WO1999010427A1 - Rheology modification of polymers prepared using metallocenes - Google Patents
Rheology modification of polymers prepared using metallocenes Download PDFInfo
- Publication number
- WO1999010427A1 WO1999010427A1 PCT/US1998/017685 US9817685W WO9910427A1 WO 1999010427 A1 WO1999010427 A1 WO 1999010427A1 US 9817685 W US9817685 W US 9817685W WO 9910427 A1 WO9910427 A1 WO 9910427A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polymer
- sulfonyl azide
- ethylene
- polymers
- poly
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G81/00—Macromolecular compounds obtained by interreacting polymers in the absence of monomers, e.g. block polymers
- C08G81/02—Macromolecular compounds obtained by interreacting polymers in the absence of monomers, e.g. block polymers at least one of the polymers being obtained by reactions involving only carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/36—Sulfur-, selenium-, or tellurium-containing compounds
- C08K5/43—Compounds containing sulfur bound to nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
- C08L23/08—Copolymers of ethene
- C08L23/0807—Copolymers of ethene with unsaturated hydrocarbons only containing more than three carbon atoms
- C08L23/0815—Copolymers of ethene with aliphatic 1-olefins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/22—Mixtures comprising a continuous polymer matrix in which are dispersed crosslinked particles of another polymer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
- C08L23/06—Polyethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
- C08L23/08—Copolymers of ethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
- C08L23/08—Copolymers of ethene
- C08L23/0807—Copolymers of ethene with unsaturated hydrocarbons only containing more than three carbon atoms
- C08L23/0838—Copolymers of ethene with aromatic monomers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/04—Homopolymers or copolymers of ethene
- C08L23/08—Copolymers of ethene
- C08L23/0846—Copolymers of ethene with unsaturated hydrocarbons containing other atoms than carbon or hydrogen atoms
- C08L23/0853—Vinylacetate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/16—Elastomeric ethene-propene or ethene-propene-diene copolymers, e.g. EPR and EPDM rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2312/00—Crosslinking
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2314/00—Polymer mixtures characterised by way of preparation
- C08L2314/06—Metallocene or single site catalysts
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2666/00—Composition of polymers characterized by a further compound in the blend, being organic macromolecular compounds, natural resins, waxes or and bituminous materials, non-macromolecular organic substances, inorganic substances or characterized by their function in the composition
- C08L2666/02—Organic macromolecular compounds, natural resins, waxes or and bituminous materials
- C08L2666/04—Macromolecular compounds according to groups C08L7/00 - C08L49/00, or C08L55/00 - C08L57/00; Derivatives thereof
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/13—Hollow or container type article [e.g., tube, vase, etc.]
- Y10T428/1334—Nonself-supporting tubular film or bag [e.g., pouch, envelope, packet, etc.]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/13—Hollow or container type article [e.g., tube, vase, etc.]
- Y10T428/1352—Polymer or resin containing [i.e., natural or synthetic]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/13—Hollow or container type article [e.g., tube, vase, etc.]
- Y10T428/1352—Polymer or resin containing [i.e., natural or synthetic]
- Y10T428/1386—Natural or synthetic rubber or rubber-like compound containing
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/13—Hollow or container type article [e.g., tube, vase, etc.]
- Y10T428/1352—Polymer or resin containing [i.e., natural or synthetic]
- Y10T428/139—Open-ended, self-supporting conduit, cylinder, or tube-type article
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31855—Of addition polymer from unsaturated monomers
- Y10T428/31909—Next to second addition polymer from unsaturated monomers
- Y10T428/31913—Monoolefin polymer
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31855—Of addition polymer from unsaturated monomers
- Y10T428/31909—Next to second addition polymer from unsaturated monomers
- Y10T428/31913—Monoolefin polymer
- Y10T428/31917—Next to polyene polymer
Definitions
- This invention relates to coupling of polyolefins, more specifically coupling of polyolefins using insertion into carbon hydrogen (C-H) bonds.
- the term "rheology modification” means change in melt viscosity of a polymer as determined by dynamic mechanical spectroscopy.
- the melt strength increases while maintaining the high shear viscosity (that is viscosity measured at a shear of 100 rad/sec by DMS) so that a polymer exhibits more resistance to stretching during elongation of molten polymer at low shear conditions (that is viscosity measured at a shear of 0.1 rad/sec by DMS) and does not sacrifice the output at high shear conditions.
- Polyolefins are frequently rheology modified using nonselective chemistries involving free radicals generated for instance using peroxides or high energy radiation.
- chemistries involving free radical generation at elevated temperatures also degrade the molecular weight, especially in polymers containing tertiary hydrogen such as polystyrene, polypropylene, polyethylene copolymers etc.
- the reaction of polypropylene with peroxides and pentaerythritol triacrylate is reported by Wang et al . , in Journal of
- US 3,058,944; 3,336,268; and 3,530,108 include the reaction of certain poly (sulfonyl azide) compounds with isotactic polypropylene or other polyolefins by nitrene insertion into C-H bonds.
- the product reported in US 3,058,944 is crosslinked.
- the product reported in US 3,530,108 is foamed and cured with cycloalkane-di (sulfonyl azide) of a given formula.
- bridged polymers because polymer chains are "bridged” with sulfonamide bridges .
- the disclosed process includes a mixing step such as milling or mixing of the pol (sulfonyl azide) and polymer in solution or dispersion then a heating step where the temperature is sufficient to decompose the poly (sulfonyl azide) (100°C to 225° depending on the azide decomposition temperature) .
- the starting polypropylene polymer for the claimed process has a molecular weight of at least 275,000.
- Blends taught in US 3,336,268 have up to 25 percent ethylene propylene elastomer.
- United States Patent 3,631,182 taught the use of azido formate for crosslinking polyolefins.
- United States Patent 3,341,418 taught the use of sulfonyl azide and azidoformate compounds to crosslink certain thermoplastics (PP (polypropylene) , PS (polystyrene) , PVC (poly (vinyl chloride)) and their blends with certain rubbers (polyisobutene, EPDM, (ethylene propylene diene rubber) ) .
- PP polypropylene
- PS polystyrene
- PVC poly (vinyl chloride)
- EPDM ethylene propylene diene rubber
- Canadian patent 797,917 family member of NL 6,503,188
- teachings of Canadian patent 797,917 include rheology modification using from 0.001 to 0.075 weight percent poly (sulfonyl azide) to modify homopolymer polyethylene and its blend with polyisobutylene.
- polymers rheology modified rather than crosslinked that is having less than 10 percent gel as determined by xylene extraction specifically by ASTM 2765.
- the starting material is high density polyethylene (density greater than 0.945 g/ml) (hereinafter HDPE of narrow MWD) which polymers advantageously have a combination of good processibility as indicated by higher melt strength at a constant low shear viscosity for example 0.1 rad/sec measured by DMS, and higher toughness, tensile elongation than a high density polyethylene of broader molecular weight distribution treated with sulfonyl azides according to the practice of the prior art using the same equivalents (stoichiometry) of coupling reactant to polymer, and higher toughness than that of the same starting material coupled or rheology modified using the same equivalents of a free radical coupling agent.
- HDPE of narrow MWD high density polyethylene (density greater than 0.945 g/ml)
- the product would have better organoleptics than coupled broader MWD HDPE.
- compositions would have less undesirable odor than the same starting materials coupled or rheology modified using the same chemical equivalents of free radical generating agents.
- a process of the invention would result in more consistent coupling than methods of coupling involving free radicals, that is use of the same reactants, amounts and conditions would result in consistent amounts of coupling or consistent (reproducible) property changes, especially consistent amounts of gel formation.
- a process would be less subject to effects from the presence of oxygen than would a coupling or rheology modification involving agents which generate free radicals.
- medium and lower density polyethylene that is polymers having a density of from 0.94 g/cc to about ⁇ .90 g/cc
- the polymers would desirably show a combination of processability improved over the starting material with retention of toughness, low heat seal initiation temperature, low haze, high gloss or hot tack properties characteristic of the starting material.
- polymers having a narrow molecular weight distribution or formed using single site catalysts especially polyolefins formed using transition metal catalysts other than Ziegler Natta catalysts, particularly where the molecular weight distribution is 3 or less, are surprisingly effectively coupled using poly (sulfonyl azide) coupling agents.
- the resulting coupled polymers have at least one of the desirable properties listed.
- the invention includes a process of preparing a coupled polymer comprising heating an admixture containing (1) at least one polyolefin comprising ethylene and optionally at least one comonomer which is selected from alpha olefins having at least 3 carbon atoms, dienes and combinations thereof said polyolefin having a molecular weight distribution of less than or equal to 3 and (2) a coupling amount at least one poly (sulfonyl azide) to at least the decomposition temperature of the poly (sulfonyl azide) for a period sufficient for decomposition of at least 80 weight percent of the poly (sulfonyl azide) and sufficient to result in a coupled polymer; particularly where the polyolefin is the product of polymerization of ethylene and optionally at least one other alpha-olefin in the presence of a single site catalyst (for example transition metal like V or Cr, metallocene or constrained geometry) .
- a single site catalyst for example transition metal like V or Cr,
- the amount of poly (sulfonyl azide) is preferably from 0.01 to 5 weight percent of the polyolefin.
- the poly (sulfonyl azide) and polyolefin preferably react at a temperature at least the decomposition temperature and greater than 150°C.
- the invention also includes any composition made by the process of the invention and any article made from the composition as well as processes for making the articles, particularly melt processing (especially where the article is formed from a melt of a composition of the invention) , more preferably selected by molding, blow molding or extrusion blow molding the composition.
- the invention also includes the use of a composition of the invention in any of these processes .
- thermoplastic polymer polymerized using a single site catalyst which polymer has at least one C-H bond that can react with azide including homopolymers and copolymers with narrow and broad (including bimodal) comonomer distribution such as copolymers of ethylene with one or more alpha olefin (C3 to C20) , ethylene copolymers with unsaturation (EPDM or EODM, that is ethylene-propylene- diene or ethylene-octene -diene) , or other polymers such as linear high density polyethylene, styrene based block copolymers (SBS, SEBS, SIS , that is styrene/butadiene/styrene, styrene/ethylene / butylene/styrene (hydrogenated SEBS) , styrene/isoprene/styrene ) , substantially random interpolymers of at least one alpha
- bimodal is used to refer to polymers which exhibit two peaks on a graphical representation of data from analysis appropriate to measure the property discussed. For instance, in the case of molecular weight, a gel permeation chromatography (GPC) curve is used to determine the distribution of molecular weights. These distributions are viewed statistically, that is as statistical distributions. Thus where there is one peak, the distribution has one mode and is unimodal . Polymer which exhibit two peaks by this analytical method is referred to as bimodal . Polymers which exhibit two or more are multimodal . In the case of molecular weight distribution, bimodal polymers are often blends of two polymers with different number average molecular weights.
- the blend is optionally an in-reactor blend or a blend formed by passing a first polymer made in a first reactor into a second reactor where the second polymer is produced.
- Bimodal polymers are illustrative of multimodal polymers herein. That is, where polymers having a bimodal characteristic are discussed, the multimodal counterpart is also suitable. Those skilled in the art recognize that the peaks frequently have overlapping areas and that mathematical analysis is sometimes needed to distinguish multimodal curves from broad irregular curves . In this invention, where preferred molecular weight distributions (MWD) are given, those distributions refer to the MWD of at least one component as represented by one peak of the GPC curve. The component is preferably one made using a single site catalyst.
- Preferred polymers for use in the practice of the invention are polymers prepared from ethylene, advantageously ethylene in combination with other monomers polymerizable therewith. Such monomers include alpha olefins and other monomers having at least one double bond.
- the polymer is a polyolefin, optionally a homopolymer, copolymer, or interpolymer .
- the homo or copolymers contain ethylene repeating units .
- the comonomer content is preferably greater than 1 weight percent as determined by 13 C NMR (carbon 13 nuclear magnetic resonance) and more preferably greater than 3 weight percent of any monomer copolymerizable with ethylene, preferably an alpha olefin or cyclic olefin, more preferably such an olefin of less than 20 carbon atoms, most preferably from 2 to 18 carbon atoms.
- the comonomer content is at least one comonomer polymerizable with ethylene, preferably less than 4 comonomers polymerizable with ethylene, more preferably less than 2 such comonomers .
- the ethylene polymers are optionally any interpolymers of ethylene and at least one ⁇ -olefin.
- Preferred ⁇ -olefins are represented by the following formula:
- Suitable ⁇ -olefins for use as comonomers in a solution, gas phase or slurry polymerization process or combinations thereof include 1- propylene, 1-butene, 1-isobutylene, 1-pentene, 1-hexene, 4-methyl-lpentene, 1-heptene and 1-octene, as well as other monomer types such as tetrafluoroethylene, vinyl benzocyclobutane, and cycloalkenes, for example cyclopentene, cyclohexene, cyclooctene, and 5-ethylidene-
- the ⁇ -olefin will be 1- butene, 1-pentene, 4-methyl-1-pentene, 1-hexene, 1- heptene, 1-octene, or ENB, or mixtures thereof. More preferably, the ⁇ -olefin will be 1-hexene, 1-heptene, 1- octene, or mixtures thereof. Most preferably, the ⁇ - olefin is 1-octene.
