WO2001027099A2 - Bicyclic compounds composition and method for stabilizing the same - Google Patents
Bicyclic compounds composition and method for stabilizing the same Download PDFInfo
- Publication number
- WO2001027099A2 WO2001027099A2 PCT/JP2000/007109 JP0007109W WO0127099A2 WO 2001027099 A2 WO2001027099 A2 WO 2001027099A2 JP 0007109 W JP0007109 W JP 0007109W WO 0127099 A2 WO0127099 A2 WO 0127099A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- hydroxy
- alkyl
- aryl
- glyceride
- composition
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4858—Organic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/351—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom not condensed with another ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/08—Bronchodilators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/738—Esters of keto-carboxylic acids or aldehydo-carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D221/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00
- C07D221/02—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00 condensed with carbocyclic rings or ring systems
- C07D221/04—Ortho- or peri-condensed ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D311/94—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems condensed with rings other than six-membered or with ring systems containing such rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/78—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems condensed with rings other than six-membered or with ring systems containing such rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D335/00—Heterocyclic compounds containing six-membered rings having one sulfur atom as the only ring hetero atom
- C07D335/04—Heterocyclic compounds containing six-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
Definitions
- the present invention relates to a novel composition comprising a novel bi-cyclic compound and a glyceride, and a method for stabilizing the bi-cyclic compound comprising the step admixing the same with a glyceride.
- a glyceride has been applied widely m the medical field and is useful as an immediate alimentation or an entero-protectmg agent ( JP-A-4-210631 ) .
- it is also useful as a solvent for various pharmaceutically active compounds such as active vitamin Ds, diazepam, thiazole derivatives, prostaglandms or flavonoids, as a diluent for a capsule preparation, as a vehicle of eye drop, and as a stabilizing agent (JP-A-53- 50141, JP-A-53-75320, US 4,248,867, JP-A-55-136219, US 4,247,702, JP-A-59-137413, JP-A-02-204417 , JP-A-04-46122, US 5,411,952, US 5,474,979 and US 5,981,607) .
- the prior arts are silent on the effect of glyce ⁇ des on the novel pharmaceutically active bi- cyclic compounds.
- the object of the present invention is to provide a novel composition comprising a certain bi-cyclic compound having a pharmacological activity and a glyceride, and a method for stabilizing the bi-cyclic compound by admixing the same with a glyceride.
- Another object of the present invention is to provide a novel compound having a pharmacological activity.
- This inventor studied to improve the stability of a novel bi-cyclic compound and found that a composition comprising the bi-cyclic compound and a glyceride can attain the above object.
- the present invention provides a novel composition
- A is -CH 2 OH, -COCH 2 OH, -COOH or a functional derivative thereof;
- X 2 and X are hydrogen atom, lower alkyl or halogen atom
- V x and V 2 are carbon or oxygen atoms
- Z is a carbon, oxygen, sulfur or nitrogen atom
- R ⁇ is a saturated or unsaturated bivalent lower- medium aliphatic hydrocarbon residue which is unsubstituted or substituted with halogen, an alkyl group, hydroxy, oxo, aryl or heterocyclic group;
- R 2 is a saturated or unsaturated, lower or medium aliphatic hydrocarbon residue which is unsubstituted or substituted with halogen atom, oxo, hydroxy, lower alkyl, lower alkoxy, lower alkanoyloxy, lower cycloalkyl, lower cycloalkyloxy, aryl, aryloxy, heterocyclic group or heterocyclic-oxy group; lower cycloalkyl; lower cycloalkyloxy; aryl, aryloxy, heterocyclic group or heterocyclic-oxy group;
- R 3 is a hydrogen atom, a lower alkyl, lower cycloalkyl, aryl or heterocyclic group; and a glyceride, and a method for stabilizing the above- specified bi-cyclic compound by means of dissolving said compound in a glyceride.
- the present invention also provides a novel bi- cycl c compound represented by the above formula (I).
- the term "unsaturated" in the definitions for R and R is intended to include at least one or more double bonds and/or triple bonds that are isolatedly, separately or serially present between carbon atoms of the main and/or side chains. According to the usual nomenclature, an unsaturated bond between two serial positions is represented by denoting the lower number of the two positions, and an unsaturated bond between two distal positions is represented by denoting both of the positions.
- lower or medium aliphatic hydrocarbon refers to a straight or branched chain hydrocarbon group having 1 to 14 carbon atoms (for a side chain, 1 to 3 carbon atoms are preferable) and preferably 1 to 10, especially 2 to 8 carbon atoms for R x and 1 to 10, especially 1 to 8 carbon atoms for R 2 .
- halogen atom covers fluorine, chlorine, bromine and iodine. Particularly preferable is a fluorine atom.
- lower throughout the specification is intended to include a group having 1 to 6 carbon atoms unless otherwise specified.
- lower alkyl refers to a straight or branched chain saturated hydrocarbon group containing 1 to
- lower alkoxy refers to a group of lower alkyl-O-, wherein lower alkyl is as defined above.
