linear polysulfone, polyether sulfone, polyester, and polyamide systems are also known, but each of these systems fails to provide as high thermal stability as is required in some aerospace applications.
There has been a progression of polyimide sulfone compounds synthesized to provide unique properties or combinations of properties. For example, Kwiatkowski and Brode synthesized maleic-capped, linear polyarylimides as disclosed in U.S. Pat. No. 3,839,287. Holub and Evans synthesized maleic- or nadic-capped, imido-substituted polyester compositions as disclosed in U.S. Pat. No. 3,729,446. We synthesized thermally stable polysulfone oligomers as disclosed in U.S. Pat. No. 4,476,184 or U.S. Pat. No. 4,536,559, and have continued to make advances with polyetherimidesulfones, polybenzoxazolesulfones (i.e., heterocycles), polybutadienesulfones, and “star” or “star-burst” multidimensional oligomers. We have shown surprisingly high glass transition temperatures and desirable physical properties in many of these oligomers and their composites, without losing ease of processing.
Multidimensional oligomers, such as disclosed in our U.S. Pat. No. 5,210,213; are easier to process than many other advanced composite oligomers since they can be handled at lower temperatures. Upon curing, however, the unsaturated phenylimide end caps crosslink so that the thermal resistance of the resulting composite is markedly increased with only a minor loss of stiffness, matrix stress transfer (impact resistance), toughness, elasticity, and other mechanical properties. Glass transition temperatures above 950° F. are achievable.
Commercial polyesters, when combined-with well-known diluents, such as styrene, do not exhibit satisfactory thermal and oxidative resistance to be useful for aircraft or aerospace applications. Polyarylesters are unsatisfactory, also, since the resins often are semicrystalline which makes them insoluble in laminating solvents, intractable in fusion, and subject to shrinking or warping during composite fabrication. Those polyarylesters that are soluble in conventional laminating solvents remain so in composite form, thereby limiting their usefulness in structural composites. The high concentration of ester groups contributes to resin strength and tenacity, but also makes the resin susceptible to the damaging effects of water absorption. High moisture absorption by commercial polyesters can lead to distortion of the composite when it is loaded at elevated temperature.
High performance, aerospace, polyester advanced composites, however, can be prepared using crosslinkable, end-capped polyester imide ether sulfone oligomers that have an acceptable combination of solvent resistance, toughness, impact resistance, strength, ease of processing, formability, and thermal resistance. By including Schiff base (—CH═N—), imidazole, thiazole, or oxazole linkages in the oligomer chain, the linear, advanced composites formed with polyester oligomers of our copending application U.S. Ser. No. 726, 259 now abandoned, can have semiconductive or conductive properties when appropriately doped.
Conductive and semiconductive plastics have been extensively studied (see, e.g., U.S. Pat. Nos. 4,375,427; 4,338,222; 3,966,987; 4,344,869; and 4,344,870), but these polymers do not possess the blend of properties which are essential for aerospace applications. That is, the conductive polymers do not possess the blend of (1) toughness, (2) stiffness, (3) elasticity, (4) ease of processing, (5) impact resistance (and other matrix stress transfer capabilities), (6) retention of properties (over a broad range of temperatures), and (7) high temperature resistance that is desirable on aerospace advanced composites. These prior art composites are often too brittle.
Thermally stable multidimensional oligomers having semiconductive or conductive properties when doped with suitable dopants are also known and are described in our copending applications (including U.S. Ser. No. 773,381 now U.S. Pat. No. 5,968.640 to Lubowitz, Sheppard, and Torre). The linear arms of the oligomers contain conductive linkages, such as Schiff base (—N═CH—) linkages, between aromatic groups. Sulfone and ether linkages are interspersed in the arms. Each arm is terminated with a mono- or difunctional end cap (i.e., a radical having one or two crosslinking sites) to allow controlled crosslinking upon heat-induced or chemically-induced curing.
Polyamides of this same general type are described in U.S. Pat. No. 4,876,328; polyetherimides, in U.S Pat. No. 5,104,965 and polyamideimides, in U.S. Pat. No. 5,164,967.
High performance, aerospace advanced composites can be prepared using crosslinkable, end-capped polyester sulfone oligomers of the present invention that have a desired blend of solvent resistance, toughness, impact resistance, strength, ease of processing, formability, and thermal resistance. The linear polyester oligomers include compounds selected from the group consisting of:
and are prepared by the condensation of acid halides (or acids) and dialcohols (bisphenols), wherein:
E=a crosslinkable (unsaturated) end cap to improve the solvent resistance of the cured oligomer in the advanced composite;
A=a residue of a dicarboxylic acid halide; and
B=a residue of a dialcohol.
Generally, A and B are each linear aromatic moieties that include electronegative (“sulfone”) linkages between aromatic radicals. Suitable electronegative linkages are selected from —SO2—, —S—, —O—, —CO—, —(CH3)2C—, and —(CF3)2C—. Schiff base (—CH═N—), oxazole, thiazole, and imidazole linkages can also be used in the backbone where semiconductive or conductive advanced composites are desired, since these linkages are conductive when suitably doped. Preferred Schiff base oligomers have the general formula:
wherein
R=an aromatic moiety or a short aryl chain including a plurality of aryl moieties linked with any of —CH2—, —SO2—, —S—, —O—, —CO—, —(CH3)2C—, or —(CF3)2C—; and q=0-4.
The crosslinkable end caps (E) are usually imidophenols or phenylimide acid halides that include a radical selected from the group consisting of:
wherein
R1=lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
j=0, 1, or 2;
G=—CH2—, —O—, —S—, or —SO2—;
T=methallyl or allyl; and
Me=methyl.
Multidimensional oligomers include an aromatic hub and three or more arms wherein each arm includes an ester linkage and an end cap.
Blends of the linear or multidimensional oligomers comprise mixtures of the oligomers and one or more compatible, noncrosslinking polymers, and may be interpenetrating networks (IPNs).
The oligomers and blends can be processed with suitable reinforcing additives in continuous or chopped form to make prepregs. Upon curing, the composites are relatively thermally stable, tough, and solvent resistant.
The oligomers are prepared by simultaneously condensing the end caps, dicarboxylic acid halides, and dialcohols, (and hubs, if making multidimensional oligomers) in a suitable solvent under an inert atmosphere.
wherein
R1=lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
j=0, 1, or 2;
G=—CH2—, —O—, —S—, or —SO2—;
T=methallyl or allyl;
Me=methyl;
A=the residue of a dicarboxylic acid halide (i.e. diacid halide);
B=the residue of a dialcohol; and
m=an integer, usually between 0 and 50.
The condensation can occur between alcohol (phenol) and acid halide functionalities or the corresponding acid or phenate groups.
The linear oligomers are usually prepared by reacting:
(b) n moles of an aromatic diacid halide or of a difunctional chain including a plurality of aryl rings linked with at least one linkage selected from the group consisting of —SO2—, —O—, —S—, —CO—, —(CH3)2C—, —(CF3)2C—, or mixtures thereof throughout the chain, the chain having an acid halide functionality on each end; and
(c) (n+1) moles of an aromatic dialcohol having terminal —OH functionalities,
or by reacting:
(b) (n+1) moles of an aromatic diacid halide or of a difunctional chain including a plurality of aryl rings linked with at least one linkage selected from the group consisting of —SO2—, —O—, —S—, —CO—, —(C3)2C—, —(CF3)2C—, or mixtures thereof throughout the chain, the chain having an acid halide functionality on each end; and
(c) n moles of an aromatic dialcohol (diol) having terminal —OH functionalities.
The diacid halide (or dicarboxylic acid, if desired) generally includes an aromatic radical selected from the group consisting of:
(a) phenyl; (b) naphthyl; (c) biphenyl;
wherein
D=—S—, —O—, —CO—, —SO2—, —(CH3)2C—, —(CF3)2 C—, or mixtures thereof throughout the chain;
(e) a divalent radical having conductive linkages, illustrated by Schiff base compounds selected from the group consisting of:
wherein
R is selected from the group consisting of:
phenyl; biphenyl; naphthyl; or
wherein W=—SO2— or —CH2—; and q=0-4; or
wherein R1=a C2 to C12 divalent aliphatic alicyclic, or aromatic radical, and, preferably, phenyl (as described in U.S. Pat. No. 4,556,697).
Schiff base acid halides can be prepared by the condensation of aldehydes and amines in the general reaction scheme:
or similar schemes.
Thiazole, oxazole, or imidazole linkages, especially between aryl groups, nay also be used in the conductive or semiconductive oligomers along with or in place of the Schiff base linkages.
Other diacid halides that can be used, but that are not preferred, are disclosed in U.S. Pat. No. 4,504,632, and include:
adipylchloride,
malonyl chloride,
succinyl chloride,
glutaryl chloride,
pimelic acid dichloride,
suberic acid dichloride,
azelaic acid dichloride,
sebacic acid dichloride,
dodecandioic acid dichloride,
phthaloyl chloride,
isophthaloyl chloride,
terephthaloyl chloride,
1,4-naphthalene dicarboxylic acid dichloride, and
4,4′-diphenylether dicarboxylic acid dichloride.
Polyaryl or aryl diacid halides are preferred to achieve the highest thermal stabilities in the resulting oligomers and composites insofar as aliphatic bonds are not as thermally stable as aromatic bonds. Particularly preferred compounds include intermediate electronegative (“sulfone”) linkages to improve toughness of the resulting oligomers. For purposes of this description, “sulfone” linkages include —SO2—, —S—, —CO—, and —(CF3)2C—, unless clearly limited to only —SO2—.
The dialcohol (i.e. diol, dihydric phenol, or bisphenol) is generally selected from the group consisting of:
HO—Ar—OH;
HO—Ar—L—Ar′—L—Ar—OH;
HO—Ar′—L—Ar—L—Ar′—OH;
wherein
T and T1=lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
q=0-4;
k=0-3; and
j=0, 1, or 2,
The dialcohols also include hydroquinone; bisphenol-A; p,p′-biphenol; 4,4′-dihydroxydiphenylsulfide; 4,4′-dihydroxydiphenylether; 4,4′-dihydroxydiphenylisopropane; 4,4′-dihydroxydiphenylhexafluoropropane; a dialcohol having a Schiff base segment, the radical being selected from the group consisting of:
wherein R is selected from the group consisting of:
phenyl;
biphenyl;
naphthyl; or
wherein W=—CH2— or —SO2—; or
wherein
L is as defined above;
Me=methyl;
m=an integer, generally less than 5, and preferably 0 or 1; and
D=any of —CO—, —SO2—, or —(CF3)2C—.
While bisphenol-A is preferred in the etherimide synthesis (because of cost and availability), the other dialcohols can be used to add rigidity to the oligomer without significantly increasing the average formula weight, and, therefore, can increase the solvent resistance. Random or a block copolymers are possible.
Furthermore, the dialcohols may also be selected from the those described in U.S. Pat. Nos. 4,584,364; 3,262,914; or 4,611,048.
Dialcohols of this nature are commercially available. Some may be easily synthesized by reacting halide intermediates with bis-phenates, such as by the reaction of 4,4′-dichlorodiphenylsulfone with bis(disodium biphenolate).
Schiff base diols are prepared by the condensation of aldehydes and amines under the general reaction schemes:
Polyester Schiff base compounds are then prepared by condensing the Schiff base diols with end cap acid halides (or carboxylic acids), and diacid halide chains.
or similar schemes as with the diols. The resulting polyesters are made by condensing the acid halides with suitable diols.
While illustrated for preparing Schiff base diacid halides and diols, these reaction schemes are representative of the processes used to prepare any of the diacid halides or diols used in the polymerization reactions. Inexpensive, commercially available starting compounds are used, such as aminophenol.
Dopants for creating semiconductive or conductive composites are generally selected from two of the three categories commonly used to dope other polymers, namely (I) dispersions of alkali metals (for high activity) or (II) strong chemical oxidizers, particularly alkali perchlorates (for lower activity). Arsenic compounds and elemental halogens, while active dopants, are too dangerous for general usage, and are not recommended.
The dopants react with the polymers to form charge transfer complexes. N-type semiconductors result from doping with alkali metal dispersions. P-type semiconductive result from doping with elemental iodine or perchlorates.
While research into conductive or semiconductive polymers has been intense, the resulting compounds, mainly polyacetylenes, polyphenelenes, and polyvinylacetylenes, are unsatisfactory for aerospace applications because the polymers are:
(a) unstable in air;
(b) unstable at high temperatures;
(c) brittle after doping;
(d) toxic because of the dopants; or
(e) intractable.
The polymers of the present invention exhibit greater oxidative stability and greater dimensional stability at elevated temperatures, greater impact strengths and greater dimensional stiffness over a wider range of temperatures, and toughness over those oligomers of the prior art.
While conventional theory holds that semiconductive polymers should have low ionization potentials, long conjugation lengths, and planar backbones, there is an inherent trade-off, between conductivity and toughness or ease of processing, if these constraints are followed. To overcome the processing and toughness shortcomings common with Schiff base, polyoxazole, polyimidazole; and polythiazole polymers, in the present invention, the polymers include mechanical swivels interspersed along the backbone coupling the rigid, conductive ribbons. Thermally stable swivels are afforded through “sulfone” linkages, particularly in phenoxyphenylsulfone or phenoxyphenylketone moieties.
The conductive, semiconductive, and nonconductive polymers are usually made by mixing the appropriate proportions of the reactants in a polar organic solvent at temperatures between about −30 to 100° C. Stepwise synthesis may also be used, if desired, as outlined for the Schiff base oligomers.
Because the aromatic polyester sulfone resins synthesized in accordance with this invention have appreciable molecular weight between the reactive groups, the oligomers will retain sufficient plasticity to be processible during fabrication prior to crosslinking of the end caps to thermoset composites. If possible, thermoplastic formulations with even higher molecular weights should be synthesized. The polyesters preferably have MWs between about 5000-40,000, and, more preferably, between about 15,000-25,000.
Because it may be difficult to make diols or diacid chlorides having intermediate benzimidazole, benzoxazole, or benzothiazole linkages, and because the chemistry for Schiff base compounds is well understood, it is preferred to use Schiff base segments in the oligomers for semiconductive or conductive applications.
Solubility of the oligomers becomes an increasing problem as the length of the chains increases. Therefore, shorter chains are preferred, if the resulting oligomers remain easily processed. That is, the chains should be long enough to yield thermoplastic characteristics but short enough to keep the oligomers soluble during the reaction sequence.
Blends can improve impact resistance of composites prepared from the crosslinkable oligomers without causing a significant loss of solvent resistance. The blends comprise mixtures of one or more crosslinkable oligomers and one or more polymers that are incapable of crosslinking, and generally comprise substantially equimolar amounts of one polymer and an oligomer having substantially identical backbones. The cross-linkable oligomer and comparable polymer can be blended together by mixing mutually soluble solutions of each. While the blend is preferably equimolar in the oligomer and polymer, the ratio of the oligomer and polymer can be adjusted to achieve the desired physical properties.
Although the polymer in the blend usually has the same type of backbone (i.e. polyester sulfone) and same length backbone as the oligomer the properties of the composite formed from the blend can be adjusted by altering the ratio of formula weights for the polymer and oligomer. The oligomer and polymer generally have substantially identical repeating units, but the oligomer and polymer merely need be compatible in the solution prior to sweeping out as a prepreg. Of course, if the polymer and oligomer have identical backbones, compatibility in the blend is likely to occur.
Quenching compounds can be employed, if desired to regulate the polymerization of the comparable polymer, so that it has an average formula weight substantially identical with the crosslinkable oligomer. For thermal stability, an aromatic quenching compound, such as phenol or benzoic acid chloride, is preferred.
The noncrosslinking polymer can be made by the same synthetic method as the oligomer with the substitution of a quenching cap for the crosslinking end cap.
While the best blends are probably those in which the backbones are essentially identical and of modest formula weight and those in which the oligomer and polymer are in equimolar proportions, other compositions may be prepared, as will be recognized by those of ordinary skill in the art.
Solvent resistance may decrease markedly if the comparable polymer is provided in large excess to the crosslinkable oligomer in the blend.
As discussed, the blends will generally comprise a mixture of one oligomer and a polymer of the same type. The polymer may, however, be from another family, such as an ester, imide, ether, or amide. The mixture may include multiple oligomers or multiple polymers, such as a three component mixture of oligomers and polymers. Other combinations will be recognized by those of ordinary skill in the art.
The blends may be semi-interpenetrating networks of the general type described by Egli et al., “Semi-Interpenetrating Networks of LARC-TPI” available from NASA-Langley Research Center.
Multidimensional polyesters (of the general formula: Ar—(—Q)w, wherein Ar=an aromatic radical of valence w, Q=a polyester radical having a crosslinking end cap and w is an integer greater than or equal to 3) can also be prepared by linking polyester sulfone or polyester arms to an aromatic hub. The multidimensional oligomers, then, have three or more chains radiating from the hub like rays or spokes. Each chain includes a crosslinking end-cap radical at its distal end to provide one or two crosslinking sites. Accordingly, the multidimensional oligomers have a greater number (i.e. higher density) of crosslinking bonds when cured than the linear oligomers.
Usually the hub will have three radiating chains to form a “Y” pattern. In some cases, four chains may be used. Including more chains leads to steric hindrance as the hub is too small to accommodate the radiating chains. A trisubstituted phenyl hub (such as phloroglucinol) is highly preferred with the chains being symmetrically placed about the hub. Biphenyl, naphthyl, or azaline (e.g., melamine) may also be used as the hub radical along the other aromatic moieties, if desired.
Triazine derivatives described in U.S. Pat. No. 4,617,390 (or the corresponding acid halides) can be used as the hub. Other suitable triazine derivatives (described in U.S. Pat. No. 4,574,154) that are suitable hubs can be made by reacting a compound of the general formula:
wherein R2is a divalent hydrocarbon residue containing 1-12 carbon atoms (and, preferably, ethylene) with phthalic acid anhydride to form arms that include imide linkages and terminal acid functionalities (that can be converted to acid halides, if desired).
Other acid halide (or carboxylic acid) hubs can be made by reacting polyol aromatic hubs, such as phloroglucinol, with nitrobenzoic acid or nitrophthalic acid to form ether linkages and active, terminal carboxylic acid functionalities. The nitrobenzoic acid products would have three active sites while the nitrophthalic acid products would have six; each having the respective formula:
wherein Φ=phenyl. Of course other nitro/acids can be used.
Hubs can also be formed by reacting the corresponding halo-hub (such a tribromobenzene) with aminophenol to form triamine compounds represented by the formula:
wherein Φ=phenyl; the hub being characterized by an intermediate ether and imide linkage connecting aromatic groups. Thio-analogs are also contemplated, in accordance with U.S. Pat. No. 3,933,862.
Phenoxyphenyl sulfone arms radiating from a hub with a terminal amine, carboxylic acid, or hydroxyl group are also precursors for making multidimensional polyester oligomers of the present invention.
The best results are likely to occur when the hub is phloroglucinol or cyuranic acid. In either case a suitable end-cap monomer (phenol or acid halide) can be reacted with the hub to form “short-armed,” multidimensional oligomers having three or six crosslinking sites. These compounds are the simplest multidimensional oligomers and are relatively inexpensive to synthesize.
The multidimensional oligomers are generally prepared by simultaneously reacting the hub with dialcohols; diacid halides dialcohols and diacid halides; and suitable end-cap monomers unreaction conditions similar to those for the linear oligomers. Care should be taken, however, to reduce the likelihood of undesirable side reactions. For example, a mixture of about 1 mole of the hub, w moles of dialcohol, w moles of diacid halide, and w moles of end-cap monomer is likely to form a complex mixture of multidimensional and linear oligomers and polymers. Accordingly, staged or stepwise reaction of the several reactants is preferred in this synthesis.
Thus, the oligomers can be synthesized in a homogeneous reaction scheme wherein all the reactants are mixed at one time, or in a stepwise reaction scheme wherein the radiating chains are affixed to the hub and the product of the first reaction is subsequently reacted with the end cap groups. Of course, the hub may be reacted with end-capped arms that include one reactive, terminal functionality for linking the arm to the hub. Homogeneous reaction is preferred, resulting undoubtedly in a mixture of oligomers because of the complexity of the reactions. The products of the processes (even without distillation or isolation of individual species) are preferred oligomer mixtures which can be used without further separation to form advanced composites.
Blends of the multidimensional oligomers, comparable to the blends of linear oligomers, can also be prepared, as will be understood.
The preferred aromatic moieties in the linear or multidimensional oligomers are aryl groups, such as phenyl, biphenyl, and naphthyl. Other aromatic groups can be used, if desired, since their stabilized aromatic bonds should provide the desired thermal stability. For example, azaline groups may be used. As discussed earlier, the aryl groups may include substituents, if desired, such as halogen, lower alkyl up to about 4 carbon atoms, lower alkoxy up to about 4 carbon atoms, or aryl side chains. Steric hindrance may arise in synthesizing the oligomers or in cross-linking the oligomers into cured composites, if the side chains are too large. The substituents may also effect the thermal stability of the resulting oligomers and composites. Unsubstituted phenyl groups are preferred for cost, convenience, and performance. The m-isomer may be preferred, since it tends to create a folded ribbon structure in the backbone.
Prepregs of the oligomers or blends can be prepared by conventional techniques. While woven fabrics are the typical reinforcement, the fibers can be continuous or discontinuous (in chopped or whisker form) and may be ceramic, organic, carbon (graphite), or glass, as suited for the desired application.
Composites can be formed by curing the oligomers or prepregs in conventional vacuum bag techniques. The oligomers can also be used as adhesives, varnishes, films, or coatings.
While preferred embodiments have been described, those skilled in the art will readily recognize alterations, variations, and modifications which might be made without departing from the inventive concept. Therefore, the claims should be interpreted liberally with the support of the full range of equivalents known to those of ordinary skill on the basis of this description. The claims should be limited only as is necessary in view of the pertinent prior art.
Claims (42)
wherein the polyester oligomer is formed by the simultaneous condensation of diacid halides, dialcohols, and end-cap monomers and wherein
E is a crosslinkable end cap radical to improve the solvent resistance of the cured oligomer in the advanced composite and is:
R1 is lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
j is 0, 1, or 2;
G is —CH2—, —O—, —S—, or —SO2—;
T is methallyl or allyl;
Me is methyl;
A is a residue of a dicarboxylic acid halide; and
B is a residue of a dialcohol.
2. The oligomer of claim 1 wherein both A and B are linear aromatic radicals having a plurality of aromatic groups linked together by linkages selected from the group consisting of: —CO—; —O—; —S—; —SO2—; —(CH3)2C—; —(CF3)2C—; —CH═N—; oxazole; thiazole; and imidazole.
3. The oligomer of claim 2 wherein the aromatic groups of both A and B are selected from the group consisting of phenyl, biphenyl, naphthyl, and mixtures thereof.