- the ethylene polymer rheology modified according to this invention is more preferably a SLEP (defined hereinafter) . These interpolymers preferably contain at least 2 weight percent, more preferably at least 5 weight percent, ⁇ -olefin.
- Interpolymers useful in the practice of the invention optionally and in one preferred embodiment include monomers having at least two double bonds which are preferably dienes or trienes .
- Suitable diene and triene comonomers include 7 -methyl- 1, 6-octadiene, 3,7- dimethyl-1, 6-octadiene, 5, 7 -dimethyl- 1, 6-octadiene, 3,7, 11-trimethyl-l, 6, 10-octatriene, 6-methyl-l, 5- heptadiene, 1, 3 -butadiene, 1, 6-heptadiene, 1, 7-octadiene, 1, 8-nonadiene, 1, 9-decadiene, 1, 10-undecadiene, bicyclo [2.2.1] hepta-2-5-diene (norbornadiene) , tetracyclododecene, or mixtures thereof, preferably butadiene, hexadienes, and octadienes,
- Polyolefins are formed by means within the skill in the art.
- the alpha olefin monomers and optionally other addition polymerizable monomers are polymerized under conditions within the skill in the art.
- Such conditions include those processes utilizing metallocene and other single site catalysts such as exemplified by U.S. Patents 4,937,299 (Ewen et al . ) , 5,218,071 (Tsutsui et al . ) , 5,278,272, 5,324,800, 5,084,534, 5,405,922, 4,588,794, 5,204,419 and the processes subsequently discussed in more detail .
- starting material polyolefins are preferably substantially linear ethylene polymers (SLEPs) .
- the substantially linear ethylene polymers (SLEPs) are homogeneous polymers having long chain branching. They are disclosed in U.S. Patent Nos. 5,272,236 and 5,278,272.
- SLEPs are available as polymers made by the InsiteTM Process and Catalyst Technology such as EngageTM polyolefin elastomers (POEs) commercially available from DuPont Dow Elastomers LLC and AffinityTM polyolefin plastomers (POPs) commercially available from The Dow Chemical Company.
- EngageTM polyolefin elastomers POEs
- POPs AffinityTM polyolefin plastomers
- SLEPs can be prepared via the solution, slurry, or gas phase, preferably solution phase, polymerization of ethylene and one or more optional ⁇ -olefin comonomers in the presence of a constrained geometry catalyst, such as the catalysts disclosed in European Patent Application 416,815-A.
- the substantially linear ethylene/ ⁇ -olefin polymers are made by a continuous process using suitable constrained geometry catalysts, preferably constrained geometry catalysts as disclosed in U. S. Patent No.
- Suitable cocatalysts for use herein include but are not limited to, for example, polymeric or oligomeric aluminoxanes, especially methyl aluminoxane, as well as inert, compatible, noncoordinating, ion forming compounds.
- Preferred cocatalysts are inert, noncoordinating, boron compounds.
- continuous process means a process in which reactants are continuously added and product is continuously withdrawn such that an approximation of a steady state (that is substantially constant concentration of reactants and product while carrying out the process) is achieved.
- the polymerization conditions for manufacturing the substantially linear ethylene/ ⁇ - olefin polymers of the present invention are generally those useful in the solution polymerization process, although the application of the present invention is not limited thereto. Slurry and gas phase polymerization processes are also believed to be useful, provided the proper catalysts and polymerization conditions are employed.
- Multiple reactor polymerization processes can also be used in making the substantially linear olefin polymers and copolymers to be rheologically modified according to the present invention, such as those disclosed in USP 3,914,342.
- the multiple reactors can be operated in series or in parallel, with at least one constrained geometry catalyst employed in one of the reactors .
- substantially linear means that, in addition to the short chain branches attributable to homogeneous comonomer incorporation, the ethylene polymer is further characterized as having long chain branches in that the polymer backbone is substituted with an average of 0.01 to 3 long chain branches/1000 carbons.
- Preferred substantially linear polymers for use in the invention are substituted with from 0.01 long chain branch/1000 carbons to 1 long chain branch/1000 carbons, and more preferably from 0.05 long chain branch/1000 carbons to 1 long chain branch/1000 carbons.
- the term “linear” means that the polymer lacks measurable or demonstrable long chain branches, that is, the polymer is substituted with an average of less than 0.01 long chain branch/1000 carbons.
- long chain branching means a chain length longer than the short chain branch that results from the incorporation of the ⁇ -olefin (s) into the polymer backbone.
- Each long chain branch has the same comonomer distribution as the polymer backbone and can be as long as the polymer backbone to which it is attached.
- deGroot and Chum found that the presence of octene does not change the hydrodynamic volume of the polyethylene samples in solution and, as such, one can account for the molecular weight increase attributable to octene short chain branches by knowing the mole percent octene in the sample. By deconvoluting the contribution to molecular weight increase attributable to 1-octene short chain branches, deGroot and Chum showed that GPC-DV may be used to quantify the level of long chain branches in substantially linear ethylene/octene copolymers .
- a critical shear stress at the onset of gross melt fracture as determined by gas extrusion rheometry, of greater than 4 x 10 s dynes/cm 2 or a gas extrusion rheology such that the critical shear rate at onset of surface melt fracture for the SLEP is at least 50 percent greater than the critical shear rate at the onset of surface melt fracture for a linear ethylene polymer, the linear ethylene polymer has an
- I 2 , M Copy/M n and preferably density which are each within ten percent of the SLEP and wherein the respective critical shear rates of the SLEP and the linear ethylene polymer are measured at the same melt temperature using a gas extrusion rheometer, and preferably (d) a single differential scanning calorimetry, DSC, melting peak between -30 and 150 C.
- the 110/12 ratio indicates the degree of long chain branching, that is, the higher the I10/I2 ratio, the more long chain branching in the polymer.
- the I10/I2 ratio of the substantially linear ethylene/ ⁇ - olefin polymers is at least 5.63, preferably at least 7, especially at least 8 or above, and as high as 25.
- the melt index (as measured by ASTM D-1238 (190°/ 2.16 kg weight)) for the substantially linear olefin polymers useful herein is preferably at least 0.1 grams/10 minutes (g/10 min), more preferably at least 0.5 g/10 min and especially at least 1 g/10 min up to preferably 100 g/10 min, more preferably up to 50 g/10 min, and especially up to 20 g/10 min.
- GER gas extrusion rheometer
- GER experiments are generally performed at a temperature of 190 C, at nitrogen pressures between 250 to 5500 psig using a 0.0754 mm diameter, 20:1 L/D die with an entrance angle of 180°.
- the PI is the apparent viscosity (in kpoise) of a material measured by GER at an apparent shear stress of 2.15 x IO 6 dyne/cm 2 .
- the SLEPs for use in the invention includes ethylene interpolymers and have a PI in the range of 0.01 kpoise to 50 kpoise, preferably 15 kpoise or less.
- the SLEPs used herein have a PI less than or equal to 70 percent of the PI of a linear ethylene polymer (either a Ziegler polymerized polymer or a linear uniformly branched polymer as described by Elston in US Patent 3,645,992) having an I 2 , M Clear/M n and preferably density, each within ten percent of the SLEPs.
- a linear ethylene polymer either a Ziegler polymerized polymer or a linear uniformly branched polymer as described by Elston in US Patent 3,645,992
- the rheological behavior of SLEPs can also be characterized by the Dow Rheology Index (DRI) , which expresses a polymer's "normalized relaxation time as the result of long chain branching.”
- DRI Dow Rheology Index
- SLEP Dow Rheology Index
- DRI values range from 0 for polymers which do not have any measurable long chain branching (for example, TafmerTM products available from Mitsui Petrochemical Industries and ExactTM products available from Exxon Chemical Company) to 15 and are independent of melt index.
- DRI provides improved correlations to melt elasticity and high shear flowability relative to correlations of the same attempted with melt flow ratios.
- DRI is preferably at least 0.1, and especially at least 0.5, and most especially at least 0.8.
- DRI can be calculated from the equation:
- n is the power law index of the material
- ⁇ and ⁇ are the measured viscosity and shear rate, respectively.
- Baseline determination of viscosity and shear rate data are obtained using a Rheometric Mechanical Spectrometer (RMS-800) under dynamic sweep mode from 0.1 to 100 radians/second at 190 °C and a Gas Extrusion Rheometer (GER) at extrusion pressures from 1,000 psi to 5,000 psi (6.89 to 34.5 MPa), which corresponds to shear stress from 0.086 to 0.43 MPa, using a 0.0754 mm diameter, 20:1 L/D die at 190 C.
- Specific material determinations can be performed from 140 to 190 °C as required to accommodate melt index variations.
- the loss of extrudate gloss is the point at which the surface roughness of the extrudate can only be detected by a 40X magnification.
- the critical shear rate at the onset of surface melt fracture for the SLEPs is at least 50 percent greater than the critical shear rate at the onset of surface melt fracture of a linear ethylene polymer having essentially the same I 2 and lM n .
- the critical shear stress at the onset of gross melt fracture for the SLEPs is greater than 4 x 10 e dynes/cm 2 .
- the critical shear rate at the onset of surface melt fracture (OSMF) and the onset of gross melt fracture (OGMF) will be used herein based on the changes of surface roughness and configurations of the extrudates extruded by a GER.
- the SLEPs used in the invention are also preferably characterized by a single DSC melting peak.
- the single melting peak is determined using a differential scanning calorimeter standardized with indium and deionized water. The method involves 3-7 mg sample sizes, a "first heat" to 180 °C which is held for 4 minutes, a cool down at 10
- the single melting peak may show, depending on equipment sensitivity, a "shoulder or a "hump" on the low melting side that constitutes less than 12 percent, typically, less than 9 percent, and more typically less than 6 percent of the total heat of fusion of the polymer.
- Such an artifact is observable for other homogeneously branched linear polymers such as ExactTM resins and is discerned on the basis of the slope of the single melting peak varying monotonically through the melting region of the artifact.
- Such an artifact occurs within 34 °C, typically within 27 °C, and more typically within 20 °C of the melting point of the single melting peak.
- the heat of fusion attributable to an artifact can be separately determined by specific integration of its associated area under the heat flow vs. temperature curve .
- the molecular weight distributions of ethylene ⁇ - olefin polymers are determined by gel permeation chromatography (GPC) on a Waters 150°C high temperature chromatographic unit equipped with a differential refractometer and three columns of mixed porosity. The columns are supplied by Polymer Laboratories and are commonly packed with pore sizes of 10 , 10 , 10 and 10 A (IO “4 , 10 "3 , IO “2 and 10 "1 mm).
- the solvent is 1,2,4- trichlorobenzene, from which 0.3 percent by weight solutions of the samples are prepared for injection.
- the flow rate is 1.0 milliliters/minute, unit operating temperature is 140° C and the injection size is 100 microliters .
- the molecular weight determination with respect to the polymer backbone is deduced by using narrow molecular weight distribution polystyrene standards (from Polymer Laboratories) in conjunction with their elution volumes.
- the equivalent polyethylene molecular weights are determined by using appropriate Mark-Houwink coefficients for polyethylene and polystyrene (as described by Williams and Ward in Journal of Polymer Science, Polymer Letters, Vol. 6, p. 621, 1968) to derive the following equation:
- Mw Weight average molecular weight
- the density of the linear or the substantially linear ethylene polymers (as measured in accordance with ASTM D-792) for use in the present invention is generally less than 0.95 g/cm 3 .
- the density is preferably at least
- the density of the polymer is preferably at least 0.855 g/cm 3 , more preferably at least 0.865 g/cm 3 , and even more preferably at least 0.870 g/cm 3 , up to preferably 0.900 g/cm 3 , more preferably 0.885 g/cm 3 , and even more preferably up to 0.880 g/cm 3 .
- preferred density is determined primarily by the modulus of elasticity or flexibility desired in the molded article.
- the density remains substantially constant during rheology modification according to this invention.
- preferred polymers for starting materials in the practice of this invention are slurry high density polyethylene homopolymers preferably made using single site catalysis with a narrow MWD (preferably less than or equal to 3.0 Mw/Mn, more preferably MWD less than 2.5, most preferably with a density greater than 0.945 g/ml) .
- Preferred melt index of the starting material depends on the desired application; however, the preferred melt index for injection molding is from 0.5 to 50 g/10 min; for film and thermoforming applications the preferred melt index is from 0.1 to 20 g/10 min; and for blow molding applications, the preferred melt index is from 0.01 to 20 g/10 min.
- the most preferred polymers as starting materials for this invention are ethylene copolymers with narrow MWD (that is a Mw/Mn of less than 3, more preferably less than 3.0, most preferably less than 2.5) . These can be produced using at least one C3-C20 olefin comonomer. Most preferred for copolymer is C3-C10. 0.5-5 mole percent comonomer as determined by ASTM 5017 is preferred in the starting material.
- Commercially available polymers in this category are known as TAFMERTM polymer commercially available from Mitsui Petrochemical
- ENGAGETM polyolefin elastomer commercially available from DuPont-Dow Elastomers.