- hydroxy ( lower ) alkyl refers to a lower alkyl as defined above which is substituted with at least one hydroxy group such as hydroxymethyl, 1-hydroxyethyl, 2-hydroxyethyl and 1-methyl-l-hydroxyethyl .
- lower alkanoyloxy refers to a group represented by the formula RCO-0-, wherein RCO- is an acyl group formed by oxidation of a lower alkyl group as defined above, such as acetyl.
- lower cycloalkyl refers to a cyclic group formed by cyclization of a lower alkyl group as defined above but contains three or more carbon atoms, and includes, for example, cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl .
- lower cycloalkyloxy refers to the group of lower-cycloalkyl-O-, wherein lower cycloalkyl is as defined above.
- aryl may include unsubstituted or substituted aromatic hydrocarbon rings (preferably monocyclic groups), for example, phenyl, naphthyl, tolyl, xylyl. Examples of the substituents are halogen atom and halo (lower ) alkyl, wherein halogen atom and lower alkyl are as defined above.
- aryloxy refers to a group represented by the formula ArO-, wherein Ar is aryl as defined above.
- heterocyclic group may include mono- to tri-cyclic, preferably monocyclic heterocyclic group which is 5 to 14, preferably 5 to 10 membered ring having optionally substituted carbon atom and 1 to 4, preferably 1 to 3 of 1 or 2 type of hetero atoms selected from nitrogen atom, oxygen atom and sulfer atom.
- heterocyclic group examples include furyl, thienyl, pyrrolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, lmidazolyl, pyrazolyl, furazanyl, pyranyl, py ⁇ dyl, pyridazmyl, pyrimidyl, pyrazmyl, 2-pyrrolmyl , pyrrolidmyl, 2- lmidazolmyl, lmidazolidmyl, 2-pyrazolmyl, pyrazolidmyl, pipe ⁇ dmo, piperazmyl, morpholmo, mdolyl, benzothienyl, qumolyl, isoquinolyl, purmyl, qumazolmyl, carbazolyl, acridmyl, phenanthridmyl, benzimidazolyl, benzimidazolonyl, benzothiazolyl, phenothia
- substituent m examples include halogen, and halogen substituted lower alkyl group, wherein halogen atom and lower alkyl group are as described above.
- heterocyclic-oxy group means a group represented by the formula HcO-, wherein He is a heterocyclic group as described above.
- the term "functional derivative" of A includes salts (preferably pharmaceutically acceptable salts), ethers, esters and amides.
- Suitable "pharmaceutically acceptable salts” include conventionally used non-toxic salts, for example a salt with an inorganic base such as an alkali metal salt (such as sodium salt and potassium salt), an alkaline earth metal salt (such as calcium salt and magnesium salt), an ammonium salt; or a salt with an organic base, for example, an amine salt (such as methylamine salt, dimethylamme salt, cyclohexylamine salt, benzylamme salt, pipe ⁇ dme salt, ethylenediamme salt, ethanolamme salt, diethanolamme salt, t ⁇ ethanolamine salt, tris (hydroxymethylammo) ethane salt, monomethyl- monoethanolamine salt, procaine salt and caffeine salt), a basic ammo ac d salt (such as arginine salt and lysme salt), tetraalkyl ammonium salt and the like. These salts may be prepared by a conventional process, for example from the corresponding acid and base or by salt interchange .
- ethers examples include alkyl ethers, for example, lower alkyl ethers such as methyl ether, ethyl ether, propyl ether, isopropyl ether, butyl ether, isobutyl ether, t-butyl ether, pentyl ether and 1- cyclopropyl ethyl ether; and medium or higher alkyl ethers such as octyl ether, diethylhexyl ether, lauryl ether and cetyl ether; unsaturated ethers such as oleyl ether and linolenyl ether; lower alkenyl ethers such as vinyl ether, allyl ether; lower alkynyl ethers such as ethynyl ether and propynyl ether; hydroxy (lower ) alkyl ethers such as hydroxyethyl ether and hydroxyisopropyl ether; lower alkoxy
- esters examples include aliphatic esters, for example, lower alkyl esters such as methyl ester, ethyl ester, propyl ester, isopropyl ester, butyl ester, isobutyl ester, t-butyl ester, pentyl ester and 1- cyclopropylethyl ester; lower alkenyl esters such as vinyl ester and allyl ester; lower alkynyl esters such as ethynyl ester and propynyl ester; hydroxy (lower) alkyl ester such as hydroxyethyl ester; lower alkoxy (lower) alkyl esters such as methoxymethyl ester and 1- methoxyethyl ester; and optionally substituted aryl esters such as, for example, phenyl ester, tosyl ester, t- butylphenyl ester, salicyl ester, 3, 4-di-methoxyphenyl est
- amides are mono- or di-lower alkyl amides such as methylamide, ethylamide and dimethylamide; arylamides such as anilide and toluidide; and alkyl- or aryl-sulfonylamides such as methylsulfonylamide, ethylsulfonyl-amide and tolylsulfonylamide .
- Preferred A is -COOH, -CH 2 OH, or its pharmaceutically acceptable salt, ester, ether or amide.