6. A prepreg comprising the oligomer of claim 1 and a reinforcing additive in fiber or particulate form.
7. A composite comprising the cured prepreg of claim 6 .
8. A blend comprising the oligomer of claim 1 and a compatible, non-crosslinking polymer.
9. A composite comprising the cured blend of claim 8 .
10. The blend of claim 8 comprising substantially equimolar amounts of the oligomer and polymer.
11. The blend of claim 8 wherein the polymer is a polyester sulfone.
12. The blend of claim 8 wherein the average formula weight of the oligomer is less than that of the polymer.
13. A prepreg comprising the blend of claim 8 and a reinforcing additive in fiber or particulate form.
14. A composite comprising the cured prepreg of claim 13 .
15. A composite comprising the cured oligomer of claim 1 .
16. An oligomer useful for forming advanced composites, comprising the product of the process of reacting about:
(a) 2 moles of a crosslinkable end cap acid halide of the general formula: Di—Ø—COX, wherein
Ø is phenyl;
X is halogen;
i is 1 or 2;
R1 is lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
j is 0, 1, or 2;
G is —CH2—, —O—, —S—, or —SO2—;
T is methallyl or allyl; and
Me is methyl;
(b) n moles of a dicarboxylic acid halide or of a chain including a plurality of aryl rings linked with at least one linkage selected from the group consisting of —SO2—, —O—, —S—, —CO—, —(CH3)2C—, —(CF3)2C—, and mixtures thereof throughout the chain, the chain having an acid halide functionality on each end; and
(c) (n+1) moles of a diol having terminal —OH functionalities, wherein n is an integer greater than or equal to 1.
17. The oligomer of claim 16 wherein the diol is selected from the group consisting of:
HO—Ar—OH;
HO—Ar—L—Ar′—L—Ar—OH; and
HO—Ar′—L—Ar—L—Ar′—OH;
T and T1=lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
q=0-4;
k=0-3; and
j=0, 1, or 2.
18. The oligomer of claim 16 wherein the diol is hydroquinone, bisphenol-A, p,p′-biphenol, 4,4′-dihydroxydiphenylsulfide, 4,4′-dihydroxydiphenylether, 4,4′-dihydroxydiphenylisopropane, 4,4′-dihydroxydiphenylhexafluoropropane,
wherein L=—S—, —O—, —CO—, —SO2—, —(CH3)2C—; or —(CF3)2C—;
Me=methyl;
m=an integer less than or equal to 5; and
D=any of —CO—, —SO2—, or —(CF3)2C—.
20. The oligomer of claim 18 wherein the dicarboxylic acid halide includes a radical selected from the group consisting of:
(a) phenyl;
(b) naphthyl;
(c) biphenyl;
wherein D is —S—, —O—, —CO—, —SO2—, —(CH3)2C—, —(CF3)2C—, or mixtures thereof throughout the chain;
(e) a divalent radical having conductive linkages, illustrated by Schiff base compounds selected from the group consisting of:
wherein R is:
wherein W is —SO2— or —CH2—; and q is 0-4; and
wherein R1=a C2 to C12 divalent aliphatic, alicyclic, or aromatic radical.
23. The oligomer of claim 16 wherein the dicarboxylic acid halide includes a radical selected from the group consisting of:
(a) phenyl;
(b) naphthyl;
(c) biphenyl;
wherein D is —S—, —O—, —CO—, —SO2—, —(CH3)2C—, —(CF3)2C—, and mixtures thereof throughout the chain;
(e) a divalent radical having conductive linkages, illustrated by Schiff base compounds selected from the group consisting of:
wherein W is —SO2— or —CH2—; and q is 0-4; and
wherein R1 is a C2 to C12 divalent aliphatic, alicyclic, or aromatic radical.
26. A prepreg comprising the oligomer of claim 16 and a reinforcing additive in fiber or particulate form.
27. A prepreg comprising the oligomer of claim 20 and a reinforcing additive in fiber or particulate form.
28. A blend comprising the oligomer of claim 16 and a compatible, non-crosslinking polymer.
29. A prepreg comprising, the blend of claim 28 and a reinforcing additive in fiber or particulate form.
30. A blend comprising the oligomer of claim 22 and a compatible, non-crosslinking polymer.
31. A prepreg comprising the blend of claim 30 and a reinforcing additive in fiber or particulate form.
wherein the polyester oligomer is formed by the simultaneous condensation of diacid halides, dialcohols, and end-cap monomers and wherein
E is a crosslinkable end cap radical to improve the solvent resistance of the cured oligomer in the advanced composite and is:
R1 is lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
j is 0, 1, or 2;
G is —CH2—, —O—, —S—, or —SO2—;
T is methallyl or allyl;
Me is methyl;
A is a residue of a dicarboxylic acid halide;
B is a residue of a dialcohol;
wherein both A and B are linear aromatic radicals having a plurality of aromatic groups linked together by linkages selected from the group consisting of: —CO—; —O—; —S—; —SO2—; —(CH3)2C—; —(CF3)2C—; —CH═N—; oxazole; thiazole; and imidazole; wherein the aromatic groups of both A and B are selected from the group consisting of phenyl, biphenyl, naphthyl, and mixtures thereof;
wherein D is:
—SO2—, —S—, —CO—, —O—, —(CH3)2C—, —(CF3)2C—, or mixtures thereof, and
wherein B includes one or more linkages selected from the group consisting of —CH═N oxazole, thiazole, and imidazole.
36. The oligomer of claim 35 wherein R is a phenoxyphenylsulfone.
37. An oligomer useful for forming advanced composites, comprising the product of the process of reacting about:
(a) 2 moles of a crosslinkable end cap phenol of the general formula:
(b) (n+1) moles of a dicarboxylic acid halide/or of a chain including a plurality of aryl rings linked with at least one linkage selected from the group consisting of —SO2—, —O—, —S—, —CO—, —(CH3)2C—, —(CF3)2C—, and mixtures thereof throughout the chain, the chain having an acid halide functionality on each end; and
(c) n moles of a diol having terminal —OH functionalities,
R1 is lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen, or mixtures thereof;
j is 0, 1, or 2;
G is —CH2—, —O—, —S—, or —SO2—;
T is methallyl for allyl;
Me is methyl;
R is phenyl; biphenyl; naphthyl; or a compound of the general formula:
W is —CH2— or —SO2—; and
q is 0-4.
38. The oligomer of claim 37 wherein the diol is:
hydroquinone,
bisphenol A,
p,p′-biphenol,
4,4′-dihydroxydiphenylsulfide,
4,4′-dihydroxydiphenylsulfone,
4,4′-dihydroxydiphenylether,
4,4′-dihydroxydiphenylisopropane,
4,4′-dihydroxydiphenylhexafluoropropane,
HO—Ar—OH,
HO—Ar—L—Ar′—L—Ar—OH,
T and T1=lower alkyl, lower alkoxy, aryl, aryloxy, substituted alkyl, substituted aryl, halogen or mixture thereof;
q=0-4;
k 0-3;
j=0, 1, or 2;
Me=methyl;
m=an integer less than or equal to 5; and
D=—CO—, —SO2, —(CF3)2C—, or mixtures thereof.
39. The oligomer of claim 38 wherein the dicarboxylic acid halide includes a radical selected from the group consisting of:
a) phenyl;
(b) naphthyl;
(c) biphenyl;
wherein D is —S—, —O—, —CO—, —SO2—, —(CH3)2C—, —(CF3)2C—, or mixtures thereof throughout the chain;
(e) a divalent radical having conductive linkages, illustrated by Schiff base compounds selected from the group consisting of:
wherein W is —SO2— or —CH2—; and q is 0-4; and
wherein R1 is a C2 to C12 divalent aliphatic alicyclic, or aromatic radical.
40. The oligomer of claim 37 wherein the dicarboxylic acid halide includes a radical selected from the group consisting of:
(a) phenyl;
(b) naphthyl;
(c) biphenyl;
wherein D=—S—, —O—, —CO—, —SO2—, —(CH3)2C—, (CF3)2C—, and mixtures thereof throughout the chain;
(e) a divalent radical having conductive linkages, illustrated by Schiff base compounds selected from the group consisting of:
wherein R is selected from the group consisting of:
wherein W=—SO2— or —CH2—; and q=0-4; and
wherein R=a C2 to C12 divalent aliphatic, alicyclic, or aromatic radical.
41. A prepreg comprising the oligomer of claim 39 and a reinforcing additive in fiber or particulate form.
42. A prepreg comprising the oligomer of claim 37 and a reinforcing additive in fiber or particulate form.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/463,437 US6583255B1 (en) | 1985-04-23 | 1995-06-05 | Polyester oligomer |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US72625985A | 1985-04-23 | 1985-04-23 | |
| US07/137,493 US5705598A (en) | 1985-04-23 | 1987-12-23 | Polyester sulfone oligomers and blends |
| US08/463,437 US6583255B1 (en) | 1985-04-23 | 1995-06-05 | Polyester oligomer |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US07/137,493 Division US5705598A (en) | 1981-11-13 | 1987-12-23 | Polyester sulfone oligomers and blends |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US6583255B1 true US6583255B1 (en) | 2003-06-24 |
Family
ID=26835303
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US07/137,493 Expired - Fee Related US5705598A (en) | 1981-11-13 | 1987-12-23 | Polyester sulfone oligomers and blends |
| US08/572,755 Expired - Fee Related US5739256A (en) | 1985-04-23 | 1995-06-05 | Method for making multidimensional polyester oligomers |
| US08/463,437 Expired - Fee Related US6583255B1 (en) | 1985-04-23 | 1995-06-05 | Polyester oligomer |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US07/137,493 Expired - Fee Related US5705598A (en) | 1981-11-13 | 1987-12-23 | Polyester sulfone oligomers and blends |
| US08/572,755 Expired - Fee Related US5739256A (en) | 1985-04-23 | 1995-06-05 | Method for making multidimensional polyester oligomers |
Country Status (1)
| Country | Link |
|---|---|
| US (3) | US5705598A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8822628B2 (en) | 2012-09-24 | 2014-09-02 | Ticona Llc | Crosslinkable liquid crystalline polymer |
| US8853342B2 (en) | 2012-09-24 | 2014-10-07 | Ticona Llc | Crosslinkable liquid crystalline polymer |
| US9145519B2 (en) | 2012-09-24 | 2015-09-29 | Ticona Llc | Crosslinkable aromatic polyester |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1370249A4 (en) * | 2001-02-26 | 2006-05-03 | Univ Duke | Novel dendriticpolymers and their biomedical uses |
| US20040131582A1 (en) * | 2002-02-26 | 2004-07-08 | Grinstaff Mark W. | Novel dendritic polymers and their biomedical uses |
| US20080300374A1 (en) * | 2007-05-31 | 2008-12-04 | The Boeing Company | Dinadic phenyl amine reactive endcaps |
| US8309663B2 (en) | 2007-05-31 | 2012-11-13 | The Boeing Company | Water-entrained-polyimide chemical compositions for use in high-performance composite fabrication |
| US7825211B2 (en) * | 2007-06-22 | 2010-11-02 | The Boeing Company | Single-step-processable polyimides |
| US9803050B2 (en) * | 2014-10-08 | 2017-10-31 | Ticona Llc | Dispersant for use in synthesis of polyaryletherketones |
Citations (456)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB907105A (en) | 1958-11-14 | 1962-10-03 | Nat Res Dev | New polymers having azo-links |
| US3105839A (en) | 1960-12-16 | 1963-10-01 | Ciba Ltd | Bicyclo (2. 2. 1)hept-5-ene-2, 3-dicarboxylic acid anhydrides |
| US3236808A (en) | 1961-01-03 | 1966-02-22 | Borg Warner | Polysulfonate copolymers |
| US3236705A (en) | 1962-07-02 | 1966-02-22 | Monsanto Res Corp | Solid polymeric solutions containing metal salts such as lithium perchlorate |
| US3262914A (en) | 1965-03-16 | 1966-07-26 | Borg Warner | Sulfonate-carboxylate copolymers |
| US3265708A (en) | 1961-03-01 | 1966-08-09 | Us Rubber Co | Nu-parahydroxyphenylmaleimide |
| US3267081A (en) | 1962-09-19 | 1966-08-16 | Koppers Co Inc | Benzothiazole dicarboxylic acids and polymers derived therefrom |
| US3313783A (en) | 1962-07-20 | 1967-04-11 | Teijin Ltd | Process for preparation of polybenzimidazoles |
| GB1069061A (en) | 1965-02-10 | 1967-05-17 | Ilford Ltd | N-aminophenyl-phthalimido derivatives and their use in photographic colour developing |
| US3354129A (en) | 1963-11-27 | 1967-11-21 | Phillips Petroleum Co | Production of polymers from aromatic compounds |
| US3355272A (en) | 1964-11-24 | 1967-11-28 | Union Carbide Corp | Abrasive compositions and structures thereof comprising a thermosetting resin and a thermoplastic polyarylene polyether |
| GB1099096A (en) | 1965-12-10 | 1968-01-17 | Monsanto Chemicals | Nitrogen-containing aromatic compounds |
| US3386969A (en) | 1962-11-19 | 1968-06-04 | Whittaker Corp | Fusible polybenzimidazoles |
| US3408349A (en) | 1963-12-04 | 1968-10-29 | American Cyanamid Co | Azine ion-radical salts |
| US3431235A (en) | 1966-03-02 | 1969-03-04 | Trw Inc | Polydiene resins including cyclized polydiene resins,method of manufacture and uses |
| US3435003A (en) | 1966-06-01 | 1969-03-25 | Du Pont | Cross-linked thermally reversible polymers produced from condensation polymers with pendant furan groups cross-linked with maleimides |
| US3448084A (en) | 1964-06-10 | 1969-06-03 | Gulf Oil Corp | Polyesters from tris(2-hydroxyethyl) isocyanurate |
| US3449442A (en) | 1967-11-13 | 1969-06-10 | Mobil Oil Corp | 2,4-diphenoxybromobenzene and 2,4-diphenoxychlorobenzene as new compounds and synthetic lubricants |
| US3449467A (en) | 1965-06-03 | 1969-06-10 | Union Carbide Corp | Hydroxyl terminated polyester oligomers |
| US3450711A (en) | 1965-07-01 | 1969-06-17 | American Cyanamid Co | Bis-imide containing diepoxide compounds |
| US3453236A (en) | 1967-01-18 | 1969-07-01 | Ashland Oil Inc | Aromatic polyimideamides prepared from aromatic tetracarboxylic acid dianhydrides and aminoaromatic hydrazides |
| US3454673A (en) | 1965-06-05 | 1969-07-08 | Ella Dannenbaum | Preparation of hardenable nitrogen containing synthetic resinous products |
| US3458486A (en) | 1964-09-04 | 1969-07-29 | Phillips Petroleum Co | Production of polymers from cyclic compounds |
| US3461461A (en) | 1965-11-01 | 1969-08-12 | Upjohn Co | 6-amino-4-(substituted amino)-1,2-dihydro-1-hydroxy-2-iminopyrimidines |
| US3528950A (en) | 1967-07-03 | 1970-09-15 | Trw Inc | Polyimide polymers |
| US3530087A (en) | 1967-02-21 | 1970-09-22 | Ciba Ltd | Adhesive compositions of polyepoxide and polysulfones |
| US3536270A (en) | 1967-12-04 | 1970-10-27 | Lincoln Tool & Mfg Co | Span control for a stator winding machine |
| US3562223A (en) | 1967-07-13 | 1971-02-09 | Rhone Poulenc Sa | Cross-linked resins |
| US3563951A (en) | 1967-03-06 | 1971-02-16 | Bayer Ag | Polyimides of high molecular weight aromatic polyether diamines |
| US3565549A (en) | 1968-06-20 | 1971-02-23 | Trw Inc | Process for preparing reinforced resinous structures |
| US3592841A (en) | 1967-12-01 | 1971-07-13 | Richardson Co | Keto-carboxylic acids |
| US3598768A (en) | 1969-07-09 | 1971-08-10 | Monsanto Co | Hetero-aromatic azopolymers |
| US3609181A (en) | 1964-05-28 | 1971-09-28 | Us Air Force | Polyesters of polyoxytetramethylene glycol and certain dioic acids |
| US3616193A (en) | 1968-05-14 | 1971-10-26 | Trw Inc | Thermoset polydiene resin adhesive bonded laminates and methods of making same |
| US3624042A (en) | 1962-11-29 | 1971-11-30 | Aerojet General Co | Carboxy terminated polymer and cured derivative thereof |
| US3631222A (en) | 1970-01-02 | 1971-12-28 | Minnesota Mining & Mfg | Polyarylsulfone graft and block copolymers with chains of polyamide acid or polyimide |
| US3632428A (en) | 1967-11-30 | 1972-01-04 | Trw Inc | Thermally stable coatings |
| US3635891A (en) | 1966-07-14 | 1972-01-18 | Trw Inc | Internally plasticized polyidene resins |
| US3641207A (en) | 1969-07-15 | 1972-02-08 | Uniroyal Inc | Thermoplastic resin blend of polysulfone resin and an ethylenepropylene terpolymer or graft derivative thereof |
| US3647529A (en) | 1967-07-10 | 1972-03-07 | Trw Inc | Reinforced structural plastics |
| US3652710A (en) | 1969-07-01 | 1972-03-28 | Gen Electric | Curable polyaryleneoxide compositions |
| US3658938A (en) | 1970-12-30 | 1972-04-25 | Union Carbide Corp | Polyamide-imide/polysulfone composites |
| US3663507A (en) | 1970-01-02 | 1972-05-16 | Minnesota Mining & Mfg | Linear polyarylsulfones having functional groups thereon |
| US3689464A (en) | 1970-12-18 | 1972-09-05 | Gen Electric | Imido-substituted polyamide compositions |
| US3697308A (en) | 1969-11-10 | 1972-10-10 | Trw Inc | Boron reinforced polyimide composites |
| US3697345A (en) | 1969-11-10 | 1972-10-10 | Trw Inc | Process for preparing polyimide laminates |
| US3699074A (en) | 1970-08-21 | 1972-10-17 | Trw Inc | Polyimide prepolymer molding powders |
| US3699075A (en) | 1970-10-28 | 1972-10-17 | Trw Inc | Soluble high molecular weight polyimide |
| US3708370A (en) | 1971-08-06 | 1973-01-02 | Trw Inc | Process for bonding surfaces with polyimide prepolymers |
| US3708439A (en) | 1971-09-28 | 1973-01-02 | Upjohn Co | Polybenzimidazoles |
| US3708459A (en) | 1970-06-24 | 1973-01-02 | Trw Inc | Molding power prepolymers |
| US3729446A (en) | 1969-07-01 | 1973-04-24 | Gen Electric | Imido-substituted polyester compositions |
| US3745149A (en) | 1971-09-29 | 1973-07-10 | Nasa | Preparation of polyimides from mixtures of monomeric diamines and esters of polycarboxylic acids |
| US3748312A (en) | 1972-04-03 | 1973-07-24 | Trw Inc | Thermosetting hydrazide polymers |
| US3748311A (en) | 1972-04-03 | 1973-07-24 | Trw Inc | Heterocyclic endo alicyclic end capped resins |
| US3749735A (en) | 1965-10-24 | 1973-07-31 | Eastman Kodak Co | Certificate of correction |
| US3757088A (en) | 1972-02-29 | 1973-09-04 | Hercules Inc | He same electrically conducting elastomers and electrical members embodying t |
| US3759777A (en) | 1971-01-20 | 1973-09-18 | Trw Inc | Process for producing reinforced plastic laminates from polydiene block polymers |
| US3761441A (en) | 1972-02-02 | 1973-09-25 | Union Carbide Corp | Thermosetting friction compositions and friction elements thereof containing an acrylimide resin |
| US3763101A (en) | 1972-04-03 | 1973-10-02 | Trw Inc | Thermosetting alicyclic endo end capped resins |
| US3770697A (en) | 1969-07-01 | 1973-11-06 | Gen Electric | Curable polycarbonate compositions |
| US3772250A (en) | 1970-12-08 | 1973-11-13 | Carborundum Co | Branched aromatic polyesters containing cyanuryl nucleus radicals |
| US3773718A (en) | 1971-04-30 | 1973-11-20 | Gen Electric | Method for making imido-substituted aromatic polymers |
| US3781249A (en) | 1971-12-30 | 1973-12-25 | Trw Inc | Polyamide-imides and precursors thereof |
| US3781240A (en) | 1971-12-30 | 1973-12-25 | Trw Inc | Polyimide molding powders |
| US3803081A (en) | 1967-07-03 | 1974-04-09 | Trw Inc | Polyamide molding powder prepolymers and method of preparation |
| US3812159A (en) | 1969-02-13 | 1974-05-21 | Trw Inc | Polybasic aromatic carboxylic acids,esters and anhydrides |
| US3827927A (en) | 1971-12-30 | 1974-08-06 | Trw Inc | Method of bonding using improved polyimide adhesives |
| US3839287A (en) | 1971-09-21 | 1974-10-01 | Union Carbide Corp | Polyarylimides |
| US3843593A (en) | 1972-06-05 | 1974-10-22 | Du Pont | Radar absorptive coating composition of an acrylic polymer,a polyester and an isocyanate cross-linking agent |
| US3847867A (en) | 1971-01-20 | 1974-11-12 | Gen Electric | Polyetherimides |
| US3847869A (en) | 1973-09-28 | 1974-11-12 | Gen Electric | Method for preparing polyetherimides |
| US3853815A (en) | 1970-08-17 | 1974-12-10 | Trw Inc | Thermosetting cyclized polydiene resins |
| US3859252A (en) | 1972-04-28 | 1975-01-07 | Uniroyal Inc | Polyamido polysulfone thermoplastics and intermediates thereof |
| US3879428A (en) | 1973-03-30 | 1975-04-22 | Gen Electric | Method for making aromatic bis(ether anhydride)s |
| US3879349A (en) | 1973-11-12 | 1975-04-22 | Hughes Aircraft Co | Homopolymerization of acetylene substituted polyimide polymers |
| US3879393A (en) | 1973-06-18 | 1975-04-22 | Miles Lab | Derivatives of 1,3-disubstituted 2,4(1h,3h)-quinazolinediones |
| US3887582A (en) | 1969-04-25 | 1975-06-03 | Gen Electric | Bis-imide compositions |
| US3890272A (en) | 1973-05-25 | 1975-06-17 | Univ Notre Dame Du Lac | Polymerizing bis-maleimides |
| US3895064A (en) | 1971-09-21 | 1975-07-15 | Union Carbide Corp | Polyamide-imides |
| US3896147A (en) | 1973-12-28 | 1975-07-22 | Ciba Geigy Corp | Hindered phenolic norbornane-2,3-dicarboximides |
| US3897395A (en) | 1973-05-25 | 1975-07-29 | Univ Notre Dame Du Lac | Process for chain-extending unsaturated terminated polyimides and products prepared therefrom |