- the most preferred comonomer content is between 3-25 weight percent.
- the preferred comonomer content is between 20-40 weight percent.
- the most preferred terpolymer is an EPDM such as NORDELTM ethylene/propylene/ diene polymer commercially available from DuPont-Dow Elastomers.
- Other very useful starting materials include the polymers made by the processes disclosed in WO 97/43323 and WO 97/44371. The melt index is measured according to ASTM D-1238 condition 190°C/2.16 Kg (formerly known as Condition E) .
- the polymer is reacted with a polyfunctional compound capable of insertion reactions into C-H bonds.
- a polyfunctional compound capable of insertion reactions into C-H bonds.
- Such polyfunctional compounds have at least two, preferably 2, functional groups capable of C-H insertion reactions.
- Those skilled in the art are familiar with C- H insertion reactions and functional groups capable of such reactions.
- the poly (sulfonyl azide) is any compound having at least two sulfonyl azide groups reactive with a polyolefin under reaction conditions.
- the poly (sulfonyl azide) s have a structure X- R-X wherein each X is S0 2 N 3 and R represents an unsubstituted or inertly substituted hydrocarbyl, hydrocarbyl ether or silicon-containing group, preferably having sufficient carbon, oxygen or silicon, preferably carbon, atoms to separate the sulfonyl azide groups sufficiently to permit a facile reaction between the polyolefin and the sulfonyl azide, more preferably at least 1, more preferably at least 2, most preferably at least 3 carbon, oxygen or silicon, preferably carbon, atoms between functional groups.
- each R advantageously has at least one carbon or silicon atom between X's and preferably has less than 50, more preferably less than 30, most preferably less than 20 carbon, oxygen or silicon atoms. Within these limits, larger is better for reasons including thermal and shock stability.
- R is straight-chain alkyl hydrocarbon, there are preferably less than 4 carbon atoms between the sulfonyl azide groups to reduce the propensity of the nitrene to bend back and react with itself.
- Silicon containing groups include silanes and siloxanes, preferably siloxanes.
- inertly substituted refers to substitution with atoms or groups which do not undesirably interfere with the desired reaction (s) or desired properties of the resulting coupled polymers.
- groups include fluorine, aliphatic or aromatic ether, siloxane as well as sulfonyl azide groups when more than two polyolefin chains are to be joined.
- Suitable structures include R as aryl, alkyl, aryl alkaryl, arylalkyl silane, siloxane or heterocyclic, groups and other groups which are inert and separate the sulfonyl azide groups as described.
- R includes at least one aryl group between the sulfonyl groups, most preferably at least two aryl groups (such as when R is 4,4' diphenylether or 4, 4' -biphenyl) .
- R is one aryl group, it is preferred that the group have more than one ring, as in the case of naphthylene bis(sulfonyl azides) .
- Poly (sulfonyl) azides include such compounds as 1, 5-pentane bis (sulfonyl azide), 1,8-octane bis (sulfonyl azide), 1,10-decane bis (sulfonyl azide), 1, 10-octadecane bis (sulfonyl azide), l-octyl-2, 4, 6- benzene tris (sulfonyl azide), 4, 4' -diphenyl ether bis(sulfonyl azide), 1, 6-bis (4' -sulfonazidophenyl) hexane, 2, 7-naphthalene bis (sulfonyl azide), and mixed sulfonyl azides of chlorinated aliphatic hydrocarbons containing an average of from 1 to 8 chlorine atoms and from 2 to 5 sulfonyl azide groups per molecule, and mixtures thereof.
- Preferred poly (sulfonyl azide) s include oxy-bis(4- sulfonylazidobenzene) , 2 , 7-naphthalene bis (sulfonyl azido), 4, 4' -bis (sulfonyl azido) biphenyl, 4, 4' -diphenyl ether bis (sulfonyl azide) and bis (4-sulfonyl azidophenyl) methane, and mixtures thereof.
- Sulfonyl azides are conveniently prepared by the reaction of sodium azide with the corresponding sulfonyl chloride, although oxidation of sulfonyl hydrazines with various reagents (nitrous acid, dinitrogen tetraoxide, nitrosonium tetrafluoroborate) has been used.
- Polyfunctional compounds capable of insertions into C-H bonds also include carbene- forming compounds such as salts of alkyl and aryl hydrazones and diazo compounds, and nitrene- forming compounds such as alkyl and aryl azides (R-N 3 ) , acyl azides (R-C(0)N 3 ), azidoformates (R-O- C(0)-N 3 ), sulfonyl azides (R-S0 2 -N 3 ) , phosphoryl azides ( (RO) 2 - (PO) -N 3 ) , phosphinic azides (R 2 -P (O) -N 3 ) and silyl azides (R 3 -Si-N 3 ) .
- carbene- forming compounds such as salts of alkyl and aryl hydrazones and diazo compounds
- nitrene- forming compounds such as alkyl and aryl azides (R-N 3 ) ,
- Some of the coupling agents of the invention are preferred because of their propensity to form a greater abundance of carbon-hydrogen insertion products.
- Such compounds as the salts of hydrazones, diazo compounds, azidoformates, sulfonyl azides, phosphoryl azides, and silyl azides are preferred because they form stable singlet-state electron products (carbenes and nitrenes) which carry out efficient carbon- hydrogen insertion reactions, rather than substantially 1) rearranging via such mechanisms as the Curtius-type rearrangement, as is the case with acyl azides and phosphinic azides, or 2) rapidly converting to the triplet-state electron configuration which preferentially undergoes hydrogen atom abstraction reactions, which is the case with alkyl and aryl azides.
- Phosphoryl azides may be reacted at temperatures in excess of 180°C up to 300°C, while silyl azides react preferentially at temperatures of from 250°C to 400°C.
- the poly (sulfonyl azide) is used in a rheology modifying amount, that is an amount effective to increase the low-shear viscosity (at 0.1 rad/sec) of the polymer preferably at least 5 percent as compared with the starting material polymer, but less than a crosslinking amount, that is an amount sufficient to result in at least 10 weight percent gel as measured by ASTM D2765- procedure A.
- the amount of azide sufficient to increase the low shear viscosity and result in less than 10 weight percent gel will depend on molecular weight of the azide used and polymer the amount is preferably less than 5 percent, more preferably less than 2 percent, most preferably less than 1 weight percent poly (sulfonyl azide) based on total weight of polymer when the poly (sulfonyl azide) has a molecular weight of from 200 to 2000.
- the amount of poly (sulfonyl azide) is preferably at least 0.01 weight percent, more preferably at least 0.05 weight percent, most preferably at least 0.10 weight percent based on total polymer.
- the sulfonyl azide is admixed with the polymer and heated to at least the decomposition temperature of the sulfonyl azide.
- decomposition temperature of the azide it is meant that temperature at which the azide converts to the sulfonyl nitrene, eliminating nitrogen and heat in the process, as determined by differential scanning calorimetry (DSC) .
- DSC differential scanning calorimetry
- the poly (sulfonyl azide) begins to react at a kinetically significant rate (convenient for use in the practice of the invention) at temperatures of 130°C and is almost completely reacted at 160°C in a DSC (scanning at 10°C/min) .
- Accelerated Rate Calorimetry (scanning at 2°C/ hr) shows onset of decomposition is 100°C. Extent of reaction is a function of time and temperature . At the low levels of azide used in the practice of the invention, the optimal properties are not reached until the azide is essentially fully reacted. Temperatures for use in the practice of the invention are also determined by the softening or melt temperatures of the polymer starting materials. For these reasons, the temperature is advantageously greater than 90°C, preferably greater than 120°C, more preferably greater than 150°C, most preferably greater than 180°C.
- Preferred times at the desired decomposition temperatures are times that are sufficient to result in reaction of the coupling agent with the polymer (s) without undesirable thermal degradation of the polymer matrix.
- Preferred reaction times in terms of the half life of the coupling agent that is the time required for half of the agent to be reacted at a preselected temperature, which half life is determinable by DSC is 5 half lives of the coupling agent .
- the reaction time is preferably at least 4 minutes at 200 °C.
- Admixing of the polymer and coupling agent is conveniently accomplished by any means within the skill in the art. Desired distribution is different in many cases, depending on what rheological properties are to be modified.
- a homopolymer it is desirable to have as homogeneous a distribution as possible, preferably achieving solubility of the azide in the polymer melt.
- a blend it is desirable to have low solubility in one or more of the polymer matrices such that the azide is preferentially in one or the other phase, or predominantly in the interfacial region between the two phases
- Preferred processes include at least one of (a) dry blending the coupling agent with the polymer, preferably to form a substantially uniform admixture and adding this mixture to melt processing equipment, for example a melt extruder to achieve the coupling reaction, at a temperature at least the decomposition temperature of the coupling agent; (b) introducing, for example by injection, a coupling agent in liquid form, for example dissolved in a solvent therefor or in a slurry of coupling agent in a liquid, into a device containing polymer, preferably softened, molten or melted polymer, but alternatively in particulate form, in solution or dispersion, more preferably in melt processing equipment; (c) forming a first admixture of a first amount of a first polymer and a coupling agent, advantageously at a temperature less than the decomposition temperature of the coupling agent, preferably by melt blending, and then forming a second admixture of the first admixture with a second amount of a second polymer
- process (c) is conveniently used to make a concentrate with a first polymer composition having a lower melting temperature, advantageously at a temperature below the decomposition temperature of the coupling agent, and the concentrate is melt blended into a second polymer composition having a higher melting temperature to complete the coupling reaction.
- Concentrates are especially preferred when temperatures are sufficiently high to result in loss of coupling agent by evaporation or decomposition not leading to reaction with the polymer, or other conditions would result that effect.
- some coupling occurs during the blending of the first polymer and coupling agent, but some of the coupling agent remains unreacted until the concentrate is blended into the second polymer composition.
- Each polymer or polymer composition includes at least one homopolymer, copolymer, terpolymer, or interpolymer and optionally includes additives within the skill in the art.
- the coupling agent is added in a dry form it is preferred to mix the agent and polymer in a softened or molten state below the decomposition temperature of the coupling agent then to heat the resulting admixture to a temperature at least equal to the decomposition temperature of the coupling agent .
- melt processing is used to mean any process in which the polymer is softened or melted, such as extrusion, pelletizing, molding, thermoforming, film blowing, compounding in polymer melt form, and fiber spinning.
- the polyolefin (s) and coupling agent are suitably combined in any manner which results in desired reaction thereof, preferably by mixing the coupling agent with the polymer (s) under conditions which allow sufficient mixing before reaction to avoid uneven amounts of localized reaction then subjecting the resulting admixture to heat sufficient for reaction.
- a substantially uniform admixture of coupling agent and polymer is formed before exposure to conditions in which chain coupling takes place.
- a substantially uniform admixture is one in which the distribution of coupling agent in the polymer is sufficiently homogeneous to be evidenced by a polymer having a melt viscosity after treatment according to the practice of the invention at least one of (a) higher at low angular frequency (for example 0.1 rad/sec) or (b) lower at higher angular frequency (for example 100 rad/sec) than that of the same polymer which has not been treated with the coupling agent but has been subjected to the same shear and thermal history.
- decomposition of the coupling agent occurs after mixing sufficient to result in a substantially uniform admixture of coupling agent and polymer.
- This mixing is preferably attained with the polymer in a molten or melted state, that is above the crystalline melt temperature, or in a dissolved or finely dispersed condition rather than in a solid mass or particulate form.
- the molten or melted form is more preferred to insure homogeneity rather than localized concentrations at the surface.
- Any equipment is suitably used, preferably equipment which provides sufficient mixing and temperature control in the same equipment, but advantageously practice of the invention takes place in such devices as an extruder or a static polymer mixing devise such as a Brabender blender.
- extruder is used for its broadest meaning to include such devices as a device which extrudes pellets or pelletizer.
- At least one step of the process of the invention takes place in the melt extrusion step.
- the reaction take place in a solvent or other medium, it is preferred that the reaction be in a bulk phase to avoid later steps for removal of the solvent or other medium.
- a polymer above the crystalline melt temperature is advantageous for even mixing and for reaching a reaction temperature (the decomposition temperature of the sulfonyl azide) .
- the process of the present invention takes place in a single vessel, that is mixing of the coupling agent and polymer takes place in the same vessel aa heating to the decomposition temperature of the coupling agent.
- the vessel is preferably a twin-screw extruder, but is also advantageously a single- screw extruder, a batch mixer, or a static mixing zone for mixing polymer at the back end of a production process .
- the reaction vessel more preferably has at least two zones of different temperatures into which a reaction mixture would pass, the first zone advantageously being at a temperature at least the crystalline melt temperature or the softening temperature of the polymer (s) and preferably less than the decomposition temperature of the coupling agents and the second zone being at a temperature sufficient for decomposition of the coupling agent.
- the first zone is preferably at a temperature sufficiently high to soften the polymer and allow it to combine with the coupling agent through distributive mixing to a substantially uniform admixture.