| US3909507A (en) | 1973-12-06 | 1975-09-30 | Gen Electric | Electrical conductors with strippable polymeric materials |
| US3914334A (en) | 1974-07-31 | 1975-10-21 | Trw Inc | Flexible electrical insulating polymeric sheet |
| US3919177A (en) | 1973-11-19 | 1975-11-11 | Phillips Petroleum Co | P-phenylene sulfide polymers |
| US3920768A (en) | 1974-01-14 | 1975-11-18 | Union Carbide Corp | Arylimide-epoxy resin composites |
| US3925324A (en) | 1971-12-27 | 1975-12-09 | Rhone Poulenc Sa | Process for the preparation of thermosetting polymers from n,n-bis-maleimide |
| US3933862A (en) | 1974-08-23 | 1976-01-20 | General Electric Company | Aromatic dithio dianhydrides |
| US3935320A (en) | 1973-04-04 | 1976-01-27 | Union Carbide Corporation | Tubular cellulosic casing with cationic thermosetting resin coating |
| US3935167A (en) | 1974-06-06 | 1976-01-27 | University Patents, Inc. | Acetylenic polymers, trimers thereof and the production thereof |
| US3941746A (en) | 1973-12-28 | 1976-03-02 | Ciba-Geigy Corporation | Containing hindered phenolic norbornane-2,3-dicarboximides stabilized compositions |
| US3956320A (en) | 1972-08-18 | 1976-05-11 | General Electric Company | Aromatic bis(ether phthalis anhydride) compounds |
| US3957732A (en) | 1973-02-02 | 1976-05-18 | Sumitomo Chemical Company, Limited | Alternating copolymers having functional group and process for producing same |
| US3957862A (en) | 1973-03-30 | 1976-05-18 | General Electric Company | Method of making aromatic bis(ether ortho phthalic acid)s |
| US3966726A (en) | 1971-09-23 | 1976-06-29 | Akzo N.V. | Process for the production of cyclic carboxylic acid imides |
| US3966678A (en) | 1973-04-26 | 1976-06-29 | Rhone-Poulenc S.A. | Maleic anhydride-polyaldimine prepolymers |
| US3966987A (en) | 1974-04-17 | 1976-06-29 | International Flavors & Fragrances Inc. | Electroconductive high polymer composition |
| US3970714A (en) | 1971-02-24 | 1976-07-20 | Rhone-Poulenc S.A. | Heat-stable polyimide resin modified by unsaturated polyester |
| US3972902A (en) | 1971-01-20 | 1976-08-03 | General Electric Company | 4,4'-Isopropylidene-bis(3- and 4-phenyleneoxyphthalic anhydride) |
| US3988374A (en) | 1970-06-29 | 1976-10-26 | Union Carbide Corporation | Polyamide-imides |
| GB1453625A (en) | 1972-12-18 | 1976-10-27 | Toray Industries | Polyamide imide resin and prepolymer therefor |
| US3993630A (en) | 1973-12-26 | 1976-11-23 | Ciba-Geigy Corporation | Polyimide, polyamide-imide or polyamide-acids with olefin end groups in combination with a reactive Schiff base compound |
| US3998786A (en) | 1973-05-25 | 1976-12-21 | University Of Notre Dame Du Lac | Process for preparing aromatic polyimides, polyimides prepared thereby |
| US4000146A (en) | 1974-01-31 | 1976-12-28 | Horizons Incorporated, A Division Of Horizons Research Incorporated | Triamino pyridine compounds |
| US4005134A (en) | 1975-09-11 | 1977-01-25 | General Electric Company | Method for making aromatic bis(ether dicarboxylic acid) |
| FR2272119B1 (en) | 1974-05-20 | 1977-03-18 | Rhone Poulenc Ind | |
| US4020069A (en) | 1975-07-25 | 1977-04-26 | General Electric Company | Preparation of aromatic tetracarboxylic acids |
| US4026871A (en) | 1973-05-25 | 1977-05-31 | University Of Notre Dame | Chain-extending phenolic end-capped polyimides |
| USRE29316E (en) | 1967-07-13 | 1977-07-19 | Rhone-Poulenc S.A. | Cross-linked resins |
| US4038261A (en) | 1975-05-27 | 1977-07-26 | Phillips Petroleum Company | Production of branched arylene sulfide polymers |
| DE1951632C3 (en) | 1969-10-14 | 1977-08-11 | Siemens AG, 1000 Berlin und 8000 München | Thermoplastic molding compounds based on stable, finely crystalline polyolefins |
| US4051177A (en) | 1973-12-18 | 1977-09-27 | Bayer Aktiengesellschaft | Process for the preparation of unsaturated amino compounds |
| US4055543A (en) | 1973-05-25 | 1977-10-25 | Alelio Gaetano Francis D | Chain-extending Schiff base terminated polyimides and method of preparation |
| US4058505A (en) | 1973-05-25 | 1977-11-15 | University Of Notre Dame Du Lac | Chain-extending amine end-capped polyimides |
| FR2303818B1 (en) | 1975-03-10 | 1977-11-18 | Rhone Poulenc Ind | |
| US4060515A (en) | 1973-05-25 | 1977-11-29 | University Of Notre Dame Du Lac | Chain-extending nitrile end-capped polyimides |
| US4064289A (en) | 1974-08-21 | 1977-12-20 | Hitachi, Ltd. | Method of making a semiconductor device |
| US4097456A (en) | 1977-03-28 | 1978-06-27 | Gulf Research & Development Company | Preparation of acetylene-substituted polyimide oligomers and polyimide polymers |
| US4100138A (en) | 1975-05-12 | 1978-07-11 | Hughes Aircraft Company | Ethnyl terminated polyimide composites |
| US4100137A (en) | 1974-05-30 | 1978-07-11 | Polysar Limited | Crosslinking of hydroxyl-containing polymers with polyaldehyde compounds |
| US4101488A (en) | 1974-09-26 | 1978-07-18 | Nitto Electric Industrial Co., Ltd. | Water-soluble heat-resistant insulating varnish |
| US4107147A (en) | 1977-09-02 | 1978-08-15 | General Electric Company | Polysulfoneimides |
| US4107153A (en) | 1974-09-25 | 1978-08-15 | Toshiba Chemical Products Co., Ltd. | Heat-resistant thermosetting polyimide resin |
| US4107174A (en) | 1975-06-18 | 1978-08-15 | Ciba-Geigy Corporation | Imidyl compounds |
| US4108837A (en) | 1963-07-16 | 1978-08-22 | Union Carbide Corporation | Polyarylene polyethers |
| US4108926A (en) | 1976-10-28 | 1978-08-22 | The United States Of America As Represented By The Secretary Of The Air Force | Reactive plasticizer for thermoplastic polysulfone resins |
| US4111879A (en) | 1976-07-10 | 1978-09-05 | Ihara Chemical Industry Co., Ltd. | Composition for inhibiting adhesion of shellfish and algae |
| US4115362A (en) | 1975-10-03 | 1978-09-19 | Teijin Limited | Process for preparing polyesters |
| US4115231A (en) | 1975-12-19 | 1978-09-19 | Ciba-Geigy Corporation | Crosslinkable mixtures of oligomers or polymers and diamines |
| US4116937A (en) | 1976-08-25 | 1978-09-26 | Trw Inc. | Compliant maleimide based plastics |
| US4124593A (en) | 1975-11-13 | 1978-11-07 | Ciba-Geigy Corporation | Aromatic dicarboximides |
| US4126619A (en) | 1975-12-19 | 1978-11-21 | Ciba-Geigy Corporation | Crosslinkable bis-imidyl derivatives |
| US4128574A (en) | 1976-10-04 | 1978-12-05 | General Electric Company | Method for making organic polycarboxylic acids |
| US4132716A (en) | 1975-06-18 | 1979-01-02 | Ciba-Geigy Corporation | Imidyl-benzene-dicarboxylic and -tricarboxylic acid derivatives |
| US4132715A (en) | 1976-04-09 | 1979-01-02 | Ciba-Geigy Corporation | Process for the production of maleimides |
| US4134895A (en) | 1975-06-18 | 1979-01-16 | Ciba-Geigy Corporation | Imidyl-benzenedicarboxylic acid derivatives |
| US4142870A (en) | 1976-07-01 | 1979-03-06 | Abrasives International N.V. | Abrasive articles prepared from crosslinked aromatic polyimides |
| US4158731A (en) | 1975-06-18 | 1979-06-19 | Ciba-Geigy Corporation | Crosslinkable polymeric compounds |
| US4166168A (en) | 1978-07-25 | 1979-08-28 | Plastics Engineering Company | Polymerization products of acetylene-terminated polyimide derivatives |
| US4167663A (en) | 1977-01-24 | 1979-09-11 | Baxter Travenol Laboratories, Inc. | Blood warming apparatus |
| US4168366A (en) | 1978-07-25 | 1979-09-18 | Plastics Engineering Company | Polyimide derivatives |
| US4172836A (en) | 1975-06-18 | 1979-10-30 | Ciba-Geigy Corporation | Imidyl compounds |
| US4174326A (en) | 1975-06-18 | 1979-11-13 | Ciba-Geigy Corporation | Imidyl compounds |
| US4175175A (en) | 1963-07-16 | 1979-11-20 | Union Carbide Corporation | Polyarylene polyethers |
| US4176223A (en) | 1978-12-18 | 1979-11-27 | E. I. Du Pont De Nemours And Company | Polyimide-esters and filaments prepared from 4-(4'-carboxyphthalimido)benzoic acid or 4-(4'-hydroxyphthalimido)phenol |
| US4179551A (en) | 1977-10-25 | 1979-12-18 | Trw, Inc. | Low temperature curable compliant bismaleimide compositions |
| US4183839A (en) | 1976-04-08 | 1980-01-15 | John V. Long | Polyimide resin-forming composition |
| US4187364A (en) | 1975-06-18 | 1980-02-05 | Ciba-Geigy Corporation | Crosslinkable polymers of unsaturated imides |
| US4189560A (en) | 1975-06-18 | 1980-02-19 | Ciba-Geigy Corporation | Crosslinkable polymers of unsaturated imides |
| US4193927A (en) | 1975-06-18 | 1980-03-18 | Ciba-Geigy Corporation | Imidyl compounds |
| US4197397A (en) | 1975-03-14 | 1980-04-08 | University Of Notre Dame Du Lac | Chain-extending anhydride end-capped polyimides |
| US4200731A (en) | 1979-02-01 | 1980-04-29 | The Goodyear Tire & Rubber Company | Endcapped polyesters |
| US4206106A (en) | 1979-04-05 | 1980-06-03 | Gulf Oil Corporation | Vinyl end-capped polyimide oligomers |
| US4218555A (en) | 1978-11-01 | 1980-08-19 | Gulf Oil Corporation | Process for preparation of acetylene end-capped polyimide oligomers |
| US4221895A (en) | 1978-04-18 | 1980-09-09 | The Dow Chemical Company | Thermally resistant thermosetting aromatic polymers containing pendant 1-alkynyl substituents |
| US4225498A (en) | 1977-08-09 | 1980-09-30 | Rhone-Poulenc Industries | Preparation of N-substituted oligo-imides |
| US4225497A (en) | 1978-02-19 | 1980-09-30 | Rhone-Poulenc Industries | Preparation of N-substituted oligo-imides |
| US4231934A (en) | 1977-11-02 | 1980-11-04 | Mitsui Toatsu Chemicals, Incorporated | Process for the production of N-(hydroxyphenyl) maleimides |
| US4234712A (en) | 1979-09-14 | 1980-11-18 | The United States Of America As Represented By The Secretary Of The Navy | Polyphthalocyanine resins |
| US4237262A (en) | 1979-02-14 | 1980-12-02 | Trw Inc. | Curable aliphatic epoxy-polimide compositions |
| US4239883A (en) | 1975-07-09 | 1980-12-16 | Technochemie Gmbh Verfahrenstechnik | Polyimide preparation from imido acyl halide |
| US4244853A (en) | 1979-04-06 | 1981-01-13 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Composition and method for making polyimide resin-reinforced fabric |
| US4250096A (en) | 1977-10-14 | 1981-02-10 | Ciba-Geigy Corporation | 3- and 4-Azidophthalic acid derivatives |
| US4251418A (en) | 1979-04-27 | 1981-02-17 | Gulf Oil Corporation | Polyimide oligomers |
| US4251420A (en) | 1979-04-23 | 1981-02-17 | Gulf Oil Corporation | End-capped polyimide oligomers |
| US4251419A (en) | 1979-04-05 | 1981-02-17 | Gulf Oil Corporation | Novel vinyl end-capped polyimide oligomers |
| US4255313A (en) | 1979-04-20 | 1981-03-10 | Gulf Oil Corporation | Novel end-capped polyimide oligomers |
| US4266047A (en) | 1971-12-16 | 1981-05-05 | General Electric Company | Novel polymers of olefin terminated prepolymers and amine terminated prepolymers |
| US4269961A (en) | 1977-10-25 | 1981-05-26 | Trw, Inc | Low temperature curable compliant bismaleimide compositions |
| US4271079A (en) | 1978-09-07 | 1981-06-02 | Dainippon Ink & Chemicals, Inc. | 5-(2,5-Dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride |
| US4273916A (en) | 1979-02-14 | 1981-06-16 | Trw Inc. | Curable cycloaliphatic epoxy-polyimide compositions |
| US4276407A (en) | 1979-08-16 | 1981-06-30 | Hughes Aircraft Company | Acetylene terminated imide oligomers which melt at low temperatures |
| US4288607A (en) | 1975-10-25 | 1981-09-08 | Dynamit Nobel Aktiengesellschaft | Bis-carbalkoxy compounds and their preparation |
| US4288583A (en) | 1978-12-29 | 1981-09-08 | Ciba-Geigy Corporation | Curable mixtures based on maleimides and 1-propenyl-substituted phenols |
| US4293670A (en) | 1979-12-26 | 1981-10-06 | Union Carbide Corporation | Blends of poly(aryl ether) resins and polyetherimide resins |
| US4297474A (en) | 1980-02-29 | 1981-10-27 | General Electric Company | Polyetherimides prepared by self-condensation of hydroxyaryl phthalimide salts |
| US4297472A (en) | 1977-08-26 | 1981-10-27 | Mobay Chemical Corporation | Method of producing color stable thermoplastic polymethanes and products made thereby |
| US4298720A (en) | 1979-07-23 | 1981-11-03 | Mitsui Toatsu Chemicals Incorporated | Thermosetting resin composition from maleimide compound and alkenyl phenol |
| US4299946A (en) | 1977-10-14 | 1981-11-10 | Rhone-Poulenc Industries | Imido copolymers from oligoimide and phenolic compound |
| US4299750A (en) | 1979-05-03 | 1981-11-10 | Gulf Oil Corporation | Novel partially acetylene end-capped polyimide oligomers |
| US4302575A (en) | 1979-12-26 | 1981-11-24 | General Electric Company | Heat curable polyimides |
| US4323662A (en) | 1979-07-10 | 1982-04-06 | Mitsui Toatsu Chemicals, Inc. | Thermosetting resin compositions comprising bismaleimides and alkenylaniline derivatives |
| USRE30922E (en) | 1979-04-05 | 1982-05-04 | Gulf Oil Corporation | Vinyl end-capped polyimide oligomers |
| US4338225A (en) | 1980-08-04 | 1982-07-06 | The Boeing Company | High performance resin reaction products of carboxyl terminated 1,2-polybutadiene with epoxides and acid terminated difunctional aliphatic alcohols |
| US4338222A (en) | 1980-04-11 | 1982-07-06 | Xerox Corporation | Semiconductive organic compositions |
| US4344870A (en) | 1980-03-12 | 1982-08-17 | Basf Aktiengesellschaft | Preparation of electrically conductive hetero-polyaromatics |
| US4344869A (en) | 1980-03-12 | 1982-08-17 | Basf Aktiengesellschaft | Preparation of electrically conductive hetero-poly-phenylenes |
| US4351932A (en) | 1980-03-27 | 1982-09-28 | Hitco | Bis-maleimide/divinyl aryl crosslinking agent resin system |
| US4358561A (en) | 1980-01-16 | 1982-11-09 | Standard Oil Company (Indiana) | Injection moldable amide-imide copolymers |
| US4360644A (en) | 1980-01-07 | 1982-11-23 | Basf Aktiengesellschaft | Preparation of electrically conductive polyaromatics |
| US4375427A (en) | 1979-12-13 | 1983-03-01 | Allied Corporation | Thermoplastic conductive polymers |
| US4376710A (en) | 1981-03-30 | 1983-03-15 | Hughes Aircraft Company | High load carrying polyimide lubricative composites |
| US4381363A (en) | 1981-09-10 | 1983-04-26 | The United States Of America As Represented By The Secretary Of The Air Force | Cure retarding additives for acetylene-terminated polymers |
| US4389504A (en) | 1981-10-02 | 1983-06-21 | The United States Of America As Represented By The United States National Aeronautics And Space Administration Office Of General Counsel-Code Gp | Elastomer toughened polyimide adhesives |
| US4393188A (en) | 1980-02-14 | 1983-07-12 | Hitachi Chemical Company, Ltd. | Thermosetting prepolymer from polyfunctional maleimide and bis maleimide |
| US4395497A (en) | 1980-07-08 | 1983-07-26 | Basf Aktiengesellschaft | Preparation of electrically conductive soluble heteropolyphenylenes |
| US4400613A (en) | 1978-12-04 | 1983-08-23 | General Electric Company | Temperature controller for a fusing roller |
| US4405770A (en) | 1981-08-12 | 1983-09-20 | National Starch And Chemical Corporation | Novel polyimides, and polyamic acid and ester intermediates thereof |
| US4407739A (en) | 1981-02-07 | 1983-10-04 | Basf Aktiengesellschaft | Preparation of electrically conductive polymers |
| US4409382A (en) | 1982-01-07 | 1983-10-11 | The United States Of America As Represented By The Secretary Of The Navy | Synthesis and polymerization of phthalonitrile monomers containing multiple phenoxy and sulfone linkages |
| US4410686A (en) | 1981-12-21 | 1983-10-18 | The Dow Chemical Company | Norbornyl modified polyesteramides and process for preparing same |
| US4414269A (en) | 1981-06-16 | 1983-11-08 | Trw, Inc. | Solvent resistant polysulfone and polyethersulfone compositions |
| US4417039A (en) | 1982-07-19 | 1983-11-22 | The United States Of America As Represented By The Secretary Of The Air Force | Cis-enyne aromatic and aromatic heterocyclic polymers |
| US4417044A (en) | 1982-05-25 | 1983-11-22 | General Electric Company | Process for making polyetherimides |
| US4418181A (en) | 1981-05-26 | 1983-11-29 | Plastics Engineering Company | Polyimides having bis-maleimide terminal groups |
| US4423202A (en) | 1981-05-05 | 1983-12-27 | Celanese Corporation | Process for the production of high molecular weight para ordered aromatic heterocyclic polymer |
| US4429108A (en) | 1982-09-27 | 1984-01-31 | Standard Oil Company (Indiana) | Poly[N,N'-(alkyl or aryl)-bis(4,4'-phthalimido)-1,3,4-oxadiazole-2,5-diyls] |
| US4438280A (en) | 1981-05-26 | 1984-03-20 | Plastics Engineering Company | Bis-(maleamic acid) derivatives of triamines |
| US4438273A (en) | 1981-07-24 | 1984-03-20 | Hughes Aircraft Company | Isoimide containing oligomers |
| US4446191A (en) | 1980-10-27 | 1984-05-01 | Hitachi Chemical Company, Ltd. | Laminates comprising prepregs metal clad |
| US4448925A (en) | 1982-08-06 | 1984-05-15 | Standard Oil Company (Indiana) | Injection moldable polymers containing phthalic anhydride |
| US4460783A (en) | 1980-01-21 | 1984-07-17 | Hitachi, Ltd. | Ether imides and process for producing the same |
| US4465809A (en) | 1982-11-05 | 1984-08-14 | Phillips Petroleum Company | Conversion of polymer lithium to polymer carboxylate |
| US4467011A (en) | 1982-05-28 | 1984-08-21 | Standard Oil Company (Indiana) | Injection moldable amide-imide polymers and copolymers containing amorphous polyamides |
| US4476184A (en) | 1983-08-09 | 1984-10-09 | The Boeing Company | Thermally stable polysulfone compositions for composite structures |
| US4476295A (en) | 1982-09-27 | 1984-10-09 | Standard Oil Company (Indiana) | Poly-imide-oxadiazole soluble in m-cresol |
| US4482683A (en) | 1981-07-10 | 1984-11-13 | Siemens Aktiengesellschaft | Stabilized sulfur-containing resins, such as polyphenylene sulfide and method of producing the same |
| US4485140A (en) | 1983-09-21 | 1984-11-27 | E. I. Du Pont De Nemours And Company | Melt-fusible polyimides |
| US4485231A (en) | 1981-07-24 | 1984-11-27 | Hughes Aircraft Company | Isoimide containing oligomers |
| US4489027A (en) | 1982-08-11 | 1984-12-18 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Process for preparing solvent resistant, thermoplastic aromatic poly(imidesulfone) |
| US4504620A (en) | 1984-04-16 | 1985-03-12 | E. I. Du Pont De Nemours And Company | Oxazoline polyester oligomers |
| US4504632A (en) | 1983-12-30 | 1985-03-12 | General Electric Company | Amide ether imide block copolymers |
| US4507466A (en) | 1983-01-07 | 1985-03-26 | The Dow Chemical Corporation | Dense star polymers having core, core branches, terminal groups |
| US4510272A (en) | 1983-03-16 | 1985-04-09 | Avco Corporation | Bis-maleimide-epoxy compositions and prepregs |
| US4515962A (en) | 1982-08-05 | 1985-05-07 | Ciba Geigy Corporation | Allyl or methallyl-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid imides and bisimides |
| US4519926A (en) | 1983-02-18 | 1985-05-28 | Standard Oil Company (Indiana) | Polyborate esters and their use in lubricants |
| US4520198A (en) | 1982-03-02 | 1985-05-28 | Plastics Engineering Company | Vinylacetylene-terminated polyimide derivatives |
| US4526838A (en) | 1982-09-08 | 1985-07-02 | Hitachi Chemical Company, Ltd. | Polyamino-bis-imide resin |
| US4533692A (en) | 1982-09-17 | 1985-08-06 | Sri International | Liquid crystalline polymer compositions, process, and products |
| US4533724A (en) | 1982-09-17 | 1985-08-06 | Sri International | Liquid crystalline poly (2,6-benzothiazole) compositions, process, and products |
| US4533693A (en) | 1982-09-17 | 1985-08-06 | Sri International | Liquid crystalline polymer compositions, process, and products |
| US4535117A (en) | 1983-11-02 | 1985-08-13 | Phillips Petroleum Company | Stabilization of polyphenylene sulfide |
| US4536559A (en) | 1983-06-17 | 1985-08-20 | The Boeing Company | Thermally stable polyimide polysulfone compositions for composite structures |
| US4547553A (en) | 1982-07-29 | 1985-10-15 | The Boeing Company | Polybutadiene modified polyester compositions |
| US4555563A (en) | 1984-12-07 | 1985-11-26 | The Dow Chemical Company | Imide functional polyphenols; thermosettable compositions containing same and cured products therefrom |
| US4556705A (en) | 1984-10-26 | 1985-12-03 | General Electric Company | Thermoplastic polyetherimide ester elastomers |
| US4556697A (en) | 1983-07-25 | 1985-12-03 | The Standard Oil Company | Alternating copolyamide prepared on a polymer matrix |
| US4558120A (en) | 1983-01-07 | 1985-12-10 | The Dow Chemical Company | Dense star polymer |
| US4562231A (en) | 1984-09-06 | 1985-12-31 | Atlantic Richfield Company | Polyether sulfone resin containing molding composition and method |
| US4562232A (en) | 1984-12-24 | 1985-12-31 | General Electric Company | Copolyetherester-dimer ester-block copolymers |
| US4563514A (en) | 1985-03-26 | 1986-01-07 | General Electric Company | Curable polysilarylene-polysiloxane copolymers |
| US4563498A (en) | 1984-07-05 | 1986-01-07 | General Electric Company | Curable silicone compositions and processes |
| US4564553A (en) | 1982-08-09 | 1986-01-14 | Merak S.P.A. | Manufactured articles, including fabrics comprising thermally bonded polyolefinic fibers |
| US4567216A (en) | 1983-12-22 | 1986-01-28 | Union Carbide Corporation | Thermoplastic modified epoxy compositions |
| US4567240A (en) | 1984-05-23 | 1986-01-28 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Ethynyl terminated ester oligomers and polymers therefrom |
| US4568737A (en) | 1983-01-07 | 1986-02-04 | The Dow Chemical Company | Dense star polymers and dendrimers |
| US4574148A (en) | 1985-06-21 | 1986-03-04 | Eastman Kodak Company | Polyester resins capable of forming containers having improved gas barrier properties |
| US4574144A (en) | 1984-12-17 | 1986-03-04 | General Electric Company | Sulfonated polyimides and polyamic acids and method for their preparation |
| US4574154A (en) | 1982-12-28 | 1986-03-04 | Ube Industries, Ltd. | Triazine addition compounds |
| US4576857A (en) | 1983-03-14 | 1986-03-18 | E. I. Du Pont De Nemours And Company | Melt-fusible polyimides |
| US4577034A (en) | 1985-02-14 | 1986-03-18 | The Dow Chemical Company | Autoxidation of bis(ortho dialkyl-phenoxy)benzophenones |
| US4578433A (en) | 1983-12-24 | 1986-03-25 | Basf Aktiengesellschaft | Long-term stability of the electrical conductivity of pyrrole polymers |
| US4578470A (en) | 1983-06-20 | 1986-03-25 | General Electric Company | Bis-imides containing heterocyclic aromatic rings |
| EP0175484A2 (en) | 1984-08-20 | 1986-03-26 | Amoco Corporation | Sized fibres |
| US4584364A (en) | 1984-02-06 | 1986-04-22 | The Boeing Company | Phenolic-capped imide sulfone resins |
| US4587329A (en) | 1984-08-17 | 1986-05-06 | The Dow Chemical Company | Dense star polymers having two dimensional molecular diameter |
| US4590363A (en) | 1982-07-28 | 1986-05-20 | Ersa Ernst Sachs Kg Gmbh & Co. | Circuit for controlling temperature of electric soldering tool |
| US4599383A (en) | 1984-04-28 | 1986-07-08 | Ntn-Rulon Industries Co., Ltd. | Polyamideimide resin composition |
| US4600769A (en) | 1983-12-15 | 1986-07-15 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Amine terminated bisaspartimide polymer |
| US4604437A (en) | 1984-01-31 | 1986-08-05 | Ciba-Geigy Corporation | Polymer from substituted, unsaturated, bicyclic imide |
| US4608426A (en) | 1985-01-29 | 1986-08-26 | American Cyanamid Company | Bis-maleimide resin composition |
| US4608414A (en) | 1983-07-19 | 1986-08-26 | Denki Kagaku Kogyo Kabushiki Kaisha | Thermoplastic resin composition containing an imide polymer and graft copolymer |
| US4611022A (en) | 1985-06-24 | 1986-09-09 | The Dow Chemical Company | Polymer modified polyphenol compositions and thermosettable resins thereof |
| US4611048A (en) | 1985-10-04 | 1986-09-09 | General Electric Company | Hydroxy terminated polyetherimide oligomers |
| US4614767A (en) | 1985-07-29 | 1986-09-30 | Atlantic Richfield Company | Moldable polyester-sulfone thermoplastic resin |
| US4615832A (en) | 1983-09-26 | 1986-10-07 | Bayer Aktiengesellschaft | Flameproofing agents, their preparation and their use for flameproofing polycarbonates |
| US4616070A (en) | 1984-05-08 | 1986-10-07 | Basf Aktiengesellschaft | Thermoplastic molding materials |
| US4616071A (en) | 1983-01-06 | 1986-10-07 | Ford Motor Company | Coating composition comprising bis-diene oligomers and bis-dieneophile oligomers |
| US4617390A (en) | 1982-02-23 | 1986-10-14 | Basf Aktiengesellschaft | S-triazine derivatives |
| US4624888A (en) | 1985-09-30 | 1986-11-25 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Acetylene (ethynyl) terminated polyimide siloxane and process for preparation thereof |
| US4628079A (en) | 1983-09-06 | 1986-12-09 | Bayer Aktiengesellschaft | Polyamide imides produced from either lactams or polyamides with isocyanates or anhydrides in which products are precipitated with a non-solvent |
| US4628067A (en) | 1984-01-16 | 1986-12-09 | Celanese Corporation | Microporous polybenzimidazole particulates |
| US4629777A (en) | 1983-05-18 | 1986-12-16 | Ciba-Geigy Corporation | Polyimides, a process for their preparation and their use |
| US4631337A (en) | 1983-01-07 | 1986-12-23 | The Dow Chemical Company | Hydrolytically-stable dense star polyamine |
| USH183H (en) | 1985-08-19 | 1987-01-06 | The United States Of America As Represented By The Secretary Of The Air Force | Conducting poly (p-xylylidenes) |
| US4638027A (en) | 1982-12-27 | 1987-01-20 | General Electric Company | Polycarbonate exhibiting improved heat resistance from cycloalkylidene diphenol |
| US4640944A (en) | 1984-01-31 | 1987-02-03 | Standard Oil Company (Indiana) | Injection moldable polyamide-imide-phthalamide copolymers containing polyetherimides |
| US4649080A (en) | 1984-05-08 | 1987-03-10 | Basf Aktiengesellschaft | Fiber-reinforced materials |
| US4654410A (en) | 1985-01-22 | 1987-03-31 | Asahi Glass Company, Ltd. | Aromatic polysulfone ether/polythioether sulfone copolymer and process for its preparation |
| US4657973A (en) | 1985-01-23 | 1987-04-14 | Idemitsu Petrochemical Co., Ltd. | Polycarbonate resin composition |
| US4657990A (en) | 1984-01-19 | 1987-04-14 | Imperial Chemical Industries Plc | Aromatic polyetherketones |
| US4657977A (en) | 1985-10-04 | 1987-04-14 | General Electric Company | Poly(etherimide-carbonate) block copolymers and polymer blends containing same |
| US4657987A (en) | 1984-01-27 | 1987-04-14 | General Electric Company | Polyetherimide-polyamide blends |
| US4660057A (en) | 1983-01-21 | 1987-04-21 | Konishiroku Photo Industry Co., Ltd. | Heater power controller for ink jet printer |
| US4661604A (en) | 1981-06-16 | 1987-04-28 | Trw, Inc. | Monofunctional crosslinking imidophenols |
| US4663399A (en) | 1985-08-22 | 1987-05-05 | General Electric Company | Polycarbonate-polyether block copolymers, polymer blends containing same and intermediates for the production thereof |
| US4663425A (en) | 1985-12-10 | 1987-05-05 | The United States Of America As Represented By The Secretary Of The Air Force | Ethynyl-containing aromatic polyamide resin |
| US4663423A (en) | 1984-04-16 | 1987-05-05 | Teijin Limited | Polyester and process for preparation thereof |
| US4663424A (en) | 1984-11-27 | 1987-05-05 | Bayer Aktiengesellschaft | Aromatic polyester with dimethylmaleimide end groups |
| US4663378A (en) | 1983-12-29 | 1987-05-05 | General Electric Company | Silicone-modified polyethermides |
| US4680326A (en) | 1985-04-16 | 1987-07-14 | Phillips Petroleum Company | Poly(arylene sulfide) composition with improved insulation resistance and cracking resistance |
| US4680377A (en) | 1985-01-15 | 1987-07-14 | Matsushita Electric Works, Ltd. | Polyimide prepolymer composition from unsaturated bisimide and diamine containing unreacted reactants |
| US4684714A (en) | 1983-09-27 | 1987-08-04 | The Boeing Company | Method for making polyimide oligomers |
| US4690972A (en) | 1986-04-04 | 1987-09-01 | Phillips Petroleum Company | Method of producing poly(arylene sulfide) compositions and articles made therefrom |
| US4691025A (en) | 1983-12-22 | 1987-09-01 | Amoco Corporation | Bismaleimides and prepreg resins therefrom |
| US4694064A (en) | 1986-02-28 | 1987-09-15 | The Dow Chemical Company | Rod-shaped dendrimer |
| US4695610A (en) | 1986-03-18 | 1987-09-22 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Semi-2-interpenetrating networks of high temperature systems |
| US4699975A (en) | 1985-03-07 | 1987-10-13 | Kureha Kagaku Kogyo Kabushiki Kaisha | Process for producing readily crystallizable arylenesulfide resin composition containing organic acid or anhydride |
| US4703081A (en) | 1986-04-08 | 1987-10-27 | Phillips Petroleum Company | Poly(arylene sulfide) resin ternary blends |
| US4709008A (en) | 1986-06-30 | 1987-11-24 | Interez, Inc. | Blend of tris (cyanatophenyl) alkane and bis(cyanatophenyl) alkane |
| US4708983A (en) | 1986-06-18 | 1987-11-24 | Phillips Petroleum Company | Arylene sulfide polymers of improved impact strength |
| US4709004A (en) | 1984-11-05 | 1987-11-24 | The Dow Chemical Company | Polyesteramide from hydroxy phenyl-isopropyl-amino diphenyl ether |
| US4709006A (en) | 1986-04-16 | 1987-11-24 | The United States Of America As Represented By The Secretary Of The Air Force | Polybenzobisthiazole from aryloxy naphthalic diacid |
| US4714768A (en) | 1983-08-25 | 1987-12-22 | Akademie Der Wissenschaften Der Ddr | N-(chlorocarbonyloxy)-5-norbornene-2,3-dicarboximide, process for its production and its use |
| US4716212A (en) | 1986-09-05 | 1987-12-29 | Phillips Petroleum Company | Preparation of a high molecular weight poly(arylene sulfide ketone) |
| US4719283A (en) | 1985-03-06 | 1988-01-12 | Huels Aktiengesellschaft | Preparation of polyimide from bis-imide and benzocyclobutene |
| US4727118A (en) | 1985-04-12 | 1988-02-23 | Ntn-Rulon Industries Co., Ltd. | Polyether imide resin composition containing vulcanizable organopolysiloxane |
| US4728742A (en) | 1984-06-27 | 1988-03-01 | Ciba-Geigy Corporation | Substituted, unsaturated, bicyclic imides containing hydroxyl groups, and polymers thereof |
| US4730030A (en) | 1986-07-25 | 1988-03-08 | The Dow Chemical Company | Arylcyclobutane/dienophile copolymer |
| US4737550A (en) | 1983-01-07 | 1988-04-12 | The Dow Chemical Company | Bridged dense star polymers |
| US4739115A (en) | 1980-09-25 | 1988-04-19 | Mcdonnell Douglas Corporation | Poly phosphinoguanidine compounds |
| US4739030A (en) | 1983-06-17 | 1988-04-19 | The Boeing Company | Difunctional end-cap monomers |
| US4739075A (en) | 1985-06-01 | 1988-04-19 | Director-General Of Agency Of Industrial Science & Technology | Reactive end-capping agent, addition-curable quinoxaline resin oligomer and intermediate material for fiber-reinforced composite |
| US4740563A (en) | 1986-12-02 | 1988-04-26 | General Electric Company | Crosslinked polyetherimide ester polymers |
| US4740584A (en) | 1986-09-08 | 1988-04-26 | Interez, Inc. | Blend of dicyanate esters of dihydric phenols |
| US4740564A (en) | 1986-12-02 | 1988-04-26 | General Electric Company | Thermoplastic polyetherimide esters |
| US4748227A (en) | 1986-04-25 | 1988-05-31 | Amoco Corporation | Process for preparing poly(aryl ether)ketones using a sodium carbonate/organic acid salt catalyst |
| US4755584A (en) | 1985-09-02 | 1988-07-05 | Idemitsu Kosan Company Limited | Polyester carbonate containing imido groups and method of preparing the same |
| US4755585A (en) | 1982-08-06 | 1988-07-05 | Amoco Corporation | Injection moldable polyamide imide from trimellitic anhydride end capped with aniline |
| US4757118A (en) | 1986-01-23 | 1988-07-12 | Allied Corporation | Thermosettable modified phenolic polyester imide resins |
| US4757150A (en) | 1986-07-31 | 1988-07-12 | General Electric Company | Polyetherimide bisphenol compositions |
| US4757128A (en) | 1986-08-01 | 1988-07-12 | Massachusetts Institute Of Technology | High molecular weight polyanhydride and preparation thereof |
| US4759986A (en) | 1986-10-23 | 1988-07-26 | Hoechst Celanese Corporation | Electrically conductive polybenzimidazole fibrous material |
| US4760106A (en) | 1983-05-20 | 1988-07-26 | Amoco Corporation | Impact resistant matrix resins for advanced composites |
| US4764427A (en) | 1985-08-22 | 1988-08-16 | Teijin Limited | Fiber having thermoplastic resin coating |
| US4766180A (en) | 1985-12-23 | 1988-08-23 | Shell Oil Company | Crosslinked elastomers containing benzocyclobutene derivatives as crosslinking agents |
| US4766197A (en) | 1986-10-28 | 1988-08-23 | Amoco Corporation | Modified poly(aryl ether ketones) derived from biphenol |
| US4769424A (en) | 1987-06-01 | 1988-09-06 | General Electric Company | Polyarylene sulfide block copolymers, method for making, and use |
| US4769426A (en) | 1985-08-27 | 1988-09-06 | Kureha Kagaku Kogyo Kabushiki Kaisha | Process for preparation of polyarylene thioethers having high melt crystallization temperature |
| US4769436A (en) | 1986-12-20 | 1988-09-06 | Bayer Aktiengesellschaft | Polyester polyalcohols containing amino and amide groups and poly (urea) urethane produced therefrom |
| US4774282A (en) | 1986-11-20 | 1988-09-27 | Amoco Corporation | Thermoset composition comprising aromatic cyanate ester, allyl ethers of bisphenol and bismaleimide |
| EP0283636A2 (en) | 1987-02-20 | 1988-09-28 | The Boeing Company | Polyetherimide oligomers and blends |
| US4777208A (en) | 1986-01-27 | 1988-10-11 | The Dow Chemical Company | Polymer modified unsaturated polyester for polyesteramide resin polymer concrete |
| US4778830A (en) | 1986-12-13 | 1988-10-18 | Basf Aktiengesellschaft | Polyester polyols liquid at room temperature and their preparation and use in preparing plastics by polyisocyanate addition polymerization |
| US4778898A (en) | 1985-10-25 | 1988-10-18 | Ciba-Geigy Corporation | Trisimides of allyl-substituted or methallyl-substituted bicyclo[2.2.1]hept-5-ene-2,3-dicarboximides and the use thereof |
| US4778859A (en) | 1985-04-27 | 1988-10-18 | Asahi Kasei Kogyo Kabushiki Kaisha | Tetramine derived polyimide with pendant unsaturation, and various photosensitive compositions therefrom |
| US4786713A (en) | 1987-11-06 | 1988-11-22 | Eastman Kodak Company | Copoly(arylene sulfidex-disulfide) |
| US4786685A (en) | 1986-08-01 | 1988-11-22 | Nippon Gohsei Kagaku Kogyo Kabushiki Kaisha | Thermoplastic resin composition |
| US4786669A (en) | 1986-06-09 | 1988-11-22 | Shell Oil Company | Lightly cross-linked resin materials |
| US4798882A (en) | 1986-07-23 | 1989-01-17 | Enichem Tecnoresine S.P.A. | Branched polycarbonate from methylene-bis-naphthalene compound |
| US4798685A (en) | 1986-10-20 | 1989-01-17 | Lockheed Corporation | Production of base-type conducting polymers |
| US4798686A (en) | 1985-11-29 | 1989-01-17 | Bayer Aktiengesellschaft | Organic polymers with electrical properties |
| US4801676A (en) | 1986-08-28 | 1989-01-31 | Basf Aktiengesellschaft | Fully aromatic thermotropic polyester imide |
| US4801677A (en) | 1985-01-25 | 1989-01-31 | Bayer Aktiengesellschaft | Thermotropic aromatic polyester with high thermal stability |
| US4804724A (en) | 1984-09-28 | 1989-02-14 | Amoco Corporation | Blends of a biphenyl containing poly (aryl ether sulfone) and a poly (aryl ether ketone) |
| US4804722A (en) | 1984-12-05 | 1989-02-14 | Hoechst Ag | Resin solutions for cements and coating compositions, process for their preparation and their use |
| US4806407A (en) | 1987-05-19 | 1989-02-21 | Shakespeare Company | Monofilaments, fabrics thereof and related process |
| US4808717A (en) | 1986-09-17 | 1989-02-28 | Sumitomo Chemical Company, Limited | Thermosetting resin composition |
| US4812552A (en) | 1987-04-15 | 1989-03-14 | Phillips Petroleum Company | Process for preparing poly(arylene sulfide ketone) with water addition |
| US4812518A (en) | 1985-10-03 | 1989-03-14 | Byk-Chemie Gmbh | Polyester group containing polysiloxanes for lacquers and molding compositions |
| US4812588A (en) | 1987-12-14 | 1989-03-14 | The Dow Chemical Company | Polyorganosiloxane-bridged bisbenzocyclobutene monomers |
| US4812534A (en) | 1988-04-04 | 1989-03-14 | Eastman Kodak Company | Impact toughened polyester nylon blends |
| US4814421A (en) | 1987-12-04 | 1989-03-21 | General Electric Company | Hydroxy terminated polycarbonates |
| US4814472A (en) | 1981-11-06 | 1989-03-21 | Hughes Aircraft Company | Diethynylated diphenyl hexafluoropropanes |
| US4814417A (en) | 1987-04-10 | 1989-03-21 | Chisso Corporation | Process for producing a liquid crystalline polyester polymer of a cyclohexanedicarboxylic acid and an aromatic diol with pyridine/thionyl chloride catalyst |
| US4814416A (en) | 1987-09-04 | 1989-03-21 | Huels Aktiengesellschaft | Molding compounds comprised of a thermotropic aromatic polyester |
| US4816503A (en) | 1986-06-04 | 1989-03-28 | The Dow Chemical Company | Polymer concrete having high bond strength and long working time |
| US4816556A (en) | 1985-02-22 | 1989-03-28 | E. I. Du Pont De Nemours And Company | Ordered polyetherketones |
| US4816526A (en) | 1986-12-08 | 1989-03-28 | Ici Americas Inc. | Polyimide/polyetherimide resin blends |
| US4816527A (en) | 1987-08-20 | 1989-03-28 | General Electric Company | Polycarbonate-siloxane polyetherimide copolymer blends |
| EP0309649A2 (en) | 1987-11-03 | 1989-04-05 | The Boeing Company | High performance heterocycle oligomers and blends |
| US4820770A (en) | 1988-01-25 | 1989-04-11 | Ethyl Corporation | Polyamide composition |
| US4826927A (en) | 1986-01-13 | 1989-05-02 | Ciba-Geigy Corporation | Mixtures containing polymides alkenyphenols and either epoxide group-free adducts or another polyimide |
| US4827000A (en) | 1987-09-21 | 1989-05-02 | Occidental Chemical Corporation | Process for the preparation of phenoxy phthalic anhydrides |
| US4826997A (en) | 1984-08-27 | 1989-05-02 | The Dow Chemical Company | N-Substituted arylcyclo butenyl-maleimides |
| US4829138A (en) | 1987-02-24 | 1989-05-09 | Rhone-Poulenc Chimie | Novel heat-stable siloxane/bismaleimido polymers |
| US4835197A (en) | 1985-01-04 | 1989-05-30 | Raychem Corporation | Aromatic polymer compositions |
| US4837256A (en) | 1983-06-13 | 1989-06-06 | Amoco Corporation | Crosslinkable, crystallizable polyarylethers and composites therefrom |
| US4839378A (en) | 1986-04-30 | 1989-06-13 | Meiji Seika Kabushiki Kaisha | Fluorophthalimides and agricultural and horticultural fungicides containing them |
| US4845150A (en) | 1985-09-26 | 1989-07-04 | Foster-Miller Inc. | Interpenetrated polymer films |
| US4845185A (en) | 1986-04-22 | 1989-07-04 | Nippon Steel Corporation | Soluble copolyimide from 9,9-bis (4-amino phenyl) fluorene |
| US4845167A (en) | 1986-10-29 | 1989-07-04 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Condensation polyimides containing 1,1,1-triaryl-2,2,2-trifluoroethane structures |
| US4845278A (en) | 1987-09-03 | 1989-07-04 | Semih Erhan | Crosslinkable aramids |
| US4847333A (en) | 1985-09-30 | 1989-07-11 | The Boeing Company | Blended polyamide oligomers |
| EP0323540A1 (en) | 1988-01-08 | 1989-07-12 | The Boeing Company | Conductive, thermally stable oligomers |
| US4851496A (en) | 1987-12-12 | 1989-07-25 | Huels Aktiengesellschaft | Molding material comprising a thermotropic, aromatic polyester from bis(carboxy phenoxy)diphenyl sulfone |
| US4851287A (en) | 1985-03-11 | 1989-07-25 | Hartsing Jr Tyler F | Laminate comprising three sheets of a thermoplastic resin |
| US4851501A (en) | 1983-06-17 | 1989-07-25 | The Boeing Company | Polyethersulfone prepregs, composites, and blends |
| US4851494A (en) | 1987-07-24 | 1989-07-25 | Ciba-Geigy Corporation | Crosslinkable polyether-amide |
| US4851505A (en) | 1988-04-13 | 1989-07-25 | E. I. Du Pont De Nemours And Company | Highly soluble aromatic polyimides |
| US4851280A (en) | 1988-04-01 | 1989-07-25 | E. I. Du Pont De Nemours And Company | Composite tooling for composites manufacture |
| US4861855A (en) | 1987-03-31 | 1989-08-29 | Amoco Corporation | Polyamide-imide compositions from bisphenoxyphenyl diamine tricarboxylic anhydride and tetracarboxylic dianhydride |