- the preferred embodiment for incorporation of coupling agent is to solution blend the coupling agent in solution or admixture into the polymer, to allow the polymer to imbibe (absorb or adsorb at least some of the coupling agent), and then to evaporate the solvent.
- the solvent is preferably a solvent for the coupling agent, and more preferably also for the polymer when the polymer is soluble such as in the case of polycarbonate.
- solvents include polar solvents such as acetone, THF
- the coupling agent be added to the post-reactor area of a polymer processing plant.
- the coupling agent is added in either powder or liquid form to the powdered polyethylene after the solvent is removed by decantation and prior to the drying and densification extrusion process.
- the coupling agent is preferably added in either powder or liquid form to the powdered polyethylene before the densification extrusion.
- the coupling agent is preferably added to the polymer melt stream after devolatilization and before the pelletizing extrusion process.
- rheology modified or chain coupled polymers that is the polymers which have sulfonamide, amine, alkyl-substituted or aryl -substituted carboxamide, alkyl -substituted or aryl -substituted phosphoramide , alkyl-substituted or aryl- substituted methylene coupling between different polymer chains.
- Resulting compounds advantageously show higher low shear viscosity than the original polymer due to coupling of long polymer chains to polymer backbones .
- Rheology modification leads to polymers which have controlled rheological properties, specifically improved melt strength as evidenced by increased low shear viscosity, better ability to hold oil, improved scratch and mar resistance, improved tackiness, improved green strength (melt) , higher orientation in high shear and high extensional processes such as injection molding, film extrusion (blown and cast) , calendaring, rotomolding, fiber production, profile extrusion, foams, and wire and cable insulation as measured by tan delta as described hereinafter, elasticity by viscosity at 0.1 rad/sec and 100 rad/sec, respectively. It is also believed that this process can be used to produce dispersions having improved properties of higher low shear viscosity than the unmodified polymer as measured by DMS.
- Rheology modified polymers are useful as large blow molded articles due to the higher low shear viscosity than unmodified polymer and the maintenance of the high shear viscosity for processability as indicated by high shear viscosity, foams for stable cell structure as measured by low shear viscosity, blown film for better bubble stability as measured by low shear viscosity, fibers for better spinnability as measured by high shear viscosity, cable and wire insulation for green strength to avoid sagging or deformation of the polymer on the wire as measured by low shear viscosity which are aspects of the invention.
- Polymers rheology modified according to the practice of the invention are superior to the corresponding unmodified polymer starting materials for these applications due to the elevation of viscosity, of preferably at least 5 percent at low shear rates (0.1 rad/sec) , sufficiently high melt strengths to avoid deformation during thermal processing (for example to avoid sag during thermoforming) or to achieve bubble strength during blow molding, and sufficiently low high shear rate viscosities to facilitate molding and extrusion.
- rheological attributes enable faster filling of injection molds at high rates than the unmodified starting materials, the setup of foams (stable cell structure) as indicated by formation of lower density closed cell foam, preferably with higher tensile strength, insulation properties, compression set or combinations thereof than attained in the use of coupling or rheology modification using coupling agents which generate free radicals, because of high melt viscosity .
- Advantageously toughness and tensile strength of the starting material is maintained.
- Polymers resulting from the practice of the invention are different from those resulting from practice of prior art processes as shown in CA 797,917.
- the polymers of the present invention show improved melt elasticity, that is higher tan delta as measured by DMS, better drawability, that is higher melt strength as measured by melt tension, less swelling as measured by blow molding die swell, and less shrinkage as measured by mold shrinkage than the unmodified polymer and the broad MWD (greater than 3.0 Mw/Mn) counterpart in thermoforming and large part blow molding.
- the rheology-modified ethylene polymers, processes for making them, and intermediates for making them of this invention are useful in the automotive area, industrial goods, building and construction, electrical (for example, wire and cable coatings/insulation) and tire products.
- Some of the fabricated articles include automotive hoses, single ply roofing, and wire and cable voltage insulation and jackets.
- Film and film structures particularly benefit from this invention and can be made using conventional hot blown film fabrication techniques or other biaxial orientation processes such as tenter frames or double bubble processes.
- Conventional hot blown film processes are described, for example, in The Encyclopedia of Chemical Technology. Kirk-Othmer, Third Edition, John Wiley & Sons, New York, 1981, vol. 16, pp. 416-417 and Vol. 18, pp. 191-192.
- Biaxial orientation film manufacturing process such as described in a "double bubble" process as in U.S. Patent 3,456,044 (Pahlke) , and the processes described in U.S. Patent 4,352,849 (Mueller), U.S. Patent 4,597,920 (Golike) , U.S.
- Patent 4,820,557 (Warren), U.S. Patent 4,837,084 (Warren), U.S. Patent 4,865,902 (Golike et al . ) , U.S. Patent 4,927,708 (Herran et al . ) , U.S. Patent 4,952,451 (Mueller), U.S. Patent 4,963,419 (Lustig et al . ) , and U.S. Patent 5,059,481 (Lustig et al . ) , can also be used to make film structures from the novel compositions described herein.
- the film structures can also be made as described in a tenter-frame technique, such as that used for oriented polypropylene .
- the films may be monolayer or multilayer films.
- the film made using this invention can also be coextruded with the other layer (s) or the film can be laminated onto another layer (s) in a secondary operation, such as that described in Packaging Foods With Plastics, by Wilmer A. Jenkins and James P. Harrington (1991) or that described in "Coextrusion For Barrier Packaging” by W.J. Schrenk and CR. Finch, Society of Plastics Engineers RETEC Proceedings . June 15-17 (1981), pp. 211-229. If a monolayer film is produced via tubular film (that is, blown film techniques) or flat die (that is, cast film) as described by K.R. Osborn and W.A.
- Extrusion coating is yet another technique for producing multilayer film structures using the novel compositions described herein.
- the novel compositions comprise at least one layer of the film structure.
- extrusion coating is a flat die technique.
- a sealant can be extrusion coated onto a substrate either in the form of a monolayer or a coextruded extrudate .
- the novel compositions described herein comprise at least one layer of the total multilayer film structure.
- Other layers of the multilayer structure include but are not limited to barrier layers or combinations thereof, tie layers, structural layers .
- Various materials can be used for these layers, with some of them being used as more than one layer in the same film structure.
- Some of these materials include: foil, nylon, ethylene/vinyl alcohol (EVOH) copolymers, polyvinylidene chloride (PVDC) , polyethylene terephthalate (PET) , oriented polypropylene (OPP) , ethylene/vinyl acetate (EVA) copolymers, ethylene/acrylic acid (EAA) copolymers, ethylene/methacrylic acid (EMAA) copolymers, LLDPE, HDPE, LDPE, nylon, graft adhesive polymers (for example, maleic anhydride grafted polyethylene) , and paper.
- the multilayer film structures comprise from 2 to 7 layers .
- Such articles comprising the rheology-modified polymer of this invention may be made by melt processing the rheology-modified polymer according to this invention. That process may include processing pellets or granules of polymer which have been rheology-modified according to this invention. In a preferred embodiment, the pellets or granules are substantially free of unreacted coupling agent when the coupling agent comprises a heat-activated coupling agent.
- Such articles may also be made by melt processing an intermediate comprising a homogeneous polymer which is not substantially free of unreacted coupling agent .
- Such intermediates are preferably treated with a coupling agent, but are not subjected to subsequent melt processing until the polymer is melted to make the article.
- the coupling agent may be either radiation or a heat-activated crosslinking agent.
- the rheology-modified polymers and intermediates used to make rheology-modified polymers may be used alone or in combination with one or more additional polymers in a polymer blend.
- additional polymers may be selected from any of the modified or unmodified homogeneous polymers described above for this invention, any modified or unmodified heterogeneous polymers or combinations thereof .
- Heterogeneous polyethylenes that are optionally combined with the rheology-modified polymers according to this invention fall into two broad categories, those prepared with a free radical initiator at high temperature and high pressure, and those prepared with a coordination catalyst at high temperature and relatively low pressure.
- the former are generally known as low density polyethylenes (LDPE) and are characterized by branched chains of polymerized monomer units pendant from the polymer backbone.
- LDPE polymers generally have a density between 0.910 and 0.935 g/cc.
- Ethylene polymers and copolymers prepared by the use of a coordination catalyst, such as a Ziegler or Phillips catalyst, are generally known as linear polymers because of the substantial absence of branch chains of polymerized monomer units pendant from the backbone.
- High density polyethylene generally having a density of 0.941 to 0.965 g/cc, is typically a homopolymer of ethylene, and it contains relatively few branch chains relative to the various linear copolymers of ethylene and an ⁇ - olefin.
- HDPE is well known, commercially available in various grades, and may be used in this invention. Density is measured according to the procedure of ASTM D- 792. Linear copolymers of ethylene and at least one ⁇ - olefin of 3 to 12 carbon atoms, preferably of 4 to 8 carbon atoms, are also well known and commercially available.
- the density of a linear ethylene/ ⁇ -olefin copolymer is a function of both the length of the ⁇ -olefin and the amount of such monomer in the copolymer relative to the amount of ethylene, the greater the length of the ⁇ -olefin and the greater the amount of ⁇ -olefin present, the lower the density of the copolymer.
- Linear low density polyethylene is typically a copolymer of ethylene and an ⁇ -olefin of 3 to 12 carbon atoms, preferably 4 to 8 carbon atoms (for example, 1-butene, 1-octene, etc.), that has sufficient ⁇ -olefin content to reduce the density of the copolymer to that of LDPE .
- the copolymer contains even more ⁇ -olefin, the density will drop below 0.91 g/cc and these copolymers are known as ultra low density polyethylene (ULDPE) or very low density polyethylene (VLDPE) .
- the densities of these linear polymers generally range from 0.87 to 0.91 g/cc.
- Heterogeneous linear ethylene polymers are available from The Dow Chemical Company as DowlexTM LLDPE and as AttaneTM ULDPE resins. Heterogeneous linear ethylene polymers can be prepared via the solution, slurry or gas phase polymerization of ethylene and one or more optional ⁇ - olefin comonomers in the presence of a Ziegler Natta catalyst, by processes such as are disclosed in U.S. Patent No. 4,076,698 to Anderson et al ..
- heterogeneous ethylene polymers are typically characterized as having molecular weight distributions, M w /M a l in the range of from 3.5 to 4.1. Relevant discussions of both of these classes of materials, and their methods of preparation are found in U.S. Patent No. 4,950,541 and the patents to which it refers.
- compositions of the invention and compositions produced by practice of the invention are particularly useful because of their surprising properties.
- the preferred medium density polyethylenes and ethylene copolymers (density 0.90 g/mL, comonomer content 0.5 - 5 mole percent) of the invention are particularly useful as blown films such as in trash bags, grocery sacks, sealant layers, tie layers, produce bags, garment bags, shipping sacks, medical films, stretch film, shrink film, agricultural film, construction film, geomembranes, or stretch hooders, preferably trash bags, agricultural film, construction film, and geomembranes.
- the medium density preferred embodiments are useful in cast films such as are useful in stretch films, diaper backsheets, industrial wrap, produce wrap, meat wrap, consumer wrap, shrink film, or elastic film, preferably as elastic film.
- the high density polyethylene (density greater than 0.945 g/mL and preferably MWD less than or equal to 3.0) preferred embodiments are particularly useful for thermoforming, preferably for use in refrigerator liners, thin walled containers, medical blister packs, modified atmosphere packaging; and in blow molding (including extrusion blow molding) to form such articles as oil bottles, pipe, fuel tanks including fuel filler necks, milk jugs, and trigger bottles.
- the low density ethylene copolymer preferred embodiments are particularly useful in extrusion such as to form wire and cable coatings, tubing, profiles such as gaskets and seals, sheeting, extrusion coatings such as carpet backing, multilayer packaging, tougheners, and impact modifiers for blends of polymers, preferably for wire and cable coating, tougheners and impact modifiers.
- the low density preferred embodiments are also particularly useful for calendaring to form such materials as sheeting, packaging films, and non-packaging films,- for foams particularly cushion packaging, toys, building and construction uses, automotive uses, body boards, airline seats, floral and craft uses, preferably cushion packaging, building and construction, automotive uses, and body boards; and for adhesives and sealants, particularly hot melt adhesives, pressure sensitive adhesives (whether applied in solvents or by hot melt) , caulks, and as tackifiers in other compositions.
- foams particularly cushion packaging, toys, building and construction uses, automotive uses, body boards, airline seats, floral and craft uses, preferably cushion packaging, building and construction, automotive uses, and body boards
- adhesives and sealants particularly hot melt adhesives, pressure sensitive adhesives (whether applied in solvents or by hot melt) , caulks, and as tackifiers in other compositions.
- ethylene polymers particularly high density polyethylene, and propylene polymers in the automotive field.
- melt strength it becomes possible to produce such automotive articles as fascia, bumper energy absorbers, bumper beams, door trim panels, door hardware cartridges, seat backs, seat pans, head rest cores, header trim, header energy absorbing (EA) inserts, pillars, instrument panels, instrument panel trim, bolsters, glove boxes, doors, consoles, ducts, parcel shelves, hat shelves, load floors, rocker panels, fenders and combinations thereof using such means as blow molding, injection molding, thermoforming, and gas assisted injection molding.