| US4861915A (en) | 1985-05-02 | 1989-08-29 | Amoco Corporation | Poly (aryl ether ketone) block copolymers |
| US4861924A (en) | 1988-08-25 | 1989-08-29 | Jet Research Center, Inc. | 1,3,5-trinitro-2,4,6-tripicrylbenzene |
| US4861882A (en) | 1988-07-14 | 1989-08-29 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Ethynyl terminated imidothioethers and resins therefrom |
| US4868270A (en) | 1986-08-04 | 1989-09-19 | The Boeing Company | Heterocycle sulfone oligomers and blends |
| US4871475A (en) | 1985-10-07 | 1989-10-03 | The Boeing Company | Polysulfone and polyethersulfone oligomers |
| US4874834A (en) | 1985-03-10 | 1989-10-17 | Nitto Electric Industrial Co., Ltd. | Colorless transparent polyimide shaped article and process for producing the same |
| US4876328A (en) | 1987-05-18 | 1989-10-24 | The Boeing Company | Polyamide composition |
| US4876330A (en) | 1985-03-10 | 1989-10-24 | Nitto Electric Industrial Co., Ltd. | Colorless transparent polyimide shaped article and process for producing the same |
| US4876325A (en) | 1988-04-28 | 1989-10-24 | Allied-Signal Inc. | Polyimide resin from bis-imide, polyphenol and dicyandiamide |
| US4891408A (en) | 1986-03-03 | 1990-01-02 | Amoco Corporation | Epoxy resins based on tetraglycidyl diamines |
| US4891167A (en) | 1985-05-02 | 1990-01-02 | Amoco Corporation | Block polymers containing a poly(aryl ether ketone) and methods for their production |
| US4891460A (en) | 1984-03-09 | 1990-01-02 | San-Petrochemicals Co., Ltd. | Process for producing alkylidenenorbornenes |
| US4895892A (en) | 1987-05-15 | 1990-01-23 | Kureha Kagaku Kogyo K.K. | Melt-stable poly(arylene thioether-ketone) compositions |
| US4895924A (en) | 1987-05-15 | 1990-01-23 | Kureha Kagaku Kogyo K. K. | Poly(arylene thioether-ketone) fibers and production process thereof |
| US4897527A (en) | 1985-05-09 | 1990-01-30 | John Corby Limited | Press for fabrics |
| US4902440A (en) | 1986-07-21 | 1990-02-20 | The Yokohama Rubber Co., Ltd. | UV-curable resin compositions |
| US4902335A (en) | 1987-06-22 | 1990-02-20 | Nihon Tokushu Noyaku Seizo K.K. | Benzo-fused cyclic compounds |
| US4902769A (en) | 1988-09-23 | 1990-02-20 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Low dielectric fluorinated poly(phenylene ether ketone) film and coating |
| US4902773A (en) | 1986-12-03 | 1990-02-20 | Nitrokemia Ipartelepek | Method of preparation of unsaturated polyester resins from maleic acid/anhydride |
| US4916235A (en) | 1986-11-26 | 1990-04-10 | University Of Dayton | Resin systems derived from benzocyclobutene-maleimide compounds |
| US4916210A (en) | 1988-10-20 | 1990-04-10 | Shell Oil Company | Resin from alpha, alpha', alpha"-tris(4-cyanatophenyl)-1,3,5-triisopropylbenzene |
| US4919992A (en) | 1987-07-02 | 1990-04-24 | Imperial Chemical Industries Plc | Process for making microporous products and the products thereof |
| US4923752A (en) | 1988-01-19 | 1990-05-08 | E. I. Du Pont De Nemours & Co. | Sizing for carbon fiber |
| EP0317754A3 (en) | 1987-05-04 | 1990-05-16 | The Boeing Company | Polymide oligomers and blends |
| US4927899A (en) | 1988-03-24 | 1990-05-22 | Rhone-Poulenc Chimie | Alkenyloxylated aromatic polyamidoimides |
| US4927900A (en) | 1988-03-24 | 1990-05-22 | Rhone-Poulenc Chimie | Aromatic polyamidoimides having maleimido end groups |
| US4931540A (en) | 1987-11-24 | 1990-06-05 | Hoechst Celanese Corporation | Polymers prepared from 4,4'-bis[2-(3,4-(dicarboxyphenyl)hexafluoroisopropyl] diphenyl ether dianhydride |
| US4931531A (en) | 1987-07-02 | 1990-06-05 | Mitsui Toatsu Chemicals, Incorporated | Polyimide and high-temperature adhesive thereof |
| US4935523A (en) | 1985-09-30 | 1990-06-19 | The Boeing Company | Crosslinking imidophenylamines |
| EP0305882A3 (en) | 1987-09-03 | 1990-07-11 | The Boeing Company | Amideimide oligomers & blends |
| US4958031A (en) | 1987-02-20 | 1990-09-18 | The Boeing Company | Crosslinking nitromonomers |
| US4973662A (en) | 1987-01-13 | 1990-11-27 | Director General Of Agency Of Industrial Science And Technology | Endcapping agent for polyimide, polyimide varnish, and intermediate material for fiber-reinforced composite material comprising same |
| US4980481A (en) | 1983-06-17 | 1990-12-25 | The Boeing Company | End-cap monomers and oligomers |
| US4981922A (en) | 1987-02-20 | 1991-01-01 | The Boeing Company | Blended etherimide oligomers |
| US4985568A (en) | 1985-09-30 | 1991-01-15 | The Boeing Company | Method of making crosslinking imidophenylamines |
| US4990624A (en) | 1987-02-20 | 1991-02-05 | The Boeing Company | Intermediates anhydrides useful for synthesizing etherimides |
| US4996101A (en) | 1988-08-08 | 1991-02-26 | Lockheed Corporation | Processible polyimide blends |
| US5003035A (en) | 1989-07-13 | 1991-03-26 | The United States Of America As Represented By The Secretary Of The Air Force | Crosslinkable rigid-rod benzobisazole copolymer |
| EP0418406A1 (en) | 1989-09-19 | 1991-03-27 | The Boeing Company | Coreactive oligomer blends |
| US5011905A (en) | 1987-05-04 | 1991-04-30 | The Boeing Company | Polyimide oligomers and blends |
| EP0277476A3 (en) | 1987-01-05 | 1991-09-25 | The Boeing Company | Crosslinkable oligomers |
| US5066541A (en) | 1984-09-18 | 1991-11-19 | The Boeing Company | Heterocycle oligomer blends |
| US5066776A (en) | 1989-08-24 | 1991-11-19 | Bayer Aktiengesellschaft | Process for the manufacture of polyarylene sulphides |
| US5075537A (en) | 1988-06-03 | 1991-12-24 | Warme-Undeletrotechn | Controlling and monitoring circuit for electrical seat heating means, especially of automotive vehicles |
| EP0405128A3 (en) | 1989-05-18 | 1992-01-08 | The Boeing Company | Polyimide oligomers and blends and method of curing |
| US5082905A (en) | 1984-09-18 | 1992-01-21 | The Boeing Company | Blended heterocycles |
| US5086154A (en) | 1989-07-11 | 1992-02-04 | Rhone-Poulenc Chimie | Linear aromatic poly(amideimide)s having latent maleimide endgroups |
| US5087701A (en) | 1985-09-30 | 1992-02-11 | The Boeing Company | Phthalimide acid halides |
| US5109105A (en) | 1987-06-12 | 1992-04-28 | The Boeing Company | Polyamide oligomers |
| US5111026A (en) | 1989-04-01 | 1992-05-05 | Ma Chun Ho | Control device and method for an electric rice cooker |
| US5112939A (en) | 1983-06-17 | 1992-05-12 | The Boeing Company | Oligomers having pyrimidinyl end caps |
| US5112936A (en) | 1988-09-06 | 1992-05-12 | Idemitsu Petrochemical Co., Ltd. | Branched chain stopped polycarbonate and process for production |
| US5115087A (en) | 1988-03-29 | 1992-05-19 | The Boeing Company | Coreactive imido oligomer blends |
| US5120819A (en) | 1984-09-18 | 1992-06-09 | Boeing Company | High performance heterocycles |
| EP0266662B1 (en) | 1986-11-06 | 1992-06-10 | Bayer Ag | Process for the preparation of poly(arylensulphides) with lower crystallization temperature |
| US5126410A (en) | 1984-09-18 | 1992-06-30 | The Boeing Company | Heterocycle oligomers |
| EP0292434B1 (en) | 1987-05-22 | 1992-07-08 | Ciba-Geigy Ag | Unsaturated bisimides and their polymers |
| US5144000A (en) | 1987-02-20 | 1992-09-01 | The Boeing Company | Method for forming crosslinking polyetherimide oligomers |
| US5151487A (en) | 1985-09-30 | 1992-09-29 | The Boeing Company | Method of preparing a crosslinking oligomer |
| US5155206A (en) | 1987-09-03 | 1992-10-13 | The Boeing Company | Crosslinkable polyamideimide oligomers and a method of preparation |
| US5159055A (en) | 1983-06-17 | 1992-10-27 | The Boeing Company | Coreactive oligomer blends |
| US5175233A (en) | 1983-06-17 | 1992-12-29 | The Boeing Company | Multidimensional ester or ether oligomers with pyrimidinyl end caps |
| US5175234A (en) | 1983-09-27 | 1992-12-29 | The Boeing Company | Lightly crosslinked polyimides |
| US5175304A (en) | 1987-02-20 | 1992-12-29 | The Boeing Company | Halo- or nitro-intermediates useful for synthesizing etherimides |
| US5198526A (en) | 1984-09-18 | 1993-03-30 | The Boeing Company | Heterocycle oligomers with multidimensional morphology |
| US5216117A (en) | 1987-09-03 | 1993-06-01 | The Boeing Company | Amideimide blends |
| US5227461A (en) | 1983-06-17 | 1993-07-13 | The Boeing Company | Extended difunctional end-cap monomers |
| US5230956A (en) | 1982-05-28 | 1993-07-27 | Amoco Corporation | Polyamide-imide sized fibers |
| US5254605A (en) | 1991-11-30 | 1993-10-19 | Cheil Industries, Inc. | Imide epoxy resin composition for sealing semiconductor elements |
| US5268519A (en) | 1987-02-20 | 1993-12-07 | The Boeing Company | Lightly crosslinked etherimide oligomers |
| US5286811A (en) | 1983-09-27 | 1994-02-15 | The Boeing Company | Blended polyimide oligomers and method of curing polyimides |
| US5338532A (en) | 1986-08-18 | 1994-08-16 | The Dow Chemical Company | Starburst conjugates |
| US5344894A (en) | 1983-09-27 | 1994-09-06 | The Boeing Company | Polyimide oligomers and blends |
| US5594089A (en) * | 1985-09-05 | 1997-01-14 | The Boeing Company | Heterocycle or heterocycle sulfone oligomers with multiple chemically functional end caps |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3267087A (en) * | 1966-08-16 | Polymerization catalyst | ||
| US29316A (en) * | 1860-07-24 | Water-metek | ||
| US30922A (en) * | 1860-12-18 | Improvement in platform-scales | ||
| DE1951632U (en) | 1966-06-14 | 1966-12-15 | Friedrich Schlapp | DETACHABLE CONNECTING FITTING. |
| US3536670A (en) * | 1969-05-26 | 1970-10-27 | Gen Mills Inc | Polycarbonates from bis-hydroxy(-alkyl and -aryl)pyromellitic diimides |
| JPS5859219A (en) * | 1981-10-05 | 1983-04-08 | Mitsui Toatsu Chem Inc | Production of aromatic polyester |
| US5506060A (en) | 1981-11-13 | 1996-04-09 | The Boeing Company | Method for making multidimensional ether or ester oligomers |
| US5550204A (en) | 1981-11-13 | 1996-08-27 | The Boeing Company | Ether and ester oligomers with multidimensional morphology |
| US5071941A (en) | 1983-06-17 | 1991-12-10 | The Boeing Company | Multidimensional ether sulfone oligomers |
| US4557034A (en) | 1983-10-25 | 1985-12-10 | General Machine Products Company, Inc. | Tool for mounting connectors to multi-conductor cords or wires |
| MC1581A1 (en) | 1983-12-02 | 1985-02-04 | Toutelectric | METHOD FOR CONTROLLING THE WELDING TIME |
| US4597553A (en) | 1985-01-25 | 1986-07-01 | Rorabaugh Barre L | Track-mounted support |
| US4907739A (en) | 1986-04-22 | 1990-03-13 | Gyp-Crete Corporation | Heating method and apparatus |
| KR880014009A (en) | 1987-05-04 | 1988-12-22 | 원본미기재 | Polyimide oligomers and mixtures thereof |
| KR910008339B1 (en) | 1987-09-17 | 1991-10-12 | 히다찌가세이고오교가부시끼가이샤 | Processes for preparation of polyimide-isoindoquin azoloine dione and precursor thereof |
-
1987
- 1987-12-23 US US07/137,493 patent/US5705598A/en not_active Expired - Fee Related
-
1995
- 1995-06-05 US US08/572,755 patent/US5739256A/en not_active Expired - Fee Related
- 1995-06-05 US US08/463,437 patent/US6583255B1/en not_active Expired - Fee Related
Patent Citations (483)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB907105A (en) | 1958-11-14 | 1962-10-03 | Nat Res Dev | New polymers having azo-links |
| US3105839A (en) | 1960-12-16 | 1963-10-01 | Ciba Ltd | Bicyclo (2. 2. 1)hept-5-ene-2, 3-dicarboxylic acid anhydrides |
| US3236808A (en) | 1961-01-03 | 1966-02-22 | Borg Warner | Polysulfonate copolymers |
| US3265708A (en) | 1961-03-01 | 1966-08-09 | Us Rubber Co | Nu-parahydroxyphenylmaleimide |
| US3236705A (en) | 1962-07-02 | 1966-02-22 | Monsanto Res Corp | Solid polymeric solutions containing metal salts such as lithium perchlorate |
| US3313783A (en) | 1962-07-20 | 1967-04-11 | Teijin Ltd | Process for preparation of polybenzimidazoles |
| US3267081A (en) | 1962-09-19 | 1966-08-16 | Koppers Co Inc | Benzothiazole dicarboxylic acids and polymers derived therefrom |
| US3386969A (en) | 1962-11-19 | 1968-06-04 | Whittaker Corp | Fusible polybenzimidazoles |
| US3624042A (en) | 1962-11-29 | 1971-11-30 | Aerojet General Co | Carboxy terminated polymer and cured derivative thereof |
| US4108837A (en) | 1963-07-16 | 1978-08-22 | Union Carbide Corporation | Polyarylene polyethers |
| US4175175A (en) | 1963-07-16 | 1979-11-20 | Union Carbide Corporation | Polyarylene polyethers |
| US3354129A (en) | 1963-11-27 | 1967-11-21 | Phillips Petroleum Co | Production of polymers from aromatic compounds |
| US3408349A (en) | 1963-12-04 | 1968-10-29 | American Cyanamid Co | Azine ion-radical salts |
| US3609181A (en) | 1964-05-28 | 1971-09-28 | Us Air Force | Polyesters of polyoxytetramethylene glycol and certain dioic acids |
| US3448084A (en) | 1964-06-10 | 1969-06-03 | Gulf Oil Corp | Polyesters from tris(2-hydroxyethyl) isocyanurate |
| US3458486A (en) | 1964-09-04 | 1969-07-29 | Phillips Petroleum Co | Production of polymers from cyclic compounds |
| US3355272A (en) | 1964-11-24 | 1967-11-28 | Union Carbide Corp | Abrasive compositions and structures thereof comprising a thermosetting resin and a thermoplastic polyarylene polyether |
| GB1069061A (en) | 1965-02-10 | 1967-05-17 | Ilford Ltd | N-aminophenyl-phthalimido derivatives and their use in photographic colour developing |
| US3262914A (en) | 1965-03-16 | 1966-07-26 | Borg Warner | Sulfonate-carboxylate copolymers |
| US3449467A (en) | 1965-06-03 | 1969-06-10 | Union Carbide Corp | Hydroxyl terminated polyester oligomers |
| US3454673A (en) | 1965-06-05 | 1969-07-08 | Ella Dannenbaum | Preparation of hardenable nitrogen containing synthetic resinous products |
| US3450711A (en) | 1965-07-01 | 1969-06-17 | American Cyanamid Co | Bis-imide containing diepoxide compounds |
| US3749735A (en) | 1965-10-24 | 1973-07-31 | Eastman Kodak Co | Certificate of correction |
| US3461461A (en) | 1965-11-01 | 1969-08-12 | Upjohn Co | 6-amino-4-(substituted amino)-1,2-dihydro-1-hydroxy-2-iminopyrimidines |
| GB1099096A (en) | 1965-12-10 | 1968-01-17 | Monsanto Chemicals | Nitrogen-containing aromatic compounds |
| US3431235A (en) | 1966-03-02 | 1969-03-04 | Trw Inc | Polydiene resins including cyclized polydiene resins,method of manufacture and uses |
| US3435003A (en) | 1966-06-01 | 1969-03-25 | Du Pont | Cross-linked thermally reversible polymers produced from condensation polymers with pendant furan groups cross-linked with maleimides |
| US3635891A (en) | 1966-07-14 | 1972-01-18 | Trw Inc | Internally plasticized polyidene resins |
| US3453236A (en) | 1967-01-18 | 1969-07-01 | Ashland Oil Inc | Aromatic polyimideamides prepared from aromatic tetracarboxylic acid dianhydrides and aminoaromatic hydrazides |
| US3530087A (en) | 1967-02-21 | 1970-09-22 | Ciba Ltd | Adhesive compositions of polyepoxide and polysulfones |
| US3563951A (en) | 1967-03-06 | 1971-02-16 | Bayer Ag | Polyimides of high molecular weight aromatic polyether diamines |
| US3803081A (en) | 1967-07-03 | 1974-04-09 | Trw Inc | Polyamide molding powder prepolymers and method of preparation |
| US3528950A (en) | 1967-07-03 | 1970-09-15 | Trw Inc | Polyimide polymers |
| US3647529A (en) | 1967-07-10 | 1972-03-07 | Trw Inc | Reinforced structural plastics |
| US3658764A (en) | 1967-07-13 | 1972-04-25 | Rhone Poulenc Sa | Cross-linked resins |
| US3562223A (en) | 1967-07-13 | 1971-02-09 | Rhone Poulenc Sa | Cross-linked resins |
| USRE29316E (en) | 1967-07-13 | 1977-07-19 | Rhone-Poulenc S.A. | Cross-linked resins |
| US3449442A (en) | 1967-11-13 | 1969-06-10 | Mobil Oil Corp | 2,4-diphenoxybromobenzene and 2,4-diphenoxychlorobenzene as new compounds and synthetic lubricants |
| US3632428A (en) | 1967-11-30 | 1972-01-04 | Trw Inc | Thermally stable coatings |
| US3592841A (en) | 1967-12-01 | 1971-07-13 | Richardson Co | Keto-carboxylic acids |
| US3536270A (en) | 1967-12-04 | 1970-10-27 | Lincoln Tool & Mfg Co | Span control for a stator winding machine |
| US3616193A (en) | 1968-05-14 | 1971-10-26 | Trw Inc | Thermoset polydiene resin adhesive bonded laminates and methods of making same |
| US3565549A (en) | 1968-06-20 | 1971-02-23 | Trw Inc | Process for preparing reinforced resinous structures |
| US3812159A (en) | 1969-02-13 | 1974-05-21 | Trw Inc | Polybasic aromatic carboxylic acids,esters and anhydrides |
| US3887582A (en) | 1969-04-25 | 1975-06-03 | Gen Electric | Bis-imide compositions |
| US3729446A (en) | 1969-07-01 | 1973-04-24 | Gen Electric | Imido-substituted polyester compositions |
| US3770697A (en) | 1969-07-01 | 1973-11-06 | Gen Electric | Curable polycarbonate compositions |
| US3652710A (en) | 1969-07-01 | 1972-03-28 | Gen Electric | Curable polyaryleneoxide compositions |
| US3598768A (en) | 1969-07-09 | 1971-08-10 | Monsanto Co | Hetero-aromatic azopolymers |
| US3641207A (en) | 1969-07-15 | 1972-02-08 | Uniroyal Inc | Thermoplastic resin blend of polysulfone resin and an ethylenepropylene terpolymer or graft derivative thereof |
| DE1951632C3 (en) | 1969-10-14 | 1977-08-11 | Siemens AG, 1000 Berlin und 8000 München | Thermoplastic molding compounds based on stable, finely crystalline polyolefins |
| US3697345A (en) | 1969-11-10 | 1972-10-10 | Trw Inc | Process for preparing polyimide laminates |
| US3697308A (en) | 1969-11-10 | 1972-10-10 | Trw Inc | Boron reinforced polyimide composites |
| US3631222A (en) | 1970-01-02 | 1971-12-28 | Minnesota Mining & Mfg | Polyarylsulfone graft and block copolymers with chains of polyamide acid or polyimide |
| US3663507A (en) | 1970-01-02 | 1972-05-16 | Minnesota Mining & Mfg | Linear polyarylsulfones having functional groups thereon |
| US3708459A (en) | 1970-06-24 | 1973-01-02 | Trw Inc | Molding power prepolymers |
| US3988374A (en) | 1970-06-29 | 1976-10-26 | Union Carbide Corporation | Polyamide-imides |
| US3853815A (en) | 1970-08-17 | 1974-12-10 | Trw Inc | Thermosetting cyclized polydiene resins |
| US3699074A (en) | 1970-08-21 | 1972-10-17 | Trw Inc | Polyimide prepolymer molding powders |
| US3699075A (en) | 1970-10-28 | 1972-10-17 | Trw Inc | Soluble high molecular weight polyimide |
| US3772250A (en) | 1970-12-08 | 1973-11-13 | Carborundum Co | Branched aromatic polyesters containing cyanuryl nucleus radicals |
| US3689464A (en) | 1970-12-18 | 1972-09-05 | Gen Electric | Imido-substituted polyamide compositions |
| US3658938A (en) | 1970-12-30 | 1972-04-25 | Union Carbide Corp | Polyamide-imide/polysulfone composites |
| US3972902A (en) | 1971-01-20 | 1976-08-03 | General Electric Company | 4,4'-Isopropylidene-bis(3- and 4-phenyleneoxyphthalic anhydride) |
| US3847867A (en) | 1971-01-20 | 1974-11-12 | Gen Electric | Polyetherimides |
| US3759777A (en) | 1971-01-20 | 1973-09-18 | Trw Inc | Process for producing reinforced plastic laminates from polydiene block polymers |
| US3970714A (en) | 1971-02-24 | 1976-07-20 | Rhone-Poulenc S.A. | Heat-stable polyimide resin modified by unsaturated polyester |
| US3773718A (en) | 1971-04-30 | 1973-11-20 | Gen Electric | Method for making imido-substituted aromatic polymers |
| US3708370A (en) | 1971-08-06 | 1973-01-02 | Trw Inc | Process for bonding surfaces with polyimide prepolymers |
| US3839287A (en) | 1971-09-21 | 1974-10-01 | Union Carbide Corp | Polyarylimides |
| US3895064A (en) | 1971-09-21 | 1975-07-15 | Union Carbide Corp | Polyamide-imides |
| US3966726A (en) | 1971-09-23 | 1976-06-29 | Akzo N.V. | Process for the production of cyclic carboxylic acid imides |
| US3708439A (en) | 1971-09-28 | 1973-01-02 | Upjohn Co | Polybenzimidazoles |
| US3745149A (en) | 1971-09-29 | 1973-07-10 | Nasa | Preparation of polyimides from mixtures of monomeric diamines and esters of polycarboxylic acids |
| US4266047A (en) | 1971-12-16 | 1981-05-05 | General Electric Company | Novel polymers of olefin terminated prepolymers and amine terminated prepolymers |
| US3925324A (en) | 1971-12-27 | 1975-12-09 | Rhone Poulenc Sa | Process for the preparation of thermosetting polymers from n,n-bis-maleimide |
| US3827927A (en) | 1971-12-30 | 1974-08-06 | Trw Inc | Method of bonding using improved polyimide adhesives |
| US3781240A (en) | 1971-12-30 | 1973-12-25 | Trw Inc | Polyimide molding powders |
| US3781249A (en) | 1971-12-30 | 1973-12-25 | Trw Inc | Polyamide-imides and precursors thereof |
| FR2166209B1 (en) | 1971-12-30 | 1976-01-30 | Trw Inc Us | |
| US3761441A (en) | 1972-02-02 | 1973-09-25 | Union Carbide Corp | Thermosetting friction compositions and friction elements thereof containing an acrylimide resin |
| US3757088A (en) | 1972-02-29 | 1973-09-04 | Hercules Inc | He same electrically conducting elastomers and electrical members embodying t |
| US3748312A (en) | 1972-04-03 | 1973-07-24 | Trw Inc | Thermosetting hydrazide polymers |
| US3748311A (en) | 1972-04-03 | 1973-07-24 | Trw Inc | Heterocyclic endo alicyclic end capped resins |
| US3763101A (en) | 1972-04-03 | 1973-10-02 | Trw Inc | Thermosetting alicyclic endo end capped resins |
| US3859252A (en) | 1972-04-28 | 1975-01-07 | Uniroyal Inc | Polyamido polysulfone thermoplastics and intermediates thereof |
| US3843593A (en) | 1972-06-05 | 1974-10-22 | Du Pont | Radar absorptive coating composition of an acrylic polymer,a polyester and an isocyanate cross-linking agent |
| US3956320A (en) | 1972-08-18 | 1976-05-11 | General Electric Company | Aromatic bis(ether phthalis anhydride) compounds |
| GB1453625A (en) | 1972-12-18 | 1976-10-27 | Toray Industries | Polyamide imide resin and prepolymer therefor |
| FR2210635B1 (en) | 1972-12-18 | 1978-03-03 | Toray Industries | |
| US3957732A (en) | 1973-02-02 | 1976-05-18 | Sumitomo Chemical Company, Limited | Alternating copolymers having functional group and process for producing same |
| US3957862A (en) | 1973-03-30 | 1976-05-18 | General Electric Company | Method of making aromatic bis(ether ortho phthalic acid)s |
| US3879428A (en) | 1973-03-30 | 1975-04-22 | Gen Electric | Method for making aromatic bis(ether anhydride)s |
| US3935320A (en) | 1973-04-04 | 1976-01-27 | Union Carbide Corporation | Tubular cellulosic casing with cationic thermosetting resin coating |
| US3966678A (en) | 1973-04-26 | 1976-06-29 | Rhone-Poulenc S.A. | Maleic anhydride-polyaldimine prepolymers |
| US4075171A (en) | 1973-05-25 | 1978-02-21 | University Of Notre Dame Du Lac | Process for preparing aromatic polyimides and some polyimides prepared thereby |
| US3897395A (en) | 1973-05-25 | 1975-07-29 | Univ Notre Dame Du Lac | Process for chain-extending unsaturated terminated polyimides and products prepared therefrom |
| US4055543A (en) | 1973-05-25 | 1977-10-25 | Alelio Gaetano Francis D | Chain-extending Schiff base terminated polyimides and method of preparation |
| US4026871A (en) | 1973-05-25 | 1977-05-31 | University Of Notre Dame | Chain-extending phenolic end-capped polyimides |
| US4058505A (en) | 1973-05-25 | 1977-11-15 | University Of Notre Dame Du Lac | Chain-extending amine end-capped polyimides |
| US3890272A (en) | 1973-05-25 | 1975-06-17 | Univ Notre Dame Du Lac | Polymerizing bis-maleimides |
| US3998786A (en) | 1973-05-25 | 1976-12-21 | University Of Notre Dame Du Lac | Process for preparing aromatic polyimides, polyimides prepared thereby |
| US4060515A (en) | 1973-05-25 | 1977-11-29 | University Of Notre Dame Du Lac | Chain-extending nitrile end-capped polyimides |
| US3879393A (en) | 1973-06-18 | 1975-04-22 | Miles Lab | Derivatives of 1,3-disubstituted 2,4(1h,3h)-quinazolinediones |
| US3847869A (en) | 1973-09-28 | 1974-11-12 | Gen Electric | Method for preparing polyetherimides |
| US3879349A (en) | 1973-11-12 | 1975-04-22 | Hughes Aircraft Co | Homopolymerization of acetylene substituted polyimide polymers |
| US3919177A (en) | 1973-11-19 | 1975-11-11 | Phillips Petroleum Co | P-phenylene sulfide polymers |
| US3909507A (en) | 1973-12-06 | 1975-09-30 | Gen Electric | Electrical conductors with strippable polymeric materials |
| US4051177A (en) | 1973-12-18 | 1977-09-27 | Bayer Aktiengesellschaft | Process for the preparation of unsaturated amino compounds |
| US3993630A (en) | 1973-12-26 | 1976-11-23 | Ciba-Geigy Corporation | Polyimide, polyamide-imide or polyamide-acids with olefin end groups in combination with a reactive Schiff base compound |
| US3941746A (en) | 1973-12-28 | 1976-03-02 | Ciba-Geigy Corporation | Containing hindered phenolic norbornane-2,3-dicarboximides stabilized compositions |
| US3896147A (en) | 1973-12-28 | 1975-07-22 | Ciba Geigy Corp | Hindered phenolic norbornane-2,3-dicarboximides |
| US3920768A (en) | 1974-01-14 | 1975-11-18 | Union Carbide Corp | Arylimide-epoxy resin composites |
| US4000146A (en) | 1974-01-31 | 1976-12-28 | Horizons Incorporated, A Division Of Horizons Research Incorporated | Triamino pyridine compounds |
| US3966987A (en) | 1974-04-17 | 1976-06-29 | International Flavors & Fragrances Inc. | Electroconductive high polymer composition |
| US4013600A (en) | 1974-05-20 | 1977-03-22 | Rhone-Poulenc Industries | Aqueous compositions of polyimides containing hetero-polysaccharide |
| FR2272119B1 (en) | 1974-05-20 | 1977-03-18 | Rhone Poulenc Ind | |
| US4100137A (en) | 1974-05-30 | 1978-07-11 | Polysar Limited | Crosslinking of hydroxyl-containing polymers with polyaldehyde compounds |
| US3935167A (en) | 1974-06-06 | 1976-01-27 | University Patents, Inc. | Acetylenic polymers, trimers thereof and the production thereof |
| US3914334A (en) | 1974-07-31 | 1975-10-21 | Trw Inc | Flexible electrical insulating polymeric sheet |
| US4064289A (en) | 1974-08-21 | 1977-12-20 | Hitachi, Ltd. | Method of making a semiconductor device |
| US3933862A (en) | 1974-08-23 | 1976-01-20 | General Electric Company | Aromatic dithio dianhydrides |
| US4107153A (en) | 1974-09-25 | 1978-08-15 | Toshiba Chemical Products Co., Ltd. | Heat-resistant thermosetting polyimide resin |
| US4101488A (en) | 1974-09-26 | 1978-07-18 | Nitto Electric Industrial Co., Ltd. | Water-soluble heat-resistant insulating varnish |
| FR2303818B1 (en) | 1975-03-10 | 1977-11-18 | Rhone Poulenc Ind | |
| US4197397A (en) | 1975-03-14 | 1980-04-08 | University Of Notre Dame Du Lac | Chain-extending anhydride end-capped polyimides |
| US4100138A (en) | 1975-05-12 | 1978-07-11 | Hughes Aircraft Company | Ethnyl terminated polyimide composites |
| US4038261A (en) | 1975-05-27 | 1977-07-26 | Phillips Petroleum Company | Production of branched arylene sulfide polymers |
| US4189560A (en) | 1975-06-18 | 1980-02-19 | Ciba-Geigy Corporation | Crosslinkable polymers of unsaturated imides |
| US4193927A (en) | 1975-06-18 | 1980-03-18 | Ciba-Geigy Corporation | Imidyl compounds |
| US4172836A (en) | 1975-06-18 | 1979-10-30 | Ciba-Geigy Corporation | Imidyl compounds |
| US4187364A (en) | 1975-06-18 | 1980-02-05 | Ciba-Geigy Corporation | Crosslinkable polymers of unsaturated imides |
| US4158731A (en) | 1975-06-18 | 1979-06-19 | Ciba-Geigy Corporation | Crosslinkable polymeric compounds |
| US4174326A (en) | 1975-06-18 | 1979-11-13 | Ciba-Geigy Corporation | Imidyl compounds |
| US4134895A (en) | 1975-06-18 | 1979-01-16 | Ciba-Geigy Corporation | Imidyl-benzenedicarboxylic acid derivatives |
| US4107174A (en) | 1975-06-18 | 1978-08-15 | Ciba-Geigy Corporation | Imidyl compounds |
| US4132716A (en) | 1975-06-18 | 1979-01-02 | Ciba-Geigy Corporation | Imidyl-benzene-dicarboxylic and -tricarboxylic acid derivatives |
| US4239883A (en) | 1975-07-09 | 1980-12-16 | Technochemie Gmbh Verfahrenstechnik | Polyimide preparation from imido acyl halide |
| US4020069A (en) | 1975-07-25 | 1977-04-26 | General Electric Company | Preparation of aromatic tetracarboxylic acids |
| US4005134A (en) | 1975-09-11 | 1977-01-25 | General Electric Company | Method for making aromatic bis(ether dicarboxylic acid) |
| US4115362A (en) | 1975-10-03 | 1978-09-19 | Teijin Limited | Process for preparing polyesters |
| US4288607A (en) | 1975-10-25 | 1981-09-08 | Dynamit Nobel Aktiengesellschaft | Bis-carbalkoxy compounds and their preparation |
| US4124593A (en) | 1975-11-13 | 1978-11-07 | Ciba-Geigy Corporation | Aromatic dicarboximides |
| US4126619A (en) | 1975-12-19 | 1978-11-21 | Ciba-Geigy Corporation | Crosslinkable bis-imidyl derivatives |
| US4115231A (en) | 1975-12-19 | 1978-09-19 | Ciba-Geigy Corporation | Crosslinkable mixtures of oligomers or polymers and diamines |
| US4365068A (en) | 1975-12-19 | 1982-12-21 | Ciba-Geigy Corporation | Crosslinkable bis-imidyl derivatives |
| US4183839A (en) | 1976-04-08 | 1980-01-15 | John V. Long | Polyimide resin-forming composition |
| US4132715A (en) | 1976-04-09 | 1979-01-02 | Ciba-Geigy Corporation | Process for the production of maleimides |
| US4142870A (en) | 1976-07-01 | 1979-03-06 | Abrasives International N.V. | Abrasive articles prepared from crosslinked aromatic polyimides |
| US4111879A (en) | 1976-07-10 | 1978-09-05 | Ihara Chemical Industry Co., Ltd. | Composition for inhibiting adhesion of shellfish and algae |
| US4116937A (en) | 1976-08-25 | 1978-09-26 | Trw Inc. | Compliant maleimide based plastics |
| US4128574A (en) | 1976-10-04 | 1978-12-05 | General Electric Company | Method for making organic polycarboxylic acids |
| US4108926A (en) | 1976-10-28 | 1978-08-22 | The United States Of America As Represented By The Secretary Of The Air Force | Reactive plasticizer for thermoplastic polysulfone resins |
| US4167663A (en) | 1977-01-24 | 1979-09-11 | Baxter Travenol Laboratories, Inc. | Blood warming apparatus |
| US4097456A (en) | 1977-03-28 | 1978-06-27 | Gulf Research & Development Company | Preparation of acetylene-substituted polyimide oligomers and polyimide polymers |
| GB2002378B (en) | 1977-08-09 | 1982-03-10 | Rhone Poulenc Ind | Preparation of oligo-imides |
| US4225498A (en) | 1977-08-09 | 1980-09-30 | Rhone-Poulenc Industries | Preparation of N-substituted oligo-imides |
| US4297472A (en) | 1977-08-26 | 1981-10-27 | Mobay Chemical Corporation | Method of producing color stable thermoplastic polymethanes and products made thereby |
| US4107147A (en) | 1977-09-02 | 1978-08-15 | General Electric Company | Polysulfoneimides |
| US4299946A (en) | 1977-10-14 | 1981-11-10 | Rhone-Poulenc Industries | Imido copolymers from oligoimide and phenolic compound |
| US4250096A (en) | 1977-10-14 | 1981-02-10 | Ciba-Geigy Corporation | 3- and 4-Azidophthalic acid derivatives |
| US4269961A (en) | 1977-10-25 | 1981-05-26 | Trw, Inc | Low temperature curable compliant bismaleimide compositions |
| US4179551A (en) | 1977-10-25 | 1979-12-18 | Trw, Inc. | Low temperature curable compliant bismaleimide compositions |
| US4289699A (en) | 1977-11-02 | 1981-09-15 | Mitsui Toatsu Chemicals, Incorporated | Process for the production of N-(hydroxyphenyl) maleimides |
| US4231934A (en) | 1977-11-02 | 1980-11-04 | Mitsui Toatsu Chemicals, Incorporated | Process for the production of N-(hydroxyphenyl) maleimides |
| US4225497A (en) | 1978-02-19 | 1980-09-30 | Rhone-Poulenc Industries | Preparation of N-substituted oligo-imides |
| US4221895A (en) | 1978-04-18 | 1980-09-09 | The Dow Chemical Company | Thermally resistant thermosetting aromatic polymers containing pendant 1-alkynyl substituents |
| US4168366A (en) | 1978-07-25 | 1979-09-18 | Plastics Engineering Company | Polyimide derivatives |
| US4166168A (en) | 1978-07-25 | 1979-08-28 | Plastics Engineering Company | Polymerization products of acetylene-terminated polyimide derivatives |
| US4271079A (en) | 1978-09-07 | 1981-06-02 | Dainippon Ink & Chemicals, Inc. | 5-(2,5-Dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride |
| US4218555A (en) | 1978-11-01 | 1980-08-19 | Gulf Oil Corporation | Process for preparation of acetylene end-capped polyimide oligomers |
| US4400613A (en) | 1978-12-04 | 1983-08-23 | General Electric Company | Temperature controller for a fusing roller |
| US4176223A (en) | 1978-12-18 | 1979-11-27 | E. I. Du Pont De Nemours And Company | Polyimide-esters and filaments prepared from 4-(4'-carboxyphthalimido)benzoic acid or 4-(4'-hydroxyphthalimido)phenol |
| US4288583A (en) | 1978-12-29 | 1981-09-08 | Ciba-Geigy Corporation | Curable mixtures based on maleimides and 1-propenyl-substituted phenols |
| US4200731A (en) | 1979-02-01 | 1980-04-29 | The Goodyear Tire & Rubber Company | Endcapped polyesters |
| US4237262A (en) | 1979-02-14 | 1980-12-02 | Trw Inc. | Curable aliphatic epoxy-polimide compositions |
| US4273916A (en) | 1979-02-14 | 1981-06-16 | Trw Inc. | Curable cycloaliphatic epoxy-polyimide compositions |
| US4206106A (en) | 1979-04-05 | 1980-06-03 | Gulf Oil Corporation | Vinyl end-capped polyimide oligomers |
| US4251419A (en) | 1979-04-05 | 1981-02-17 | Gulf Oil Corporation | Novel vinyl end-capped polyimide oligomers |
| USRE30922E (en) | 1979-04-05 | 1982-05-04 | Gulf Oil Corporation | Vinyl end-capped polyimide oligomers |
| US4244853A (en) | 1979-04-06 | 1981-01-13 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Composition and method for making polyimide resin-reinforced fabric |
| US4255313A (en) | 1979-04-20 | 1981-03-10 | Gulf Oil Corporation | Novel end-capped polyimide oligomers |
| US4251420A (en) | 1979-04-23 | 1981-02-17 | Gulf Oil Corporation | End-capped polyimide oligomers |
| US4251418A (en) | 1979-04-27 | 1981-02-17 | Gulf Oil Corporation | Polyimide oligomers |
| US4299750A (en) | 1979-05-03 | 1981-11-10 | Gulf Oil Corporation | Novel partially acetylene end-capped polyimide oligomers |
| US4323662A (en) | 1979-07-10 | 1982-04-06 | Mitsui Toatsu Chemicals, Inc. | Thermosetting resin compositions comprising bismaleimides and alkenylaniline derivatives |
| US4298720A (en) | 1979-07-23 | 1981-11-03 | Mitsui Toatsu Chemicals Incorporated | Thermosetting resin composition from maleimide compound and alkenyl phenol |
| US4276407A (en) | 1979-08-16 | 1981-06-30 | Hughes Aircraft Company | Acetylene terminated imide oligomers which melt at low temperatures |
| US4234712A (en) | 1979-09-14 | 1980-11-18 | The United States Of America As Represented By The Secretary Of The Navy | Polyphthalocyanine resins |
| US4375427A (en) | 1979-12-13 | 1983-03-01 | Allied Corporation | Thermoplastic conductive polymers |
| US4302575A (en) | 1979-12-26 | 1981-11-24 | General Electric Company | Heat curable polyimides |
| US4293670A (en) | 1979-12-26 | 1981-10-06 | Union Carbide Corporation | Blends of poly(aryl ether) resins and polyetherimide resins |
| US4360644A (en) | 1980-01-07 | 1982-11-23 | Basf Aktiengesellschaft | Preparation of electrically conductive polyaromatics |
| US4358561A (en) | 1980-01-16 | 1982-11-09 | Standard Oil Company (Indiana) | Injection moldable amide-imide copolymers |
| US4460783A (en) | 1980-01-21 | 1984-07-17 | Hitachi, Ltd. | Ether imides and process for producing the same |
| US4393188A (en) | 1980-02-14 | 1983-07-12 | Hitachi Chemical Company, Ltd. | Thermosetting prepolymer from polyfunctional maleimide and bis maleimide |
| US4297474A (en) | 1980-02-29 | 1981-10-27 | General Electric Company | Polyetherimides prepared by self-condensation of hydroxyaryl phthalimide salts |
| US4344869A (en) | 1980-03-12 | 1982-08-17 | Basf Aktiengesellschaft | Preparation of electrically conductive hetero-poly-phenylenes |
| US4344870A (en) | 1980-03-12 | 1982-08-17 | Basf Aktiengesellschaft | Preparation of electrically conductive hetero-polyaromatics |
| US4351932A (en) | 1980-03-27 | 1982-09-28 | Hitco | Bis-maleimide/divinyl aryl crosslinking agent resin system |
| US4338222A (en) | 1980-04-11 | 1982-07-06 | Xerox Corporation | Semiconductive organic compositions |
| US4395497A (en) | 1980-07-08 | 1983-07-26 | Basf Aktiengesellschaft | Preparation of electrically conductive soluble heteropolyphenylenes |
| US4338225A (en) | 1980-08-04 | 1982-07-06 | The Boeing Company | High performance resin reaction products of carboxyl terminated 1,2-polybutadiene with epoxides and acid terminated difunctional aliphatic alcohols |
| US4739115A (en) | 1980-09-25 | 1988-04-19 | Mcdonnell Douglas Corporation | Poly phosphinoguanidine compounds |
| US4446191A (en) | 1980-10-27 | 1984-05-01 | Hitachi Chemical Company, Ltd. | Laminates comprising prepregs metal clad |
| US4407739A (en) | 1981-02-07 | 1983-10-04 | Basf Aktiengesellschaft | Preparation of electrically conductive polymers |
| US4376710A (en) | 1981-03-30 | 1983-03-15 | Hughes Aircraft Company | High load carrying polyimide lubricative composites |
| US4423202A (en) | 1981-05-05 | 1983-12-27 | Celanese Corporation | Process for the production of high molecular weight para ordered aromatic heterocyclic polymer |
| US4418181A (en) | 1981-05-26 | 1983-11-29 | Plastics Engineering Company | Polyimides having bis-maleimide terminal groups |
| US4438280A (en) | 1981-05-26 | 1984-03-20 | Plastics Engineering Company | Bis-(maleamic acid) derivatives of triamines |
| US4661604A (en) | 1981-06-16 | 1987-04-28 | Trw, Inc. | Monofunctional crosslinking imidophenols |
| US4414269A (en) | 1981-06-16 | 1983-11-08 | Trw, Inc. | Solvent resistant polysulfone and polyethersulfone compositions |
| EP0067976B1 (en) | 1981-06-16 | 1987-03-04 | The Boeing Company | Method for the preparation of cross-linkable polymers |
| CA1175998A (en) | 1981-06-16 | 1984-10-09 | Hyman R. Lubowitz | Solvent resistant polysulfone and polyethersulfone compositions |
| US4482683A (en) | 1981-07-10 | 1984-11-13 | Siemens Aktiengesellschaft | Stabilized sulfur-containing resins, such as polyphenylene sulfide and method of producing the same |
| US4485231A (en) | 1981-07-24 | 1984-11-27 | Hughes Aircraft Company | Isoimide containing oligomers |
| US4438273A (en) | 1981-07-24 | 1984-03-20 | Hughes Aircraft Company | Isoimide containing oligomers |
| US4405770A (en) | 1981-08-12 | 1983-09-20 | National Starch And Chemical Corporation | Novel polyimides, and polyamic acid and ester intermediates thereof |
| US4381363A (en) | 1981-09-10 | 1983-04-26 | The United States Of America As Represented By The Secretary Of The Air Force | Cure retarding additives for acetylene-terminated polymers |
| US4389504A (en) | 1981-10-02 | 1983-06-21 | The United States Of America As Represented By The United States National Aeronautics And Space Administration Office Of General Counsel-Code Gp | Elastomer toughened polyimide adhesives |
| US4814472A (en) | 1981-11-06 | 1989-03-21 | Hughes Aircraft Company | Diethynylated diphenyl hexafluoropropanes |
| US4410686A (en) | 1981-12-21 | 1983-10-18 | The Dow Chemical Company | Norbornyl modified polyesteramides and process for preparing same |
| US4409382A (en) | 1982-01-07 | 1983-10-11 | The United States Of America As Represented By The Secretary Of The Navy | Synthesis and polymerization of phthalonitrile monomers containing multiple phenoxy and sulfone linkages |
| US4617390A (en) | 1982-02-23 | 1986-10-14 | Basf Aktiengesellschaft | S-triazine derivatives |
| US4520198A (en) | 1982-03-02 | 1985-05-28 | Plastics Engineering Company | Vinylacetylene-terminated polyimide derivatives |
| US4417044A (en) | 1982-05-25 | 1983-11-22 | General Electric Company | Process for making polyetherimides |
| US5230956A (en) | 1982-05-28 | 1993-07-27 | Amoco Corporation | Polyamide-imide sized fibers |
| US4467011A (en) | 1982-05-28 | 1984-08-21 | Standard Oil Company (Indiana) | Injection moldable amide-imide polymers and copolymers containing amorphous polyamides |
| US4417039A (en) | 1982-07-19 | 1983-11-22 | The United States Of America As Represented By The Secretary Of The Air Force | Cis-enyne aromatic and aromatic heterocyclic polymers |
| US4590363A (en) | 1982-07-28 | 1986-05-20 | Ersa Ernst Sachs Kg Gmbh & Co. | Circuit for controlling temperature of electric soldering tool |
| US4547553A (en) | 1982-07-29 | 1985-10-15 | The Boeing Company | Polybutadiene modified polyester compositions |
| US4515962A (en) | 1982-08-05 | 1985-05-07 | Ciba Geigy Corporation | Allyl or methallyl-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid imides and bisimides |