- EA header energy absorbing
- Such automotive articles as cross car supports, door outer panels, seat trim, and combinations thereof can be produced using such means as blow molding, injection molding, and thermoforming.
- Such articles as roof liners, underbody closeouts (underbody shields) , pick-up bed liners, and wheel liners can be conveniently produced by thermoforming; while such articles as fuel filler necks and fuel tanks can be produced using blow molding, roto- molding or injection molding.
- Injection molding, thermoforming and gas assisted injection molding are useful for producing door impact beams .
- Such articles as bumper beams, door impact beams, heater trim, roof liners, ducts, pick-up bed liners, fuel filler necks, fuel transport lines and conductive fuel systems are conveniently produced by extrusion or coextrusion as well .
- Coextrusion is additionally useful for rocker panels and fenders.
- Conductive fuel systems are also optionally roto-molded or blow molded. Roto-molding is also useful for door EA inserts, seat backs, head rest cores, header EA, instrument panel trim, bolsters, and ducts; while blow molding is additionally useful for door EA inserts.
- foaming is useful for bumper energy absorbers, bumper beams, door trim panels, door EA inserts, seat trim, head rest cores, header trim, roof liners, header EA, pillars, instrument panel trim, bolsters, and pick-up bed liners.
- Compression thermoforming that is thermoforming at a pressure greater than 240 kPa, is useful to produce such articles as bumper beams, cross car supports, door impact beams, door outer panels, door hardware cartridges, seat backs, seat pans, header trim, roof liners, instrument panel trim, bolsters, ducts, parcel shelves, hat shelves, load floors, rocker panels, fenders, underbody closeouts, pick-up bed liners, wheel liners and combinations thereof.
- Practice of the invention advantageously facilitates blow molding, thermoforming and foaming ethylene polymers and propylene polymers that without reaction with poly (sulfonyl azide) would not be conveniently shaped using those methods and which have a fractional melt (as measured by the procedure of ASTM-
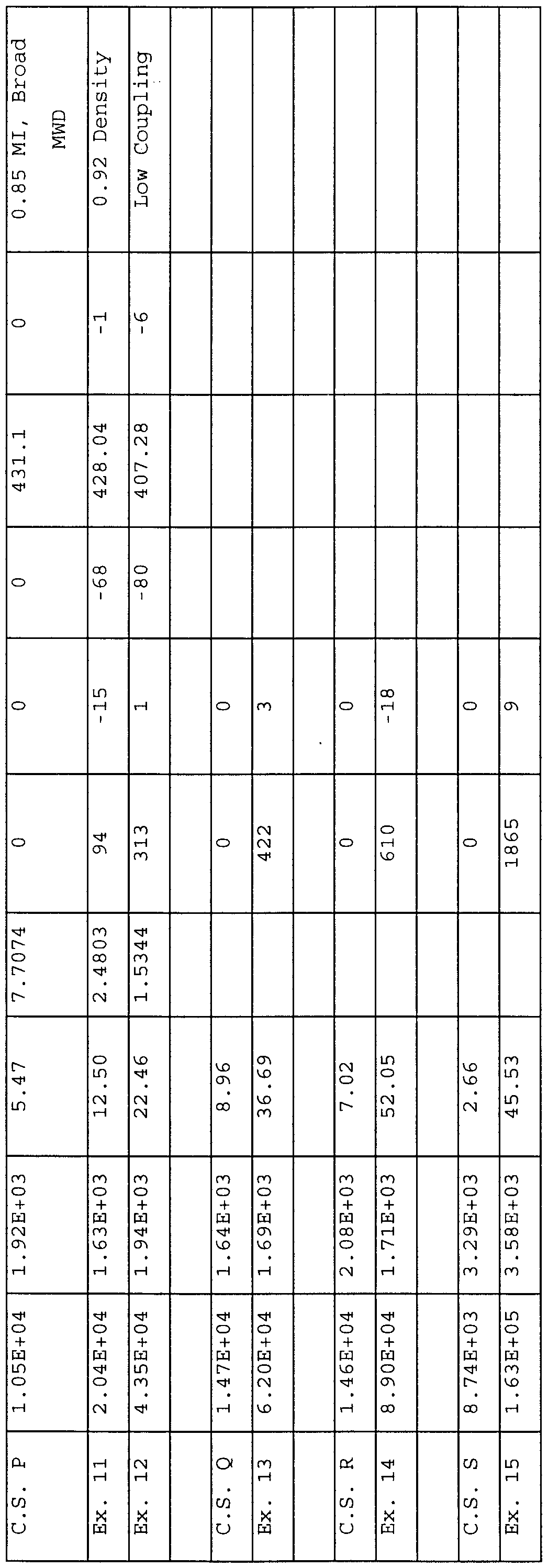
- Examples (Ex) of the invention are designated numerically while comparative samples (C.S.) are designated alphabetically and are not examples of the invention.
- a Rheometrics, Inc. RMS- 800 dynamic mechanical spectrometer with 25mm diameter parallel plates was used to determine the dynamic rheological data.
- a frequency sweep with five logarithmically spaced points per decade was run from 0.1 to 100 rad/s at 190 C.
- the strain was determined to be within the linear viscoelastic regime by performing a strain sweep at 0.1 rad/s and 190 C, by strain sweep from 2 to 30 percent strain in 2 percent steps to determine the minimum required strain to produce torques within the specification of the transducer; another strain sweep at 100 rad/s and 190 C was used to determine the maximum strain before nonlinearity occurred according to the procedure disclosed by J. M. Dealy and K. F. Wissbrun, "Melt Rheology and Its Role in Plastics Processing", Van Nostrand, New York (1990). All testing was performed in a nitrogen purge to minimize oxidative degradation.
- a Perkin Elmer model TMA 7 thermomechanical analyzer was used to measure the upper service temperature . Probe force of 102g and heating rate of 5°C/min were used. Each test specimen was a disk with thickness of 2 mm and diameter, prepared by compression molding at 205°C and air-cooling to room temperature.
- Samples are prepared using either a mixer commercially available from Haake, Inc. under the trade designation HaakeBuchler Rheomix 600 mixer with roller style blades, attached to a rheometer commercially available from Haake, Inc. under the trade designation HaakeBuchler Rheocord 9000 Torque rheometer, or using a Brabender mixer (Type R.E.E. No. A-19/S.B) with a 50 g mixing bowl.
- a Brabender mixer Type R.E.E. No. A-19/S.B
- the polymer was melted at 100°C for 2 minutes until all pellets were molten. Then 0.05 weight percent of 4,4'- oxybis (benzenesulfonyl azide) (Hereinafter BSA) CAS# [7456-68-0] was mixed into the molten polymer for 2 minutes. After intimate mixing was achieved, the temperature was adjusted to 170°C and the rotational speed was increased from 20 to 40 rpm over a period of 7 minutes to reach a maximum of 180°C. The mixture is held at this higher temperature and high rotational speed for 12 minutes, and then it was cooled to 150°C. The sample was removed from the Haake and allowed to cool to room temperature .
- BSA 4,4'- oxybis (benzenesulfonyl azide)
- Example 2 the procedure of Example 1 was repeated but using 0.1 weight percent 4, 4' -oxybis (benzenesulfonyl azide) (BSA) .
- BSA 4' -oxybis (benzenesulfonyl azide)
- EXACT 3028 commercially available from Exxon Chemical Company under the trade designation EXACT 3028 reported to be made using a metallocene catalyst, using 0.05 weight percent of BSA for Example 3, 0.1 weight percent for Example 4 and no poly (sulfonyl azide) for Comparative Samples B.
- the following is an example of using a multimodal polymer with an overall MWD > 3.00, but for which at least one component of the product has a MWD ⁇ 3.0 and is reported to be made using a metallocene catalyst.
- Elastomers LLC under the trade designation Nordel IP NDR 3720 hydrocarbon rubber (containing 1000 ppm Irganox 1076 stabilizer) and using 0.05 weight percent of BSA for Example 5, 0.1 weight percent of BSA for Example 6 and no poly (sulfonyl azide) for Comparative Samples C.
- ENGAGE EG8100 (reported to be made using a constrained geometry catalyst) with 0, 0.05, and 0.1 weight percent of BSA for C.S. J, Example 7, and Example 8, respectively.
- Examples 11 and 12 and Comparative Sample P The following is an example of rheology modification of a multimodal polymer product with an overall MWD > 3.0, but for which at least one of the components has a MWD ⁇ 3.0 and is reported to be made using a metallocene catalyst.
- Elite 5100 containing 1250 ppm calcium stearate, 500 ppm Irganox 1076 antioxidant, and 800 ppm P-EPQ antioxidant
- Example 13 and Comparative Sample The procedure of Example 1 is repeated using an ethylene- octene copolymer with melt index of 1.0 g/ 10 min (by
- Example 1 The procedure of Example 1 is repeated using an ethylene- octene copolymer with melt index of 0.80 g/ 10 min (by
- the rheology modification efficiency is surprisingly influenced by the molecular weight distribution, molecular weight, type of comonomer, and the comonomer content.
- the trend is that the narrow distribution polymers or higher comonomer content polymers show better rheology modification efficiency.
- the degree of efficiency is dependent on the type of polymer, and this is discussed hereinafter.
- the low shear viscosity (viscosity 0.1 rad/s) increased by 103 percent over that of the base polymer with the higher shear viscosities increasing less substantially at 3 percent at 100 rad/s and 22 percent at 1000 l/s.
- a substantial increase in low shear viscosity (100 percent at 0.1 rad/s) correlatable with increases in melt strength were observed for a metallocene based EPDM with less substantial changes in high shear viscosity (3 percent at 100 rad/s) reflecting good processability.
- the higher molecular weight C.S. I showed significant modification with a low shear viscosity increase of 63 percent with increases in high shear viscosity being 4 percent at 100 rad/s.
- C.S. F The lower molecular weight, C.S. F showed more minor changes of 13 percent in the low shear viscosity and a 13 percent change in the high shear viscosity.
- the tan delta at 0.1 rad/s change also decreased substantially from 3 percent to -29 percent to -61 percent from the C.S. E and C.S. F to Examples 5 and 6. (Note: C.S. D and C.S. E with 0.05 percent azide were rerun to verify that there was a slight drop in low shear viscosity with 0.05 percent azide) .
- the higher comonomer content polymer (Example 7 and 8) has better rheology modification efficiency, probably due to higher comonomer content.
- the viscosity changes at 0.1 rad/sec are 241 percent for Example 2 and 366 percent for Example 8.
- the rheology modification efficiency is high due to the combination of narrow MWD and high comonomer content.
- the comonomer content used in these polymers is higher than a corresponding density ethylene- octene copolymer.
- the viscosity changes at 0.1 rad/sec are 241 percent for Example 2 and 2214 percent for Example 4.
- the C.S. E and C.S. F showed a minor effect of coupling in terms of minor changes in storage modulus and Tg. No Tg shift was observed for the C.S. H and C.S. I with slight increases in modulus with increasing azide level. Examples 5 and 6 showed a significant shift in storage modulus increasing by 38 percent at 25 ° C and by 167 percent at 153 ° C. The Tg was also raised 7
- Examples 1 and 2 showed a broadening and raising of the Tg with coupling and a significant increase in modulus (60 percent) in the melt regime (the temperature greater than 100 C) .
- Examples 7 and 8 showed no significant shift in Tg, a significant increase in room temperature modulus and a large increase in the high temperature modulus (471 percent) .
- Examples 3 and 4 showed a broadening and increase of Tg with coupling with significant increases in room temperature modulus (43 percent) and high temperature modulus (5914 percent) .
- Examples 9 and 10 showed an increase in Tg of 3 C, an increase in room temperature modulus of 30 percent and an increase in high temperature modulus of 1017 percent.
- N showed no significant increase in Tg but an increase in the intensity of the alpha (melting) transition, an increase in room temperature modulus of 40 percent and high temperature modulus of 605 percent.
- Examples 11 and 12 showed a similar increase in the intensity of the alpha transition, no change in room temperature modulus and a 510 percent increase in higher temperature modulus.
- the melt rheological behavior shows the largest differences between the comparative samples (not subjected to coupling reactions) due to coupling in the practice of the invention.
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR9814442A BR9814442A (en) | 1997-08-27 | 1998-08-26 | Process for preparing a coupled polymer, composition, article and use thereof |
| KR10-2000-7001910A KR100535582B1 (en) | 1997-08-27 | 1998-08-26 | Rheology modification of polymers prepared using metallocenes |
| AU89210/98A AU743302B2 (en) | 1997-08-27 | 1998-08-26 | Rheology modification of polymers prepared using metallocenes |
| JP2000507746A JP4342721B2 (en) | 1997-08-27 | 1998-08-26 | Flow modification of polymers produced using metallocenes |
| PL33908398A PL339083A1 (en) | 1997-08-27 | 1998-08-26 | Modification of rheological properties of polymers obtained using metalocenes |
| CA 2301835 CA2301835C (en) | 1997-08-27 | 1998-08-26 | Rheology modification of polymers prepared using metallocenes |
| EP19980941064 EP1007590A1 (en) | 1997-08-27 | 1998-08-26 | Rheology modification of polymers prepared using metallocenes |
| NO20000966A NO322848B1 (en) | 1997-08-27 | 2000-02-25 | Coupled polymer, method of preparation and use thereof. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US5758297P | 1997-08-27 | 1997-08-27 | |
| US60/057,582 | 1997-08-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1999010427A1 true WO1999010427A1 (en) | 1999-03-04 |
Family
ID=22011502
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1998/016214 WO1999010422A1 (en) | 1997-08-27 | 1998-08-05 | Rheology modification of low density polyethylene |
| PCT/US1998/016213 WO1999010421A1 (en) | 1997-08-27 | 1998-08-05 | Rheology modification of interpolymers of alpha-olefins and vinyl aromatic monomers |
| PCT/US1998/016215 WO1999010423A1 (en) | 1997-08-27 | 1998-08-05 | Rheology modification of elastomers |
| PCT/US1998/016810 WO1999010415A1 (en) | 1997-08-27 | 1998-08-13 | Process of rheology modification of polymers |
| PCT/US1998/017685 WO1999010427A1 (en) | 1997-08-27 | 1998-08-26 | Rheology modification of polymers prepared using metallocenes |
Family Applications Before (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1998/016214 WO1999010422A1 (en) | 1997-08-27 | 1998-08-05 | Rheology modification of low density polyethylene |
| PCT/US1998/016213 WO1999010421A1 (en) | 1997-08-27 | 1998-08-05 | Rheology modification of interpolymers of alpha-olefins and vinyl aromatic monomers |
| PCT/US1998/016215 WO1999010423A1 (en) | 1997-08-27 | 1998-08-05 | Rheology modification of elastomers |
| PCT/US1998/016810 WO1999010415A1 (en) | 1997-08-27 | 1998-08-13 | Process of rheology modification of polymers |
Country Status (21)
| Country | Link |
|---|---|
| US (9) | US6376623B1 (en) |
| EP (5) | EP1007585A1 (en) |
| JP (5) | JP2001514287A (en) |
| KR (5) | KR100562616B1 (en) |
| CN (3) | CN1273592A (en) |
| AR (5) | AR016885A1 (en) |
| AT (3) | ATE306517T1 (en) |
| AU (5) | AU743240B2 (en) |
| BR (3) | BR9814447A (en) |
| CA (5) | CA2301644C (en) |
| DE (3) | DE69834123T2 (en) |
| ES (2) | ES2247709T3 (en) |
| HU (1) | HUP0002756A3 (en) |
| ID (1) | ID25634A (en) |
| MY (4) | MY133100A (en) |
| NO (1) | NO322848B1 (en) |
| PL (1) | PL339083A1 (en) |
| TR (2) | TR200000905T2 (en) |
| TW (5) | TW591070B (en) |
| WO (5) | WO1999010422A1 (en) |
| ZA (5) | ZA987752B (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014145082A (en) * | 1998-11-02 | 2014-08-14 | Dow Global Technologies Llc | SHEAR THINNING ETHYLENE/α-OLEFIN INTERPOLYMERS AND THEIR PREPARATION METHOD |
| US8883870B2 (en) | 2010-09-15 | 2014-11-11 | Dow Global Technologies Llc | Propylene-alpha-olefin copolymer compositions with improved foaming window |
| US10471690B2 (en) | 2014-12-30 | 2019-11-12 | Dow Global Technologies Llc | Sulfonylazide derivative for tie layer |
Families Citing this family (131)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6506842B1 (en) * | 1997-01-29 | 2003-01-14 | Dupont Dow Elastomers L.L.C. | Rheology-modified thermoplastic elastomer compositions and articles fabricated therefrom |
| EP0966346B1 (en) * | 1997-03-10 | 2003-06-04 | Perstorp Ab | A process for the manufacturing of thermoplastic products with high creep strain resistance |
| US6376623B1 (en) * | 1997-08-27 | 2002-04-23 | The Dow Chemical Company | Rheology modification of elastomers |
| US6709742B2 (en) | 1998-05-18 | 2004-03-23 | Dow Global Technologies Inc. | Crosslinked elastic fibers |
| US6277916B1 (en) | 1999-02-25 | 2001-08-21 | The Dow Chemical Company | Process for preparing thermoplastic vulcanizates |
| US6365693B1 (en) | 1999-05-07 | 2002-04-02 | Dupont Dow Elastomers, L.L.C. | Fluoroalkanesulfonyl azide copolymers |
| US6472473B1 (en) | 1999-06-24 | 2002-10-29 | Dow Global Technology Inc. | Polyolefin composition with improved impact properties |
| EP1457518A1 (en) * | 1999-12-03 | 2004-09-15 | Dow Global Technologies, Inc. | Grafted thermoplastic compositions and fabricated articles therefrom |
| US6395791B1 (en) | 1999-12-03 | 2002-05-28 | The Dow Chemical Company | Grafted thermoplastic compositions and fabricated articles therefrom |
| US6291618B1 (en) | 1999-12-23 | 2001-09-18 | The Dow Chemical Company | Phosphazene azide coupling agents and their use in rheology modification of polymers |
| EP1276611B1 (en) | 2000-01-24 | 2004-04-21 | Dow Global Technologies Inc. | Composition and films thereof |
| DE60117536T2 (en) * | 2000-05-04 | 2006-11-09 | Dow Global Technologies, Inc., Midland | MOLECULAR MELT AND METHOD FOR THE PRODUCTION AND USE THEREOF |
| CA2410483C (en) * | 2000-05-26 | 2009-08-11 | Dow Global Technologies Inc. | Polyethylene rich/polypropylene blends and their uses |
| US6548600B2 (en) | 2000-09-22 | 2003-04-15 | Dupont Dow Elastomers L.L.C. | Thermoplastic elastomer compositions rheology-modified using peroxides and free radical coagents |
| US20040041347A1 (en) * | 2000-11-30 | 2004-03-04 | Beach Jefferey Kurtis | Permanently lubricated gasket |
| US7469905B2 (en) | 2000-11-30 | 2008-12-30 | Springseal Inc. | Permanently lubricated film gasket and method of manufacture |
| WO2002051890A1 (en) * | 2000-12-22 | 2002-07-04 | Dow Global Technologies Inc. | Propylene copolymer foams |
| WO2003029344A1 (en) * | 2001-10-01 | 2003-04-10 | Dow Global Technologies Inc. | Blow moldable propylene polymer compositions |
| US20050070673A1 (en) * | 2001-10-01 | 2005-03-31 | Novak Leo R. | Thermoformable propylene polymer compositions |
| WO2003040229A1 (en) * | 2001-11-02 | 2003-05-15 | Dow Global Technologies Inc. | Molecular melt and methods for making and using the molecular melt |
| EP1860214B1 (en) | 2002-03-11 | 2009-04-29 | Dow Global Technologies Inc. | Reversible, heat-set, elastic fibers, and method of making and articles made from same |
| KR20040089730A (en) * | 2002-03-11 | 2004-10-21 | 다우 글로벌 테크놀로지스 인크. | Reversible, heat-set, elastic fibers, and method of making and articles made from same |
| US6921563B2 (en) * | 2002-05-24 | 2005-07-26 | Ticona Gmbh | Multi-layer laminate, packaging material and packages made therefrom |
| US6734253B2 (en) * | 2002-07-19 | 2004-05-11 | Dow Global Technologies, Inc. | Scratch and mar resistant propylene polymer composition |
| US6864195B2 (en) * | 2002-08-15 | 2005-03-08 | Bfs Diversified Products, Llc | Heat weldable roofing membrane |
| US6846884B2 (en) * | 2002-09-27 | 2005-01-25 | Union Carbide Chemicals & Plastics Technology Corporation | Control of resin properties |
| US7338994B2 (en) * | 2002-10-01 | 2008-03-04 | Dow Global Technologies Inc. | Rheology-modified thermoplastic elastomer compositions for extruded profiles |
| US6908687B2 (en) | 2002-12-30 | 2005-06-21 | Exxonmobil Oil Corporation | Heat-shrinkable polymeric films |
| US7101628B2 (en) * | 2003-03-20 | 2006-09-05 | Bfs Diversified Products, Llc | Thermoplastic vulcanizate and membrane for covering a roof made therefrom |
| US6930148B2 (en) * | 2003-04-08 | 2005-08-16 | Texas Petrochemicals Lp | Enhanced polyisobutylene modified hot melt adhesive formulation |
| CN1806004B (en) * | 2003-06-10 | 2010-06-16 | 陶氏环球技术公司 | Film layers made from ethylene polymer blends |
| US20060169477A1 (en) * | 2003-07-24 | 2006-08-03 | Gross Laurence H | Cable insulation system with flexibility, high temperature deformation resistance, and reduced degree of stickiness |
| US7632086B2 (en) * | 2003-10-03 | 2009-12-15 | Exxonmobil Chemical Patents Inc. | Melt fracture reduction |
| BRPI0415577A (en) * | 2003-10-22 | 2007-04-17 | Procter & Gamble | composition combined with a blow molded thermoplastic package |
| US7906586B2 (en) * | 2003-12-09 | 2011-03-15 | Dow Global Technologies Llc | Thermoplastic olefinic compositions |
| DE602004014718D1 (en) * | 2004-03-12 | 2008-08-14 | Borealis Tech Oy | Crosslinked pressure tube made of polyethylene and process for its preparation |
| US8129009B2 (en) * | 2004-04-13 | 2012-03-06 | E. I. Du Pont De Nemours And Company | Composition comprising ethylene copolymer |
| JP2005320469A (en) * | 2004-05-11 | 2005-11-17 | Idemitsu Kosan Co Ltd | Polycarbonate resin composition and molded article |
| US20070244234A1 (en) * | 2004-06-14 | 2007-10-18 | Polyone Corporation | Durable and Low Friction Thermoplastic Elastomer Compositions |
| US20060020063A1 (en) * | 2004-07-22 | 2006-01-26 | Guenther Gerhard K | Controlled finishes for free surface polyethylene resins |
| EP1819496A4 (en) * | 2004-12-08 | 2009-10-28 | Advanced Elastomer Systems | Molded gasket and method of making |
| EP1831303B1 (en) * | 2004-12-17 | 2010-04-14 | Dow Global Technologies Inc. | Rheology modified polyethylene compositions |
| EP1674504A1 (en) * | 2004-12-22 | 2006-06-28 | Total Petrochemicals Research Feluy | Geo-membrane applications |
| CN101094889B (en) * | 2005-01-03 | 2010-12-15 | 陶氏环球技术公司 | Elastomeric resin compositions with improved resistance to draw resonance |
| CN101111527B (en) * | 2005-01-31 | 2010-09-15 | 三井化学株式会社 | Ethylene series resin and molding obtained therefrom |
| BRPI0607992A2 (en) * | 2005-03-04 | 2009-10-27 | Dow Global Technologies Inc | polyethylene composition, film, and thermoformed article |
| US7642330B2 (en) * | 2005-03-31 | 2010-01-05 | Exxonmobil Chemical Patents Inc. | Method of selecting polyolefins based on rheological properties |
| US20060222796A1 (en) * | 2005-04-04 | 2006-10-05 | Morris Barry A | Structure comprising metallized film and ethylene copolymer |
| DE602006005645D1 (en) * | 2005-05-12 | 2009-04-23 | Dow Global Technologies Inc | Thermoformed and extruded film with reduced gloss |
| DE102005023040A1 (en) * | 2005-05-13 | 2006-11-16 | Basell Polyolefine Gmbh | Polyolefinic molding composition for producing pipes with improved resistance comprises thermoplastic polyolefin, and organic polyoxy compound and/or organic polyhydroxy compound |
| US7399737B2 (en) | 2005-06-13 | 2008-07-15 | Exxonmobil Chemical Patents Inc. | Lube additives |
| US20070045904A1 (en) * | 2005-08-31 | 2007-03-01 | Ulcej John A | Blow molding extrusion apparatus and method |
| KR20080060289A (en) | 2005-10-26 | 2008-07-01 | 다우 글로벌 테크놀로지스 인크. | Multi-layer, elastic articles |
| US7491761B2 (en) * | 2005-11-01 | 2009-02-17 | Solutia Incorporated | Poly(vinyl butyral) pellets |
| AU2006317055A1 (en) | 2005-11-23 | 2007-05-31 | Dow Global Technologies Inc. | Heterogeneous, compositionally phase separated, ethylene alpha-olefin interpolymers |