| US4448925A (en) | 1982-08-06 | 1984-05-15 | Standard Oil Company (Indiana) | Injection moldable polymers containing phthalic anhydride |
| US4755585A (en) | 1982-08-06 | 1988-07-05 | Amoco Corporation | Injection moldable polyamide imide from trimellitic anhydride end capped with aniline |
| US4564553A (en) | 1982-08-09 | 1986-01-14 | Merak S.P.A. | Manufactured articles, including fabrics comprising thermally bonded polyolefinic fibers |
| US4489027A (en) | 1982-08-11 | 1984-12-18 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Process for preparing solvent resistant, thermoplastic aromatic poly(imidesulfone) |
| US4526838A (en) | 1982-09-08 | 1985-07-02 | Hitachi Chemical Company, Ltd. | Polyamino-bis-imide resin |
| US4533692A (en) | 1982-09-17 | 1985-08-06 | Sri International | Liquid crystalline polymer compositions, process, and products |
| US4533693A (en) | 1982-09-17 | 1985-08-06 | Sri International | Liquid crystalline polymer compositions, process, and products |
| US4533724A (en) | 1982-09-17 | 1985-08-06 | Sri International | Liquid crystalline poly (2,6-benzothiazole) compositions, process, and products |
| US4429108A (en) | 1982-09-27 | 1984-01-31 | Standard Oil Company (Indiana) | Poly[N,N'-(alkyl or aryl)-bis(4,4'-phthalimido)-1,3,4-oxadiazole-2,5-diyls] |
| US4476295A (en) | 1982-09-27 | 1984-10-09 | Standard Oil Company (Indiana) | Poly-imide-oxadiazole soluble in m-cresol |
| US4465809A (en) | 1982-11-05 | 1984-08-14 | Phillips Petroleum Company | Conversion of polymer lithium to polymer carboxylate |
| US4638027A (en) | 1982-12-27 | 1987-01-20 | General Electric Company | Polycarbonate exhibiting improved heat resistance from cycloalkylidene diphenol |
| US4574154A (en) | 1982-12-28 | 1986-03-04 | Ube Industries, Ltd. | Triazine addition compounds |
| US4616071A (en) | 1983-01-06 | 1986-10-07 | Ford Motor Company | Coating composition comprising bis-diene oligomers and bis-dieneophile oligomers |
| US4737550A (en) | 1983-01-07 | 1988-04-12 | The Dow Chemical Company | Bridged dense star polymers |
| US4558120A (en) | 1983-01-07 | 1985-12-10 | The Dow Chemical Company | Dense star polymer |
| US4568737A (en) | 1983-01-07 | 1986-02-04 | The Dow Chemical Company | Dense star polymers and dendrimers |
| US4631337A (en) | 1983-01-07 | 1986-12-23 | The Dow Chemical Company | Hydrolytically-stable dense star polyamine |
| US4507466A (en) | 1983-01-07 | 1985-03-26 | The Dow Chemical Corporation | Dense star polymers having core, core branches, terminal groups |
| US4660057A (en) | 1983-01-21 | 1987-04-21 | Konishiroku Photo Industry Co., Ltd. | Heater power controller for ink jet printer |
| US4519926A (en) | 1983-02-18 | 1985-05-28 | Standard Oil Company (Indiana) | Polyborate esters and their use in lubricants |
| US4576857A (en) | 1983-03-14 | 1986-03-18 | E. I. Du Pont De Nemours And Company | Melt-fusible polyimides |
| US4510272A (en) | 1983-03-16 | 1985-04-09 | Avco Corporation | Bis-maleimide-epoxy compositions and prepregs |
| US4629777A (en) | 1983-05-18 | 1986-12-16 | Ciba-Geigy Corporation | Polyimides, a process for their preparation and their use |
| US4760106A (en) | 1983-05-20 | 1988-07-26 | Amoco Corporation | Impact resistant matrix resins for advanced composites |
| US4837256A (en) | 1983-06-13 | 1989-06-06 | Amoco Corporation | Crosslinkable, crystallizable polyarylethers and composites therefrom |
| US4536559A (en) | 1983-06-17 | 1985-08-20 | The Boeing Company | Thermally stable polyimide polysulfone compositions for composite structures |
| US4739030A (en) | 1983-06-17 | 1988-04-19 | The Boeing Company | Difunctional end-cap monomers |
| US5175233A (en) | 1983-06-17 | 1992-12-29 | The Boeing Company | Multidimensional ester or ether oligomers with pyrimidinyl end caps |
| US5227461A (en) | 1983-06-17 | 1993-07-13 | The Boeing Company | Extended difunctional end-cap monomers |
| US5159055A (en) | 1983-06-17 | 1992-10-27 | The Boeing Company | Coreactive oligomer blends |
| US5112939A (en) | 1983-06-17 | 1992-05-12 | The Boeing Company | Oligomers having pyrimidinyl end caps |
| US4851501A (en) | 1983-06-17 | 1989-07-25 | The Boeing Company | Polyethersulfone prepregs, composites, and blends |
| US5210213A (en) | 1983-06-17 | 1993-05-11 | The Boeing Company | Dimensional, crosslinkable oligomers |
| EP0310735A1 (en) | 1983-06-17 | 1989-04-12 | The Boeing Company | Polysulfone and polyethersulfone oligomers |
| US4980481A (en) | 1983-06-17 | 1990-12-25 | The Boeing Company | End-cap monomers and oligomers |
| EP0132547B1 (en) | 1983-06-20 | 1989-02-22 | General Electric Company | Bisimides and method for their use in polyimide synthesis |
| US4578470A (en) | 1983-06-20 | 1986-03-25 | General Electric Company | Bis-imides containing heterocyclic aromatic rings |
| US4608414A (en) | 1983-07-19 | 1986-08-26 | Denki Kagaku Kogyo Kabushiki Kaisha | Thermoplastic resin composition containing an imide polymer and graft copolymer |
| US4556697A (en) | 1983-07-25 | 1985-12-03 | The Standard Oil Company | Alternating copolyamide prepared on a polymer matrix |
| US4476184A (en) | 1983-08-09 | 1984-10-09 | The Boeing Company | Thermally stable polysulfone compositions for composite structures |
| US4714768A (en) | 1983-08-25 | 1987-12-22 | Akademie Der Wissenschaften Der Ddr | N-(chlorocarbonyloxy)-5-norbornene-2,3-dicarboximide, process for its production and its use |
| US4628079A (en) | 1983-09-06 | 1986-12-09 | Bayer Aktiengesellschaft | Polyamide imides produced from either lactams or polyamides with isocyanates or anhydrides in which products are precipitated with a non-solvent |
| US4485140A (en) | 1983-09-21 | 1984-11-27 | E. I. Du Pont De Nemours And Company | Melt-fusible polyimides |
| US4615832A (en) | 1983-09-26 | 1986-10-07 | Bayer Aktiengesellschaft | Flameproofing agents, their preparation and their use for flameproofing polycarbonates |
| US5344894A (en) | 1983-09-27 | 1994-09-06 | The Boeing Company | Polyimide oligomers and blends |
| US5175234A (en) | 1983-09-27 | 1992-12-29 | The Boeing Company | Lightly crosslinked polyimides |
| US4684714A (en) | 1983-09-27 | 1987-08-04 | The Boeing Company | Method for making polyimide oligomers |
| US5286811A (en) | 1983-09-27 | 1994-02-15 | The Boeing Company | Blended polyimide oligomers and method of curing polyimides |
| US4535117A (en) | 1983-11-02 | 1985-08-13 | Phillips Petroleum Company | Stabilization of polyphenylene sulfide |
| US4600769A (en) | 1983-12-15 | 1986-07-15 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Amine terminated bisaspartimide polymer |
| US4567216A (en) | 1983-12-22 | 1986-01-28 | Union Carbide Corporation | Thermoplastic modified epoxy compositions |
| US4691025A (en) | 1983-12-22 | 1987-09-01 | Amoco Corporation | Bismaleimides and prepreg resins therefrom |
| US4578433A (en) | 1983-12-24 | 1986-03-25 | Basf Aktiengesellschaft | Long-term stability of the electrical conductivity of pyrrole polymers |
| US4663378A (en) | 1983-12-29 | 1987-05-05 | General Electric Company | Silicone-modified polyethermides |
| US4504632A (en) | 1983-12-30 | 1985-03-12 | General Electric Company | Amide ether imide block copolymers |
| US4628067A (en) | 1984-01-16 | 1986-12-09 | Celanese Corporation | Microporous polybenzimidazole particulates |
| US4657990A (en) | 1984-01-19 | 1987-04-14 | Imperial Chemical Industries Plc | Aromatic polyetherketones |
| US4657987A (en) | 1984-01-27 | 1987-04-14 | General Electric Company | Polyetherimide-polyamide blends |
| EP0152372B1 (en) | 1984-01-31 | 1991-12-27 | Ciba-Geigy Ag | Substituted unsaturated bicyclic imides and their polymers |
| US4742166A (en) | 1984-01-31 | 1988-05-03 | Ciba-Geigy Corporation | Substituted, unsaturated, bicyclic imides and polymers thereof |
| US4604437A (en) | 1984-01-31 | 1986-08-05 | Ciba-Geigy Corporation | Polymer from substituted, unsaturated, bicyclic imide |
| US4640944A (en) | 1984-01-31 | 1987-02-03 | Standard Oil Company (Indiana) | Injection moldable polyamide-imide-phthalamide copolymers containing polyetherimides |
| US4584364A (en) | 1984-02-06 | 1986-04-22 | The Boeing Company | Phenolic-capped imide sulfone resins |
| US4891460A (en) | 1984-03-09 | 1990-01-02 | San-Petrochemicals Co., Ltd. | Process for producing alkylidenenorbornenes |
| US4504620A (en) | 1984-04-16 | 1985-03-12 | E. I. Du Pont De Nemours And Company | Oxazoline polyester oligomers |
| US4663423A (en) | 1984-04-16 | 1987-05-05 | Teijin Limited | Polyester and process for preparation thereof |
| US4599383A (en) | 1984-04-28 | 1986-07-08 | Ntn-Rulon Industries Co., Ltd. | Polyamideimide resin composition |
| US4616070A (en) | 1984-05-08 | 1986-10-07 | Basf Aktiengesellschaft | Thermoplastic molding materials |
| US4649080A (en) | 1984-05-08 | 1987-03-10 | Basf Aktiengesellschaft | Fiber-reinforced materials |
| US4567240A (en) | 1984-05-23 | 1986-01-28 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Ethynyl terminated ester oligomers and polymers therefrom |
| US4728742A (en) | 1984-06-27 | 1988-03-01 | Ciba-Geigy Corporation | Substituted, unsaturated, bicyclic imides containing hydroxyl groups, and polymers thereof |
| US4563498A (en) | 1984-07-05 | 1986-01-07 | General Electric Company | Curable silicone compositions and processes |
| US4587329A (en) | 1984-08-17 | 1986-05-06 | The Dow Chemical Company | Dense star polymers having two dimensional molecular diameter |
| CA1269576A (en) | 1984-08-20 | 1990-05-29 | Bill W. Cole | Sizing agents |
| EP0175484A2 (en) | 1984-08-20 | 1986-03-26 | Amoco Corporation | Sized fibres |
| US4826997A (en) | 1984-08-27 | 1989-05-02 | The Dow Chemical Company | N-Substituted arylcyclo butenyl-maleimides |
| US4562231A (en) | 1984-09-06 | 1985-12-31 | Atlantic Richfield Company | Polyether sulfone resin containing molding composition and method |
| US5120819A (en) | 1984-09-18 | 1992-06-09 | Boeing Company | High performance heterocycles |
| US5066541A (en) | 1984-09-18 | 1991-11-19 | The Boeing Company | Heterocycle oligomer blends |
| US5198526A (en) | 1984-09-18 | 1993-03-30 | The Boeing Company | Heterocycle oligomers with multidimensional morphology |
| US4965336A (en) | 1984-09-18 | 1990-10-23 | The Boeing Company | High performance heterocycle oligomers and blends |
| US5082905A (en) | 1984-09-18 | 1992-01-21 | The Boeing Company | Blended heterocycles |
| US5126410A (en) | 1984-09-18 | 1992-06-30 | The Boeing Company | Heterocycle oligomers |
| US4804724A (en) | 1984-09-28 | 1989-02-14 | Amoco Corporation | Blends of a biphenyl containing poly (aryl ether sulfone) and a poly (aryl ether ketone) |
| US4556705A (en) | 1984-10-26 | 1985-12-03 | General Electric Company | Thermoplastic polyetherimide ester elastomers |
| US4709004A (en) | 1984-11-05 | 1987-11-24 | The Dow Chemical Company | Polyesteramide from hydroxy phenyl-isopropyl-amino diphenyl ether |
| US4663424A (en) | 1984-11-27 | 1987-05-05 | Bayer Aktiengesellschaft | Aromatic polyester with dimethylmaleimide end groups |
| US4804722A (en) | 1984-12-05 | 1989-02-14 | Hoechst Ag | Resin solutions for cements and coating compositions, process for their preparation and their use |
| US4555563A (en) | 1984-12-07 | 1985-11-26 | The Dow Chemical Company | Imide functional polyphenols; thermosettable compositions containing same and cured products therefrom |
| US4574144A (en) | 1984-12-17 | 1986-03-04 | General Electric Company | Sulfonated polyimides and polyamic acids and method for their preparation |
| US4562232A (en) | 1984-12-24 | 1985-12-31 | General Electric Company | Copolyetherester-dimer ester-block copolymers |
| US4835197A (en) | 1985-01-04 | 1989-05-30 | Raychem Corporation | Aromatic polymer compositions |
| US4680377A (en) | 1985-01-15 | 1987-07-14 | Matsushita Electric Works, Ltd. | Polyimide prepolymer composition from unsaturated bisimide and diamine containing unreacted reactants |
| US4654410A (en) | 1985-01-22 | 1987-03-31 | Asahi Glass Company, Ltd. | Aromatic polysulfone ether/polythioether sulfone copolymer and process for its preparation |
| US4657973A (en) | 1985-01-23 | 1987-04-14 | Idemitsu Petrochemical Co., Ltd. | Polycarbonate resin composition |
| US4801677A (en) | 1985-01-25 | 1989-01-31 | Bayer Aktiengesellschaft | Thermotropic aromatic polyester with high thermal stability |
| US4608426A (en) | 1985-01-29 | 1986-08-26 | American Cyanamid Company | Bis-maleimide resin composition |
| US4577034A (en) | 1985-02-14 | 1986-03-18 | The Dow Chemical Company | Autoxidation of bis(ortho dialkyl-phenoxy)benzophenones |
| US4816556A (en) | 1985-02-22 | 1989-03-28 | E. I. Du Pont De Nemours And Company | Ordered polyetherketones |
| US4719283A (en) | 1985-03-06 | 1988-01-12 | Huels Aktiengesellschaft | Preparation of polyimide from bis-imide and benzocyclobutene |
| US4699975A (en) | 1985-03-07 | 1987-10-13 | Kureha Kagaku Kogyo Kabushiki Kaisha | Process for producing readily crystallizable arylenesulfide resin composition containing organic acid or anhydride |
| US4874834A (en) | 1985-03-10 | 1989-10-17 | Nitto Electric Industrial Co., Ltd. | Colorless transparent polyimide shaped article and process for producing the same |
| US4876330A (en) | 1985-03-10 | 1989-10-24 | Nitto Electric Industrial Co., Ltd. | Colorless transparent polyimide shaped article and process for producing the same |
| US4851287A (en) | 1985-03-11 | 1989-07-25 | Hartsing Jr Tyler F | Laminate comprising three sheets of a thermoplastic resin |
| US4563514A (en) | 1985-03-26 | 1986-01-07 | General Electric Company | Curable polysilarylene-polysiloxane copolymers |
| US4727118A (en) | 1985-04-12 | 1988-02-23 | Ntn-Rulon Industries Co., Ltd. | Polyether imide resin composition containing vulcanizable organopolysiloxane |
| US4680326A (en) | 1985-04-16 | 1987-07-14 | Phillips Petroleum Company | Poly(arylene sulfide) composition with improved insulation resistance and cracking resistance |
| US4778859A (en) | 1985-04-27 | 1988-10-18 | Asahi Kasei Kogyo Kabushiki Kaisha | Tetramine derived polyimide with pendant unsaturation, and various photosensitive compositions therefrom |
| US4861915A (en) | 1985-05-02 | 1989-08-29 | Amoco Corporation | Poly (aryl ether ketone) block copolymers |
| US4891167A (en) | 1985-05-02 | 1990-01-02 | Amoco Corporation | Block polymers containing a poly(aryl ether ketone) and methods for their production |
| US4897527A (en) | 1985-05-09 | 1990-01-30 | John Corby Limited | Press for fabrics |
| US4739075A (en) | 1985-06-01 | 1988-04-19 | Director-General Of Agency Of Industrial Science & Technology | Reactive end-capping agent, addition-curable quinoxaline resin oligomer and intermediate material for fiber-reinforced composite |
| US4574148A (en) | 1985-06-21 | 1986-03-04 | Eastman Kodak Company | Polyester resins capable of forming containers having improved gas barrier properties |
| US4611022A (en) | 1985-06-24 | 1986-09-09 | The Dow Chemical Company | Polymer modified polyphenol compositions and thermosettable resins thereof |
| US4614767A (en) | 1985-07-29 | 1986-09-30 | Atlantic Richfield Company | Moldable polyester-sulfone thermoplastic resin |
| USH183H (en) | 1985-08-19 | 1987-01-06 | The United States Of America As Represented By The Secretary Of The Air Force | Conducting poly (p-xylylidenes) |
| US4663399A (en) | 1985-08-22 | 1987-05-05 | General Electric Company | Polycarbonate-polyether block copolymers, polymer blends containing same and intermediates for the production thereof |
| US4764427A (en) | 1985-08-22 | 1988-08-16 | Teijin Limited | Fiber having thermoplastic resin coating |
| US4769426A (en) | 1985-08-27 | 1988-09-06 | Kureha Kagaku Kogyo Kabushiki Kaisha | Process for preparation of polyarylene thioethers having high melt crystallization temperature |
| US4755584A (en) | 1985-09-02 | 1988-07-05 | Idemitsu Kosan Company Limited | Polyester carbonate containing imido groups and method of preparing the same |
| US5594089A (en) * | 1985-09-05 | 1997-01-14 | The Boeing Company | Heterocycle or heterocycle sulfone oligomers with multiple chemically functional end caps |
| US4845150A (en) | 1985-09-26 | 1989-07-04 | Foster-Miller Inc. | Interpenetrated polymer films |
| US4847333A (en) | 1985-09-30 | 1989-07-11 | The Boeing Company | Blended polyamide oligomers |
| US5151487A (en) | 1985-09-30 | 1992-09-29 | The Boeing Company | Method of preparing a crosslinking oligomer |
| US5087701A (en) | 1985-09-30 | 1992-02-11 | The Boeing Company | Phthalimide acid halides |
| US4935523A (en) | 1985-09-30 | 1990-06-19 | The Boeing Company | Crosslinking imidophenylamines |
| US4624888A (en) | 1985-09-30 | 1986-11-25 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Acetylene (ethynyl) terminated polyimide siloxane and process for preparation thereof |
| US4985568A (en) | 1985-09-30 | 1991-01-15 | The Boeing Company | Method of making crosslinking imidophenylamines |
| US4812518A (en) | 1985-10-03 | 1989-03-14 | Byk-Chemie Gmbh | Polyester group containing polysiloxanes for lacquers and molding compositions |
| US4611048A (en) | 1985-10-04 | 1986-09-09 | General Electric Company | Hydroxy terminated polyetherimide oligomers |
| US4657977A (en) | 1985-10-04 | 1987-04-14 | General Electric Company | Poly(etherimide-carbonate) block copolymers and polymer blends containing same |
| US4871475A (en) | 1985-10-07 | 1989-10-03 | The Boeing Company | Polysulfone and polyethersulfone oligomers |
| US4778898A (en) | 1985-10-25 | 1988-10-18 | Ciba-Geigy Corporation | Trisimides of allyl-substituted or methallyl-substituted bicyclo[2.2.1]hept-5-ene-2,3-dicarboximides and the use thereof |
| US4798686A (en) | 1985-11-29 | 1989-01-17 | Bayer Aktiengesellschaft | Organic polymers with electrical properties |
| US4663425A (en) | 1985-12-10 | 1987-05-05 | The United States Of America As Represented By The Secretary Of The Air Force | Ethynyl-containing aromatic polyamide resin |
| US4766180A (en) | 1985-12-23 | 1988-08-23 | Shell Oil Company | Crosslinked elastomers containing benzocyclobutene derivatives as crosslinking agents |
| US4826927A (en) | 1986-01-13 | 1989-05-02 | Ciba-Geigy Corporation | Mixtures containing polymides alkenyphenols and either epoxide group-free adducts or another polyimide |
| US4757118A (en) | 1986-01-23 | 1988-07-12 | Allied Corporation | Thermosettable modified phenolic polyester imide resins |
| US4777208A (en) | 1986-01-27 | 1988-10-11 | The Dow Chemical Company | Polymer modified unsaturated polyester for polyesteramide resin polymer concrete |
| US4694064A (en) | 1986-02-28 | 1987-09-15 | The Dow Chemical Company | Rod-shaped dendrimer |
| US4891408A (en) | 1986-03-03 | 1990-01-02 | Amoco Corporation | Epoxy resins based on tetraglycidyl diamines |
| US4695610A (en) | 1986-03-18 | 1987-09-22 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Semi-2-interpenetrating networks of high temperature systems |
| US4690972A (en) | 1986-04-04 | 1987-09-01 | Phillips Petroleum Company | Method of producing poly(arylene sulfide) compositions and articles made therefrom |
| US4703081A (en) | 1986-04-08 | 1987-10-27 | Phillips Petroleum Company | Poly(arylene sulfide) resin ternary blends |
| US4709006A (en) | 1986-04-16 | 1987-11-24 | The United States Of America As Represented By The Secretary Of The Air Force | Polybenzobisthiazole from aryloxy naphthalic diacid |
| US4845185A (en) | 1986-04-22 | 1989-07-04 | Nippon Steel Corporation | Soluble copolyimide from 9,9-bis (4-amino phenyl) fluorene |
| US4748227A (en) | 1986-04-25 | 1988-05-31 | Amoco Corporation | Process for preparing poly(aryl ether)ketones using a sodium carbonate/organic acid salt catalyst |
| US4839378A (en) | 1986-04-30 | 1989-06-13 | Meiji Seika Kabushiki Kaisha | Fluorophthalimides and agricultural and horticultural fungicides containing them |
| US4816503A (en) | 1986-06-04 | 1989-03-28 | The Dow Chemical Company | Polymer concrete having high bond strength and long working time |