| US8153243B2 (en) | 2005-12-09 | 2012-04-10 | Dow Global Technologies Llc | Interpolymers suitable for multilayer films |
| US20070182106A1 (en) * | 2006-02-06 | 2007-08-09 | Biesenberger Jeffrey J | Lubeless pipe gasket and method of fabrication |
| CN101437878B (en) | 2006-03-10 | 2012-09-05 | 陶氏环球技术有限责任公司 | Polyethylene resins for sheet and thermoforming applications |
| US7462391B2 (en) * | 2006-05-17 | 2008-12-09 | The Goodyear Tire & Rubber Company | Multi-layered veneer for a tire sidewall |
| BR122018012418B1 (en) | 2006-09-21 | 2019-09-24 | Union Carbide Chemicals & Plastics Technology Llc. | METHOD FOR CONTROLING A POLYMER COMPOSITION PRODUCTION PROCESS |
| BRPI0717496B1 (en) | 2006-10-06 | 2018-06-26 | Bridgestone Corporation | Tire component and method for preparing a polymer |
| US8474830B2 (en) | 2007-06-07 | 2013-07-02 | Springseal, Inc. | Gasket |
| US20090018253A1 (en) * | 2007-07-09 | 2009-01-15 | Eastman Chemical Company | Perfromance additives for thermoplastic elastomers |
| DE102008050219B4 (en) * | 2007-10-05 | 2020-06-10 | The Yokohama Rubber Co., Ltd. | Rubber composition for tires, manufacture and use of the like and vulcanized composition |
| US8404773B2 (en) | 2008-03-17 | 2013-03-26 | Dow Global Technologies Llc | Coating composition, method of producing the same, articles made therefrom, and method of making such articles |
| US8727387B2 (en) * | 2008-05-27 | 2014-05-20 | Springseal, Inc. | Pipe coupling assembly |
| EP2315802A4 (en) * | 2008-07-31 | 2012-04-18 | 3M Innovative Properties Co | Azide compositions and methods of making and using thereof |
| WO2010033601A1 (en) * | 2008-09-16 | 2010-03-25 | Dow Global Technologies Inc. | Polymeric compositions and foams, methods of making the same, and articles prepared from the same |
| US7960024B2 (en) | 2009-01-27 | 2011-06-14 | Milliken & Company | Multi-layered fiber |
| US8114507B2 (en) * | 2009-01-27 | 2012-02-14 | Milliken & Company | Multi-layered fiber |
| US8119549B2 (en) | 2009-01-27 | 2012-02-21 | Milliken & Company | Consolidated fibrous structure |
| US8147957B2 (en) * | 2009-01-27 | 2012-04-03 | Milliken & Company | Consolidated fibrous structure |
| US8029633B2 (en) * | 2009-01-27 | 2011-10-04 | Milliken & Company | Method of forming a consolidated fibrous structure |
| US20100239796A1 (en) * | 2009-03-23 | 2010-09-23 | Gagne Joseph Donald | Lap sealable laminate and packaging made therefrom |
| US20110319987A1 (en) * | 2009-05-20 | 2011-12-29 | Arsenal Medical | Medical implant |
| EP2432425B1 (en) * | 2009-05-20 | 2018-08-08 | 480 Biomedical, Inc. | Medical implant |
| US9265633B2 (en) | 2009-05-20 | 2016-02-23 | 480 Biomedical, Inc. | Drug-eluting medical implants |
| US8888840B2 (en) * | 2009-05-20 | 2014-11-18 | Boston Scientific Scimed, Inc. | Drug eluting medical implant |
| CN102803306A (en) * | 2009-06-23 | 2012-11-28 | 陶氏环球技术有限责任公司 | Controlled-rheology Polypropylene |
| KR20120107066A (en) * | 2009-07-01 | 2012-09-28 | 다우 글로벌 테크놀로지스 엘엘씨 | Ethylenic polymer and its use |
| KR101761549B1 (en) | 2009-07-14 | 2017-07-26 | 다우 글로벌 테크놀로지스 엘엘씨 | Vacuum thermoformed, extruded sheeting with improved reduced gloss |
| DE202009013150U1 (en) * | 2009-10-01 | 2011-02-17 | Peguform Gmbh | Mounting system for mounting a bumper cover |
| CN102597120B (en) | 2009-11-11 | 2015-05-27 | 3M创新有限公司 | Polymeric compositions and method of making and articles thereof |
| BR112012011058B1 (en) | 2009-11-23 | 2021-06-08 | Dow Global Technologies Llc | composition, film and article |
| CN102958994A (en) | 2010-04-14 | 2013-03-06 | 陶氏环球技术有限责任公司 | Fuel containers made from polyethylene compositions with improved creep resistance |
| CN102985476B (en) * | 2010-05-27 | 2015-08-19 | 陶氏环球技术有限责任公司 | Polymer composition, its preparation method and the goods prepared by said composition |
| US8269107B2 (en) | 2010-05-28 | 2012-09-18 | General Cable Technologies Corporation | Halogen-free flame retardant polyolefin |
| BR112013012819B1 (en) * | 2010-11-24 | 2021-09-14 | Dow Global Technologies Llc | TOP LAYER OF AN ARTIFICIAL LEATHER MULTILAYER STRUCTURE, ARTIFICIAL LEATHER MULTILAYER STRUCTURE, ARTICLE AND PROCESS TO PREPARE A COMPOSITION |
| BRPI1107080A2 (en) | 2010-12-30 | 2013-04-24 | Braskem Sa | article forming by blowing and compression |
| US8801049B2 (en) | 2011-04-29 | 2014-08-12 | Springseal, Inc. | Pipe coupling system and method |
| KR102127083B1 (en) * | 2012-08-29 | 2020-06-25 | 다우 글로벌 테크놀로지스 엘엘씨 | Ethylene-based polymer compositions and foams |
| JP5711713B2 (en) * | 2012-10-01 | 2015-05-07 | 住友電気工業株式会社 | Multi-layer heat recovery article |
| US9087629B2 (en) | 2012-12-13 | 2015-07-21 | General Cable Technologies Corporation | Fire and water resistant cable |
| US9115274B2 (en) | 2012-12-13 | 2015-08-25 | General Cable Technologies Corporation | Fire and water resistant cable cover |
| JP6002027B2 (en) | 2012-12-20 | 2016-10-05 | 住友電気工業株式会社 | Multi-layer heat recovery article, wire splice and wire harness |
| JP5651161B2 (en) | 2012-12-20 | 2015-01-07 | 住友電気工業株式会社 | Multi-layer heat recovery article, wire splice and wire harness |
| ES2688532T3 (en) | 2013-01-18 | 2018-11-05 | Basf Se | Acrylic dispersion based coating compositions |
| KR102367286B1 (en) | 2013-01-18 | 2022-02-24 | 셀가드 엘엘씨 | Surface modifying agents, modified materials and methods |
| MX2015010042A (en) | 2013-02-14 | 2015-10-30 | Dow Global Technologies Llc | Ethylene-based polymer compositions with improved processibility. |
| US10654948B2 (en) | 2013-03-13 | 2020-05-19 | Chevron Phillips Chemical Company Lp | Radically coupled resins and methods of making and using same |
| US10577440B2 (en) | 2013-03-13 | 2020-03-03 | Chevron Phillips Chemical Company Lp | Radically coupled resins and methods of making and using same |
| US10329071B2 (en) * | 2013-03-15 | 2019-06-25 | Dow Global Technologies Llc | EPDM packaging system and process |
| MX363375B (en) * | 2013-05-02 | 2019-03-21 | Dow Global Technologies Llc | A polyethylene composition and articles made therefrom. |
| KR20210054023A (en) | 2013-05-17 | 2021-05-12 | 밀텍 코포레이션 | Actinic and electron beam radiation curable water based electrode binders and electrodes incorporating same |
| US9475928B2 (en) | 2013-06-24 | 2016-10-25 | Dow Global Technologies Llc | Reinforced polypropylene composition |
| KR102034340B1 (en) | 2013-06-24 | 2019-10-18 | 다우 글로벌 테크놀로지스 엘엘씨 | Reinforced polypropylene composition |
| KR101684648B1 (en) * | 2014-04-21 | 2016-12-08 | 주식회사 엘지화학 | Elastic diene terpolymer and preparation method thereof |
| WO2014209082A1 (en) | 2013-06-28 | 2014-12-31 | 주식회사 엘지화학 | Trinary elastic copolymer comprising diene and method for preparing same |
| KR101585204B1 (en) | 2013-06-28 | 2016-01-13 | 주식회사 엘지화학 | Elastic diene terpolymer and preparation method thereof |
| US9493593B2 (en) | 2013-06-28 | 2016-11-15 | Lg Chem, Ltd. | Elastic diene terpolymer and preparation method thereof |
| US9650460B2 (en) | 2013-06-28 | 2017-05-16 | Lg Chem, Ltd. | Elastic diene terpolymer and preparation method thereof |
| US9777206B2 (en) | 2013-12-10 | 2017-10-03 | General Cable Technologies Corporation | Thermally conductive compositions and cables thereof |
| KR20160119079A (en) | 2014-02-07 | 2016-10-12 | 제너럴 케이블 테크놀로지즈 코오포레이션 | Methods of forming cables with improved coverings |
| JP6694390B2 (en) | 2014-02-24 | 2020-05-13 | ダウ グローバル テクノロジーズ エルエルシー | Thermoplastic polyolefin with reduced gloss for non-carpet floors |
| KR101783897B1 (en) | 2014-12-10 | 2017-10-10 | 주식회사 엘지화학 | Polyolefin pellet for preparing fiber and fiber comprising the same |
| WO2016109560A1 (en) | 2014-12-30 | 2016-07-07 | General Cable Technologies Corporation | Multi-layer cables |
| US10183924B2 (en) | 2015-12-30 | 2019-01-22 | Dow Global Technologies Llc | Sulfonylazide derivative for diene polymer |
| TWI750182B (en) * | 2016-06-23 | 2021-12-21 | 美商陶氏全球科技有限責任公司 | Aliphatic sulfonyl azide anhydride for tie layer |
| TWI746568B (en) * | 2016-06-23 | 2021-11-21 | 美商陶氏全球科技有限責任公司 | Production of sulfonyl azide anhydride |
| DE102016121554A1 (en) * | 2016-11-10 | 2018-05-17 | Wobben Properties Gmbh | Multilayer composite component |
| WO2018191713A1 (en) | 2017-04-13 | 2018-10-18 | Braskem America, Inc. | Azide-modified olefin as polymeric coupling agent |
| WO2018195756A1 (en) * | 2017-04-25 | 2018-11-01 | 南通大学 | Test tube stand and receiving plate unit with magnetic base used for super clean bench |
| US11584844B2 (en) | 2017-12-29 | 2023-02-21 | Dow Global Technologies Llc | Process for preparing crosslinked polyolefin particles |
| KR20200103669A (en) | 2017-12-29 | 2020-09-02 | 다우 글로벌 테크놀로지스 엘엘씨 | How to modify a polycarbonate blend |
| JP7305642B2 (en) | 2017-12-29 | 2023-07-10 | ダウ グローバル テクノロジーズ エルエルシー | Polyolefin-acrylic particles |
| KR102295069B1 (en) * | 2018-08-17 | 2021-08-26 | 주식회사 엘지화학 | Separator for electrochemical device and manufacturing method thereof |
| KR102474477B1 (en) * | 2018-11-29 | 2022-12-05 | 롯데케미칼 주식회사 | Polyolefin composition having imporoved stress resistance |
| US11073214B2 (en) * | 2019-03-29 | 2021-07-27 | Rapak, Llc | Duckbill valve and method for making a duckbill valve |
| SG11202112115XA (en) | 2019-05-30 | 2021-12-30 | Dow Global Technologies Llc | Method of extruding linear low-density polyethylene (lldpe) without surface fractures in the melt |
| FR3099766B1 (en) | 2019-08-07 | 2021-07-30 | Michelin & Cie | Ethylene-rich diene block polymer having a statistical block and a polyethylene block. |
| FR3116536B1 (en) | 2020-11-26 | 2022-11-25 | Michelin & Cie | An ethylene-rich diene triblock polymer having a random block and two polyethylene terminal blocks. |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3203936A (en) * | 1961-03-15 | 1965-08-31 | Hercules Powder Co Ltd | Cross-linking ethylene polymers |
| DE1569025A1 (en) * | 1964-03-13 | 1970-07-09 | Hercules Inc | Improvements in or relating to polymers containing bridge bonds |
| US5118531A (en) * | 1987-12-18 | 1992-06-02 | Uniroyal Chemical Company, Inc. | Pumpability sealant composition |
| EP0702032A2 (en) * | 1994-09-19 | 1996-03-20 | Sentinel Products Corp. | Cross-linked foam structures of essentially linear polyolefines and process for manufacture |
Family Cites Families (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA797917A (en) | 1968-10-29 | Hercules Incorporated | Bridged polymers | |
| NL282628A (en) | 1961-03-15 | |||
| US3203937A (en) | 1961-03-15 | 1965-08-31 | Hercules Powder Co Ltd | Cross-linking ethylene polymers |
| US3282864A (en) | 1963-05-31 | 1966-11-01 | Phillips Petroleum Co | Di(sulfonylazides) as crosslinking agents for hydrocarbon polymers |
| US3336268A (en) | 1963-10-14 | 1967-08-15 | Hercules Inc | Bridged polypropylene |
| US3336258A (en) | 1963-11-26 | 1967-08-15 | Du Pont | Aromatic polyamide-acids cross-linked with ditertiary diamines |
| US3530108A (en) | 1964-06-12 | 1970-09-22 | Hercules Inc | Elastic polypropylene film and process |
| US3341480A (en) | 1964-07-02 | 1967-09-12 | Hercules Inc | Cellular polyolefin |
| FR1478692A (en) | 1965-04-15 | 1967-07-12 | ||
| US3298975A (en) | 1966-03-18 | 1967-01-17 | Hercules Inc | Expandable partially-foamed and cured polypropylene composition |
| US3855184A (en) | 1971-01-27 | 1974-12-17 | E Bostick | Crosslinking of aromatic polymers with organic polysulfonazides |
| NL7207689A (en) | 1972-06-07 | 1973-12-11 | ||
| FR2245702B1 (en) * | 1973-08-24 | 1976-05-07 | Rhone Poulenc Ind | |
| JPS5744456B2 (en) | 1974-04-10 | 1982-09-21 | ||
| DE2911719C2 (en) | 1979-03-24 | 1982-07-01 | Fa. Carl Freudenberg, 6940 Weinheim | Process for the production of lightweight foams from thermoplastic crystalline plastics |
| US4579905A (en) | 1983-08-15 | 1986-04-01 | General Electric Company | Graftlinked polymers and process for making |
| US4714716A (en) | 1984-11-16 | 1987-12-22 | The Dow Chemical Company | Lightly crosslinked linear olefinic polymer foams and process for making |
| US4694025A (en) | 1984-11-16 | 1987-09-15 | The Dow Chemical Company | Alcohol control of lightly crosslinked foamed polymer production |
| GB2205103A (en) | 1987-05-21 | 1988-11-30 | Shell Int Research | Modified block copolymers and process for the preparation thereof |
| US5037895A (en) | 1988-08-04 | 1991-08-06 | Phillips Petroleum Company | Process for improving thermoplastic hydrocarbon polymers with polysulfonazide and free radical catalyst |
| US5594078A (en) | 1991-07-23 | 1997-01-14 | Phillips Petroleum Company | Process for producing broad molecular weight polyolefin |
| US5498581A (en) | 1994-06-01 | 1996-03-12 | Phillips Petroleum Company | Method for making and using a supported metallocene catalyst system |
| ES2139926T3 (en) * | 1994-09-02 | 2000-02-16 | Dow Chemical Co | THERMOSTABLE ELASTOMERS. |
| US5869591A (en) * | 1994-09-02 | 1999-02-09 | The Dow Chemical Company | Thermoset interpolymers and foams |
| US5654454A (en) | 1995-05-30 | 1997-08-05 | Phillips Petroleum Company | Metallocene preparation and use |
| AR016890A1 (en) * | 1997-08-27 | 2001-08-01 | Dow Chemical Co | PROCEDURE FOR THE PREPARATION OF COUPLED MIXTURES OF ALPHA-OLEFINE / ALPHATIC VINYL OR MONOMERO INTERPOLIMEROS OF ALPHATICOIMPED VINYL MONOMERO WITH POLYOLEFINS, COMPOSITIONS, FOAM ARTICLES AND PROCESS FOR THE FORMATION OF AN ARTICLE |
| US6376623B1 (en) * | 1997-08-27 | 2002-04-23 | The Dow Chemical Company | Rheology modification of elastomers |
| DE19941841A1 (en) | 1999-09-02 | 2001-03-08 | Claas Selbstfahr Erntemasch | Self-propelled agricultural machine |
| US6291618B1 (en) * | 1999-12-23 | 2001-09-18 | The Dow Chemical Company | Phosphazene azide coupling agents and their use in rheology modification of polymers |
-
1998
- 1998-08-05 US US09/129,161 patent/US6376623B1/en not_active Expired - Lifetime
- 1998-08-05 US US09/129,163 patent/US6521306B1/en not_active Expired - Lifetime
- 1998-08-05 AT AT98938337T patent/ATE306517T1/en active
- 1998-08-05 EP EP19980938336 patent/EP1007585A1/en not_active Withdrawn
- 1998-08-05 KR KR1020007002005A patent/KR100562616B1/en not_active IP Right Cessation
- 1998-08-05 JP JP2000507740A patent/JP2001514287A/en active Pending
- 1998-08-05 AU AU86881/98A patent/AU743240B2/en not_active Ceased
- 1998-08-05 ES ES98938337T patent/ES2247709T3/en not_active Expired - Lifetime
- 1998-08-05 DE DE1998634123 patent/DE69834123T2/en not_active Expired - Lifetime
- 1998-08-05 KR KR1020007002004A patent/KR20010023366A/en not_active Application Discontinuation
- 1998-08-05 AT AT98938338T patent/ATE322519T1/en not_active IP Right Cessation
- 1998-08-05 EP EP98938337A patent/EP1007586B1/en not_active Expired - Lifetime
- 1998-08-05 KR KR1020007002009A patent/KR100562615B1/en not_active IP Right Cessation
- 1998-08-05 DE DE1998631871 patent/DE69831871T2/en not_active Expired - Lifetime
- 1998-08-05 BR BR9814447A patent/BR9814447A/en not_active Application Discontinuation
- 1998-08-05 US US09/129,155 patent/US6211302B1/en not_active Expired - Fee Related
- 1998-08-05 WO PCT/US1998/016214 patent/WO1999010422A1/en active IP Right Grant
- 1998-08-05 JP JP2000507741A patent/JP2001514288A/en active Pending
- 1998-08-05 CA CA 2301644 patent/CA2301644C/en not_active Expired - Fee Related
- 1998-08-05 CA CA 2301602 patent/CA2301602A1/en not_active Abandoned
- 1998-08-05 CN CN98809860A patent/CN1273592A/en active Pending
- 1998-08-05 EP EP19980938338 patent/EP1007587B1/en not_active Expired - Lifetime
- 1998-08-05 AU AU86883/98A patent/AU8688398A/en not_active Abandoned
- 1998-08-05 JP JP2000507742A patent/JP4344471B2/en not_active Expired - Fee Related
- 1998-08-05 CA CA 2301836 patent/CA2301836C/en not_active Expired - Fee Related
- 1998-08-05 WO PCT/US1998/016213 patent/WO1999010421A1/en not_active Application Discontinuation
- 1998-08-05 TR TR200000905T patent/TR200000905T2/en unknown
- 1998-08-05 AU AU86882/98A patent/AU8688298A/en not_active Abandoned
- 1998-08-05 WO PCT/US1998/016215 patent/WO1999010423A1/en active IP Right Grant
- 1998-08-13 CN CN98810324A patent/CN1276811A/en active Pending
- 1998-08-13 AT AT98939934T patent/ATE239048T1/en not_active IP Right Cessation
- 1998-08-13 JP JP2000507736A patent/JP2001514284A/en active Pending
- 1998-08-13 BR BR9814441A patent/BR9814441A/en not_active IP Right Cessation
- 1998-08-13 US US09/133,244 patent/US6143829A/en not_active Expired - Lifetime
- 1998-08-13 AU AU88283/98A patent/AU8828398A/en not_active Abandoned
- 1998-08-13 EP EP19980939934 patent/EP1007583B1/en not_active Expired - Lifetime
- 1998-08-13 DE DE1998614098 patent/DE69814098T2/en not_active Expired - Lifetime
- 1998-08-13 ES ES98939934T patent/ES2192783T3/en not_active Expired - Lifetime
- 1998-08-13 KR KR1020007001979A patent/KR20010023341A/en not_active Application Discontinuation
- 1998-08-13 CA CA 2301527 patent/CA2301527A1/en not_active Abandoned
- 1998-08-13 WO PCT/US1998/016810 patent/WO1999010415A1/en not_active Application Discontinuation
- 1998-08-25 MY MYPI98003863A patent/MY133100A/en unknown
- 1998-08-25 MY MYPI98003864A patent/MY121755A/en unknown
- 1998-08-25 MY MYPI98003868A patent/MY128986A/en unknown
- 1998-08-26 EP EP19980941064 patent/EP1007590A1/en not_active Withdrawn
- 1998-08-26 PL PL33908398A patent/PL339083A1/en unknown
- 1998-08-26 ZA ZA9807752A patent/ZA987752B/en unknown
- 1998-08-26 ZA ZA9807753A patent/ZA987753B/en unknown
- 1998-08-26 AU AU89210/98A patent/AU743302B2/en not_active Ceased
- 1998-08-26 ID ID20000351A patent/ID25634A/en unknown
- 1998-08-26 AR ARP980104237 patent/AR016885A1/en unknown
- 1998-08-26 AR ARP980104238 patent/AR016886A1/en unknown
- 1998-08-26 ZA ZA9807745A patent/ZA987745B/en unknown
- 1998-08-26 CA CA 2301835 patent/CA2301835C/en not_active Expired - Lifetime
- 1998-08-26 ZA ZA9807750A patent/ZA987750B/en unknown
- 1998-08-26 HU HU0002756A patent/HUP0002756A3/en unknown
- 1998-08-26 AR ARP980104240 patent/AR016888A1/en unknown
- 1998-08-26 WO PCT/US1998/017685 patent/WO1999010427A1/en active IP Right Grant
- 1998-08-26 AR ARP980104241 patent/AR016889A1/en unknown
- 1998-08-26 KR KR10-2000-7001910A patent/KR100535582B1/en not_active IP Right Cessation
- 1998-08-26 CN CNB988094347A patent/CN1166730C/en not_active Expired - Lifetime
- 1998-08-26 US US09/140,603 patent/US6528136B1/en not_active Expired - Lifetime
- 1998-08-26 TR TR200001175T patent/TR200001175T2/en unknown
- 1998-08-26 BR BR9814442A patent/BR9814442A/en not_active Application Discontinuation
- 1998-08-26 AR ARP980104239 patent/AR016887A1/en unknown
- 1998-08-26 JP JP2000507746A patent/JP4342721B2/en not_active Expired - Lifetime
- 1998-08-26 ZA ZA9807746A patent/ZA987746B/en unknown
- 1998-08-26 MY MYPI98003897A patent/MY126503A/en unknown
- 1998-09-29 TW TW87114112A patent/TW591070B/en active
- 1998-09-29 TW TW87114107A patent/TW584641B/en active
- 1998-09-29 TW TW87114109A patent/TW588059B/en active
- 1998-10-01 TW TW87114111A patent/TW584652B/en active
-
2000
- 2000-02-25 NO NO20000966A patent/NO322848B1/en unknown
- 2000-09-29 TW TW91132924A patent/TW200300428A/en unknown
- 2000-10-23 US US09/694,649 patent/US6359073B1/en not_active Expired - Lifetime
-
2001
- 2001-10-29 US US10/021,134 patent/US6506848B2/en not_active Expired - Lifetime
- 2001-12-10 US US10/013,152 patent/US6552129B2/en not_active Expired - Lifetime
-
2003
- 2003-01-21 US US10/347,713 patent/US6777502B2/en not_active Expired - Lifetime
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3203936A (en) * | 1961-03-15 | 1965-08-31 | Hercules Powder Co Ltd | Cross-linking ethylene polymers |
| DE1569025A1 (en) * | 1964-03-13 | 1970-07-09 | Hercules Inc | Improvements in or relating to polymers containing bridge bonds |
| US5118531A (en) * | 1987-12-18 | 1992-06-02 | Uniroyal Chemical Company, Inc. | Pumpability sealant composition |
| EP0702032A2 (en) * | 1994-09-19 | 1996-03-20 | Sentinel Products Corp. | Cross-linked foam structures of essentially linear polyolefines and process for manufacture |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014145082A (en) * | 1998-11-02 | 2014-08-14 | Dow Global Technologies Llc | SHEAR THINNING ETHYLENE/α-OLEFIN INTERPOLYMERS AND THEIR PREPARATION METHOD |
| US8883870B2 (en) | 2010-09-15 | 2014-11-11 | Dow Global Technologies Llc | Propylene-alpha-olefin copolymer compositions with improved foaming window |
| US10471690B2 (en) | 2014-12-30 | 2019-11-12 | Dow Global Technologies Llc | Sulfonylazide derivative for tie layer |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6528136B1 (en) | Rheology modification of polymers prepared using metallocenes | |
| MXPA00002017A (en) | Rheology modification of polymers prepared using metallocenes | |
| CZ2000710A3 (en) | Modification process of polymer rheology that are prepared by employing metallocenes | |
| MXPA00002008A (en) | Rheology modification of interpolymers of alpha-olefins and vinyl aromatic monomers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 98809434.7 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH GM HR HU ID IL IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 89210/98 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2301835 Country of ref document: CA Ref document number: 2301835 Country of ref document: CA Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2000 507746 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2000/002017 Country of ref document: MX Ref document number: 1020007001910 Country of ref document: KR Ref document number: PV2000-710 Country of ref document: CZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1998941064 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000/01175 Country of ref document: TR |
|
| WWP | Wipo information: published in national office |
Ref document number: 1998941064 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: PV2000-710 Country of ref document: CZ |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020007001910 Country of ref document: KR |
|
| WWR | Wipo information: refused in national office |
Ref document number: PV2000-710 Country of ref document: CZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 89210/98 Country of ref document: AU |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1998941064 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1020007001910 Country of ref document: KR |