| US4786669A (en) | 1986-06-09 | 1988-11-22 | Shell Oil Company | Lightly cross-linked resin materials |
| US4708983A (en) | 1986-06-18 | 1987-11-24 | Phillips Petroleum Company | Arylene sulfide polymers of improved impact strength |
| US4709008A (en) | 1986-06-30 | 1987-11-24 | Interez, Inc. | Blend of tris (cyanatophenyl) alkane and bis(cyanatophenyl) alkane |
| US4902440A (en) | 1986-07-21 | 1990-02-20 | The Yokohama Rubber Co., Ltd. | UV-curable resin compositions |
| US4798882A (en) | 1986-07-23 | 1989-01-17 | Enichem Tecnoresine S.P.A. | Branched polycarbonate from methylene-bis-naphthalene compound |
| US4730030A (en) | 1986-07-25 | 1988-03-08 | The Dow Chemical Company | Arylcyclobutane/dienophile copolymer |
| US4757150A (en) | 1986-07-31 | 1988-07-12 | General Electric Company | Polyetherimide bisphenol compositions |
| US4786685A (en) | 1986-08-01 | 1988-11-22 | Nippon Gohsei Kagaku Kogyo Kabushiki Kaisha | Thermoplastic resin composition |
| US4757128A (en) | 1986-08-01 | 1988-07-12 | Massachusetts Institute Of Technology | High molecular weight polyanhydride and preparation thereof |
| US4868270A (en) | 1986-08-04 | 1989-09-19 | The Boeing Company | Heterocycle sulfone oligomers and blends |
| US5338532A (en) | 1986-08-18 | 1994-08-16 | The Dow Chemical Company | Starburst conjugates |
| US4801676A (en) | 1986-08-28 | 1989-01-31 | Basf Aktiengesellschaft | Fully aromatic thermotropic polyester imide |
| US4716212A (en) | 1986-09-05 | 1987-12-29 | Phillips Petroleum Company | Preparation of a high molecular weight poly(arylene sulfide ketone) |
| US4740584A (en) | 1986-09-08 | 1988-04-26 | Interez, Inc. | Blend of dicyanate esters of dihydric phenols |
| US4808717A (en) | 1986-09-17 | 1989-02-28 | Sumitomo Chemical Company, Limited | Thermosetting resin composition |
| US4798685A (en) | 1986-10-20 | 1989-01-17 | Lockheed Corporation | Production of base-type conducting polymers |
| US4759986A (en) | 1986-10-23 | 1988-07-26 | Hoechst Celanese Corporation | Electrically conductive polybenzimidazole fibrous material |
| US4766197A (en) | 1986-10-28 | 1988-08-23 | Amoco Corporation | Modified poly(aryl ether ketones) derived from biphenol |
| US4845167A (en) | 1986-10-29 | 1989-07-04 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Condensation polyimides containing 1,1,1-triaryl-2,2,2-trifluoroethane structures |
| EP0266662B1 (en) | 1986-11-06 | 1992-06-10 | Bayer Ag | Process for the preparation of poly(arylensulphides) with lower crystallization temperature |
| US4774282A (en) | 1986-11-20 | 1988-09-27 | Amoco Corporation | Thermoset composition comprising aromatic cyanate ester, allyl ethers of bisphenol and bismaleimide |
| US4916235A (en) | 1986-11-26 | 1990-04-10 | University Of Dayton | Resin systems derived from benzocyclobutene-maleimide compounds |
| US4740564A (en) | 1986-12-02 | 1988-04-26 | General Electric Company | Thermoplastic polyetherimide esters |
| US4740563A (en) | 1986-12-02 | 1988-04-26 | General Electric Company | Crosslinked polyetherimide ester polymers |
| US4902773A (en) | 1986-12-03 | 1990-02-20 | Nitrokemia Ipartelepek | Method of preparation of unsaturated polyester resins from maleic acid/anhydride |
| US4816526A (en) | 1986-12-08 | 1989-03-28 | Ici Americas Inc. | Polyimide/polyetherimide resin blends |
| US4778830A (en) | 1986-12-13 | 1988-10-18 | Basf Aktiengesellschaft | Polyester polyols liquid at room temperature and their preparation and use in preparing plastics by polyisocyanate addition polymerization |
| US4769436A (en) | 1986-12-20 | 1988-09-06 | Bayer Aktiengesellschaft | Polyester polyalcohols containing amino and amide groups and poly (urea) urethane produced therefrom |
| EP0277476A3 (en) | 1987-01-05 | 1991-09-25 | The Boeing Company | Crosslinkable oligomers |
| US4973662A (en) | 1987-01-13 | 1990-11-27 | Director General Of Agency Of Industrial Science And Technology | Endcapping agent for polyimide, polyimide varnish, and intermediate material for fiber-reinforced composite material comprising same |
| US4958031A (en) | 1987-02-20 | 1990-09-18 | The Boeing Company | Crosslinking nitromonomers |
| EP0283636A2 (en) | 1987-02-20 | 1988-09-28 | The Boeing Company | Polyetherimide oligomers and blends |
| US5268519A (en) | 1987-02-20 | 1993-12-07 | The Boeing Company | Lightly crosslinked etherimide oligomers |
| US5175304A (en) | 1987-02-20 | 1992-12-29 | The Boeing Company | Halo- or nitro-intermediates useful for synthesizing etherimides |
| US5144000A (en) | 1987-02-20 | 1992-09-01 | The Boeing Company | Method for forming crosslinking polyetherimide oligomers |
| US4990624A (en) | 1987-02-20 | 1991-02-05 | The Boeing Company | Intermediates anhydrides useful for synthesizing etherimides |
| US4851495A (en) | 1987-02-20 | 1989-07-25 | The Boeing Company | Polyetherimide oligomer |
| US4981922A (en) | 1987-02-20 | 1991-01-01 | The Boeing Company | Blended etherimide oligomers |
| US4829138A (en) | 1987-02-24 | 1989-05-09 | Rhone-Poulenc Chimie | Novel heat-stable siloxane/bismaleimido polymers |
| US4861855A (en) | 1987-03-31 | 1989-08-29 | Amoco Corporation | Polyamide-imide compositions from bisphenoxyphenyl diamine tricarboxylic anhydride and tetracarboxylic dianhydride |
| US4814417A (en) | 1987-04-10 | 1989-03-21 | Chisso Corporation | Process for producing a liquid crystalline polyester polymer of a cyclohexanedicarboxylic acid and an aromatic diol with pyridine/thionyl chloride catalyst |
| US4812552A (en) | 1987-04-15 | 1989-03-14 | Phillips Petroleum Company | Process for preparing poly(arylene sulfide ketone) with water addition |
| EP0317754A3 (en) | 1987-05-04 | 1990-05-16 | The Boeing Company | Polymide oligomers and blends |
| US5011905A (en) | 1987-05-04 | 1991-04-30 | The Boeing Company | Polyimide oligomers and blends |
| EP0289798B1 (en) | 1987-05-04 | 1996-03-13 | The Boeing Company | Crosslinking imidophenylamines |
| US5116935A (en) | 1987-05-04 | 1992-05-26 | The Boeing Company | Polyimide oligomers and blends and method of curing |
| US4895924A (en) | 1987-05-15 | 1990-01-23 | Kureha Kagaku Kogyo K. K. | Poly(arylene thioether-ketone) fibers and production process thereof |
| US4895892A (en) | 1987-05-15 | 1990-01-23 | Kureha Kagaku Kogyo K.K. | Melt-stable poly(arylene thioether-ketone) compositions |
| EP0294555A3 (en) | 1987-05-18 | 1990-07-11 | The Boeing Company | Polyamide oligomers |
| US4876328A (en) | 1987-05-18 | 1989-10-24 | The Boeing Company | Polyamide composition |
| EP0292677A3 (en) | 1987-05-18 | 1990-05-16 | The Boeing Company | Blended polyamide oligomers |
| US4806407A (en) | 1987-05-19 | 1989-02-21 | Shakespeare Company | Monofilaments, fabrics thereof and related process |
| EP0292434B1 (en) | 1987-05-22 | 1992-07-08 | Ciba-Geigy Ag | Unsaturated bisimides and their polymers |
| US4769424A (en) | 1987-06-01 | 1988-09-06 | General Electric Company | Polyarylene sulfide block copolymers, method for making, and use |
| US5109105A (en) | 1987-06-12 | 1992-04-28 | The Boeing Company | Polyamide oligomers |
| US4902335A (en) | 1987-06-22 | 1990-02-20 | Nihon Tokushu Noyaku Seizo K.K. | Benzo-fused cyclic compounds |
| US4919992A (en) | 1987-07-02 | 1990-04-24 | Imperial Chemical Industries Plc | Process for making microporous products and the products thereof |
| US4931531A (en) | 1987-07-02 | 1990-06-05 | Mitsui Toatsu Chemicals, Incorporated | Polyimide and high-temperature adhesive thereof |
| US4851494A (en) | 1987-07-24 | 1989-07-25 | Ciba-Geigy Corporation | Crosslinkable polyether-amide |
| US4816527A (en) | 1987-08-20 | 1989-03-28 | General Electric Company | Polycarbonate-siloxane polyetherimide copolymer blends |
| US4845278A (en) | 1987-09-03 | 1989-07-04 | Semih Erhan | Crosslinkable aramids |
| US5155206A (en) | 1987-09-03 | 1992-10-13 | The Boeing Company | Crosslinkable polyamideimide oligomers and a method of preparation |
| EP0305882A3 (en) | 1987-09-03 | 1990-07-11 | The Boeing Company | Amideimide oligomers & blends |
| US5239046A (en) | 1987-09-03 | 1993-08-24 | The Boeing Company | Amideimide sizing for carbon fiber |
| US5104967A (en) | 1987-09-03 | 1992-04-14 | The Boeing Company | Amideimide oligomers and blends |
| US5216117A (en) | 1987-09-03 | 1993-06-01 | The Boeing Company | Amideimide blends |
| US4814416A (en) | 1987-09-04 | 1989-03-21 | Huels Aktiengesellschaft | Molding compounds comprised of a thermotropic aromatic polyester |
| US4827000A (en) | 1987-09-21 | 1989-05-02 | Occidental Chemical Corporation | Process for the preparation of phenoxy phthalic anhydrides |
| EP0309649A2 (en) | 1987-11-03 | 1989-04-05 | The Boeing Company | High performance heterocycle oligomers and blends |
| US4786713A (en) | 1987-11-06 | 1988-11-22 | Eastman Kodak Company | Copoly(arylene sulfidex-disulfide) |
| EP0311735A3 (en) | 1987-11-17 | 1991-04-10 | The Boeing Company | Heterocycle sulfone oligomers and blends |
| US4931540A (en) | 1987-11-24 | 1990-06-05 | Hoechst Celanese Corporation | Polymers prepared from 4,4'-bis[2-(3,4-(dicarboxyphenyl)hexafluoroisopropyl] diphenyl ether dianhydride |
| US4814421A (en) | 1987-12-04 | 1989-03-21 | General Electric Company | Hydroxy terminated polycarbonates |
| US4851496A (en) | 1987-12-12 | 1989-07-25 | Huels Aktiengesellschaft | Molding material comprising a thermotropic, aromatic polyester from bis(carboxy phenoxy)diphenyl sulfone |
| US4812588A (en) | 1987-12-14 | 1989-03-14 | The Dow Chemical Company | Polyorganosiloxane-bridged bisbenzocyclobutene monomers |
| EP0323540A1 (en) | 1988-01-08 | 1989-07-12 | The Boeing Company | Conductive, thermally stable oligomers |
| US4923752A (en) | 1988-01-19 | 1990-05-08 | E. I. Du Pont De Nemours & Co. | Sizing for carbon fiber |
| US4820770A (en) | 1988-01-25 | 1989-04-11 | Ethyl Corporation | Polyamide composition |
| EP0334778B1 (en) | 1988-03-24 | 1992-04-15 | Ciba-Geigy Ag | Aromatic polyamide-imides functionalised with alkenyloxy groups, a method for their preparation and their use to prepare crosslinked polymers |
| EP0336856B1 (en) | 1988-03-24 | 1993-03-03 | Ciba-Geigy Ag | With maleimide groups functionalized aromatic polyamideimides, process for their preparation and their use for preparing cross-linked polymers |
| US4927899A (en) | 1988-03-24 | 1990-05-22 | Rhone-Poulenc Chimie | Alkenyloxylated aromatic polyamidoimides |
| US4927900A (en) | 1988-03-24 | 1990-05-22 | Rhone-Poulenc Chimie | Aromatic polyamidoimides having maleimido end groups |
| US5115087A (en) | 1988-03-29 | 1992-05-19 | The Boeing Company | Coreactive imido oligomer blends |
| US4851280A (en) | 1988-04-01 | 1989-07-25 | E. I. Du Pont De Nemours And Company | Composite tooling for composites manufacture |
| US4812534A (en) | 1988-04-04 | 1989-03-14 | Eastman Kodak Company | Impact toughened polyester nylon blends |
| US4851505A (en) | 1988-04-13 | 1989-07-25 | E. I. Du Pont De Nemours And Company | Highly soluble aromatic polyimides |
| US4876325A (en) | 1988-04-28 | 1989-10-24 | Allied-Signal Inc. | Polyimide resin from bis-imide, polyphenol and dicyandiamide |
| US5075537A (en) | 1988-06-03 | 1991-12-24 | Warme-Undeletrotechn | Controlling and monitoring circuit for electrical seat heating means, especially of automotive vehicles |
| US4861882A (en) | 1988-07-14 | 1989-08-29 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Ethynyl terminated imidothioethers and resins therefrom |
| US4996101A (en) | 1988-08-08 | 1991-02-26 | Lockheed Corporation | Processible polyimide blends |
| US4861924A (en) | 1988-08-25 | 1989-08-29 | Jet Research Center, Inc. | 1,3,5-trinitro-2,4,6-tripicrylbenzene |
| US5112936A (en) | 1988-09-06 | 1992-05-12 | Idemitsu Petrochemical Co., Ltd. | Branched chain stopped polycarbonate and process for production |
| US4902769A (en) | 1988-09-23 | 1990-02-20 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Low dielectric fluorinated poly(phenylene ether ketone) film and coating |
| US4916210A (en) | 1988-10-20 | 1990-04-10 | Shell Oil Company | Resin from alpha, alpha', alpha"-tris(4-cyanatophenyl)-1,3,5-triisopropylbenzene |
| US5111026A (en) | 1989-04-01 | 1992-05-05 | Ma Chun Ho | Control device and method for an electric rice cooker |
| EP0405128A3 (en) | 1989-05-18 | 1992-01-08 | The Boeing Company | Polyimide oligomers and blends and method of curing |
| US5086154A (en) | 1989-07-11 | 1992-02-04 | Rhone-Poulenc Chimie | Linear aromatic poly(amideimide)s having latent maleimide endgroups |
| US5003035A (en) | 1989-07-13 | 1991-03-26 | The United States Of America As Represented By The Secretary Of The Air Force | Crosslinkable rigid-rod benzobisazole copolymer |
| US5066776A (en) | 1989-08-24 | 1991-11-19 | Bayer Aktiengesellschaft | Process for the manufacture of polyarylene sulphides |
| EP0418406A1 (en) | 1989-09-19 | 1991-03-27 | The Boeing Company | Coreactive oligomer blends |
| US5254605A (en) | 1991-11-30 | 1993-10-19 | Cheil Industries, Inc. | Imide epoxy resin composition for sealing semiconductor elements |
Non-Patent Citations (30)
| Title |
|---|
| Bartolotta, Predicting Fatigue Lives of Metal-Matrix/Fiber Composites NASA Tech Briefs pp. 28, 30, Apr. 1994. |
| Bryant, et al., Phenylethynyl End-Capping Reagents and Reactive Diluents NASA Tech Briefs pp. 36-37, Apr. 1994. |
| Bryant, et al., Synthesis and Properties of Phenylethynyl-Terminated Polyimides Polymer PrePrints, vol. 34, No. 1, 566-567, Mar. 1993. |
| Buckley, et al., Processable Polyimides for High Temperature Applications 36th International SAMPE Symposium pp. 1172-1181, Apr. 1991. |
| Crivello, et al. Polyimidothioether-Polysulfide Block Polymers Polymer Sci., Polymer Chem. Ed., vol. 3, pp. 1819-1842, 1975. |
| Edwards, et al. Constituents of the Higher Fungi. Part XIII.1 2-Arly-3-methoxymaleic Anhydrides from Pulvinic Acid Derivatives. A Convenient Method for Determination of Structure of Fungaland Lichen Pulvinic Acid Derivatives, Journal of The Chemical Society pp. 1538-1542, 1973. |
| Elsenbaumer et al., Highly Conductive Meta Derivatives of Poly(phenylene Sulfide) J. Polymer Sci: Polymer Phys. Ed., vol. 20, 1781-1787. 1982. |
| Frazer, High Temperature Resistant Polymers Interscience Publishers, John Wiley & Sons, Inc., 139-213, 1968. |
| Heidemann, "Oligomers" Encyclopedia of Polymer Science and Technology vol. 9 Molding to Petroleum Resins 485-506, 1968. |
| Jensen, et al., Phenylethynyl-Terminated Ploy (Arlene Ethers) NASA Tech Briefs p. 37, Apr. 1994. |
| Kwiatkowski, et al., Thermosetting Diphenyl Sulfone-Baed Malcimides Journal of Polymer Science, vol. 13, pp. 961-972, 1975. |
| Lyle, et al., Polyarylene Ethers: Maleimides, Nadimides and Blends The Interdisciplinary Symposium on Recent Advances in Polyimides and Other High Performance Polymers, San Diego, California pp. K-1-K-7, Jan. 1990. |
| Mittal (ed), Polyimides Plenum Press, NY, vol. 1 & 2 (selected pages), |
| Patel et al,, Poly-Schiff Bases, I. Preparation of Poly-Schiff Bases from 4,4′-Diacetyl Diphenyl Ether (DDE) with Various Diamines J. of Polymer Sci: Polymer Chem. Ed., vol. 20, 1985-1992, 1982. |
| Patel et al,, Poly-Schiff Bases, I. Preparation of Poly-Schiff Bases from 4,4'-Diacetyl Diphenyl Ether (DDE) with Various Diamines J. of Polymer Sci: Polymer Chem. Ed., vol. 20, 1985-1992, 1982. |
| Radlmann, et al., New Synthesis of Poly(ether Ketones). (44195h) Chem. Abstracts vol. 72, 1970, p. 44187. 1970. |
| Roberts, et al., Effect of Solution Concentration and Aging Conditions on PMR-15 Resin SAMPE Journal, pp. 24-28, 213, Mar./Apr. 1986. |
| Second-generation polyimide raises continuous-use temperatures Advanced Composites, May/Jun. 1988. |
| Serafini et al., Thermally Stable Polyimides from Solutions of Monomeric Reactants Journal of Applied Polymer Science, vol. 16, pp. 905-915, 1972. |
| Serafini, et al., A Review of Processable High Temperature Resistant Addition-type Laminating Resins Mittal (ed) Polyimides-Synthesis, Characterization and Applications, Plenum Press, NY, vol. 1, pp. 89-95, 1973. |
| Southcott, et al., "The Development of Processable, Fully Imidized, Polyimides for High-Temperature Applications" High Perform. Polym. 6, pp. 1-12, Printed in UK, 1994. |
| Spillman et al., Copolymers of Poly(Para-Phenylene Terephthalamide) Containing a Thermally Activated Cross-Linking Agent PMSE vol. 68, Spring Meetings 139-140, 1993. |
| St. Clair et al., The Development of Aerospace Polyimide Adhesives Mittal (ed), Polyimides-Synthesis Characterization and Applications, Plenum Press, NY, vol. 2, pp. 977-1041, 1973. |
| St. Clair, et al., Additives Lower Pickup of Moisture by Polyimides NASA Tech Briefs, 80-81, Apr., 1989. |
| Stenson, Polycyanurates Find Applications; Their Chemistry Remains Puzzling Science/Technology, 208 ACS National Meeting, Washington, D,C., C&EN Northeast News Bureau 30-31, Sep. 1994. |
| Stoakley, et al., Low-Dielectric-Constant Polyimide/Glass Composites NASA Tech. Briefs p. 24, Apr. 1994. |
| Vannucci, et al., Improved PMR Polyimides for Heat-Stable Laminates NASA Tech Briefs pp. 30-31, Apr. 1994. |
| Vanucci et al., 700° F. Properties of Autoclave Cured PMR-II Composites NASA Tech. Memo 100923. Sep., 1988. |
| Vanucci, PMR Polyimide Compositions for Improved Peformance at 371° C. NTIS N87-16071, Apr., 1987. |
| Walton, A New Conjugated Network Polymer as an Electrical Conductor and Thermally Stable Plastic Am. Chem., Soc. Org. Coat Plast. Chem., vol. 42, 595-599, 1980. |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8822628B2 (en) | 2012-09-24 | 2014-09-02 | Ticona Llc | Crosslinkable liquid crystalline polymer |
| US8853342B2 (en) | 2012-09-24 | 2014-10-07 | Ticona Llc | Crosslinkable liquid crystalline polymer |
| US9145519B2 (en) | 2012-09-24 | 2015-09-29 | Ticona Llc | Crosslinkable aromatic polyester |
Also Published As
| Publication number | Publication date |
|---|---|
| US5739256A (en) | 1998-04-14 |
| US5705598A (en) | 1998-01-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA1319214C (en) | Blended polyamide oligomers | |
| US4876328A (en) | Polyamide composition | |
| CA1336098C (en) | Heterocycle sulfone oligomers and blends | |
| EP0305882A2 (en) | Amideimide oligomers & blends | |
| US4965336A (en) | High performance heterocycle oligomers and blends | |
| US5082905A (en) | Blended heterocycles | |
| US5968640A (en) | Conductive, thermally stable oligomers | |
| US5512676A (en) | Extended amideimide hub for multidimensional oligomers | |
| US5109105A (en) | Polyamide oligomers | |
| US5216117A (en) | Amideimide blends | |
| US6569954B1 (en) | Composites from blends of advanced oligomers | |
| EP0289798A2 (en) | Crosslinking imidophenylamines | |
| US5126410A (en) | Heterocycle oligomers | |
| US5151487A (en) | Method of preparing a crosslinking oligomer | |
| US5120819A (en) | High performance heterocycles | |
| US5066541A (en) | Heterocycle oligomer blends | |
| US5198526A (en) | Heterocycle oligomers with multidimensional morphology | |
| US5087701A (en) | Phthalimide acid halides | |
| US6583255B1 (en) | Polyester oligomer | |
| US5594089A (en) | Heterocycle or heterocycle sulfone oligomers with multiple chemically functional end caps | |
| US4985568A (en) | Method of making crosslinking imidophenylamines | |
| EP0289695A2 (en) | Polyimide oligomers and blends | |
| US5367083A (en) | Extended acid halide capping monomers | |
| EP0323540A1 (en) | Conductive, thermally stable oligomers | |
| US5817738A (en) | Conductive, multidimensional oligomers and blends |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: CENTER FOR ADVANCE TECHNOLOGY & INNOVATION OF RACI Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:BOEING COMPANY, THE;REEL/FRAME:015552/0610 Effective date: 20040702 |
|
| REMI | Maintenance fee reminder mailed | ||
| LAPS | Lapse for failure to pay maintenance fees | ||
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20070624 |