- Preferred combination of X ⁇ and X 2 is that at least one of X ⁇ and X 2 is halogen atom, and more preferably, both of them are halogen, especially fluorine atoms.
- Preferred x 0, or where one of R 4 and R 5 is hydrogen, another is hydroxy,
- Preferred W 2 is where R 4 and R 5 are both hydrogen atoms
- Preferred Z is an oxygen atom.
- Preferred R x is an unsubstituted saturated or unsaturated bivalent lower-medium aliphatic hydrocarbon residue. It may preferably have 1-10 carbon atoms, more preferably, 2-8 carbon atoms.
- Preferred R 2 is a saturated or unsaturated bivalent lower-medium aliphatic hydrocarbon residue. It may preferably have 1-10 carbon atoms, more preferably, 1- 8 carbon atoms .
- Preferred R 3 is a hydrogen atom.
- the bi-cyclic compounds according to the present invention encompass not only the compounds represented by the above formula (I) but also optic isomers, steric isomers, and tautomeric isomers thereof.
- Tautomer II a bi-cyclic compound having the formula as shown below (Tautomer II) may be in equilibrium with its tautomeric isomer, 13, 14-dihydro-15- keto-prostaglandin compound (tautomer I) (USP 5,166,174, USP 5,225,439, USP 5,284,858, USP 5,380,709, USP 5,428,062 and US 5,886,034, these cited references are herein incorporated by reference.)
- Tautomer II Tautomer II
- the tautomeric compounds as above exist predominantly in the form of the bi-cyclic compound.
- hydrogen bonding occurs between the water molecule and, for example, the keto group at the hydrocarbon chain, thereby hindering bi- cyclic ring formation.
- the halogen atom(s) at X 1 and/or X 2 promote bi-cyclic ring formation, such as the compound 1 or 2 below.
- the bi- cyclic/mono-cyclic structures may be present in a ratio of 6:1 in D 2 0; 10:1 in CD 3 OD-D 2 0 and 96:4 in CDC1 3 .
- a preferable embodiment of the present invention is the composition in which the bi-cyclic form is present in ratio of bi-cyclic/mono-cyclic of at least 50:50, preferably 90:10, or even greater to substantially all bi-cyclic compound; 100 % bi-cyclic compound is within this invention.
- Preferred embodiment of the compound of the present invention include the Compounds 1 and 2 shown below: Compound 1 :
- the compounds of the present invention possess some pharmacological activities such as bronchodialator.
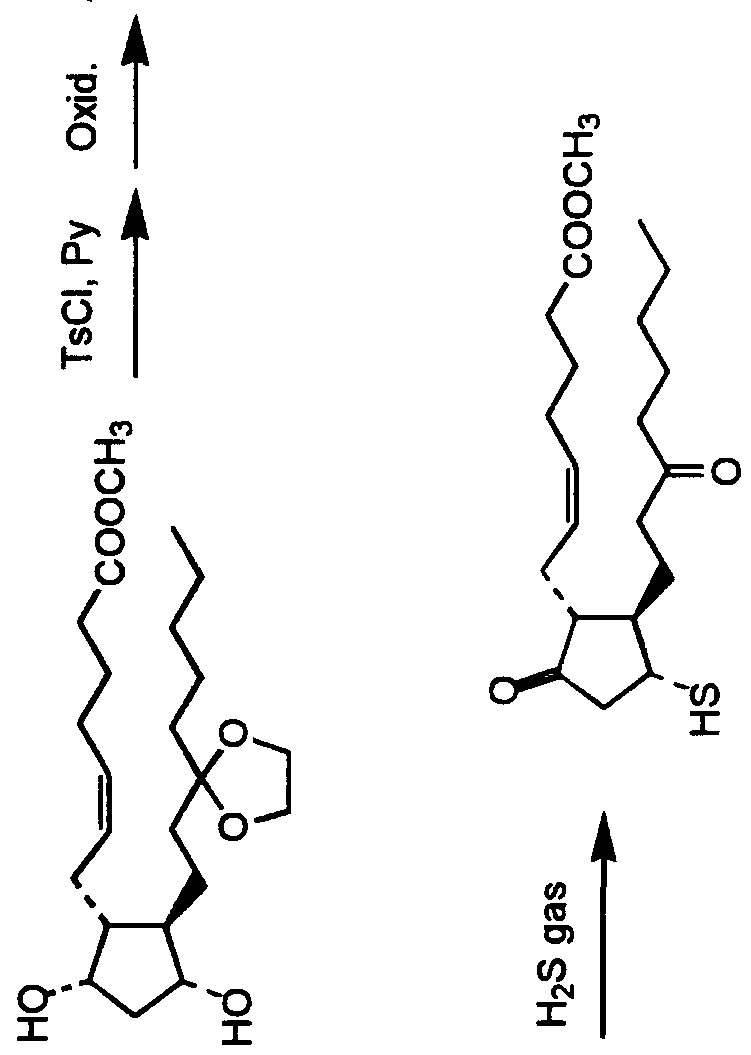
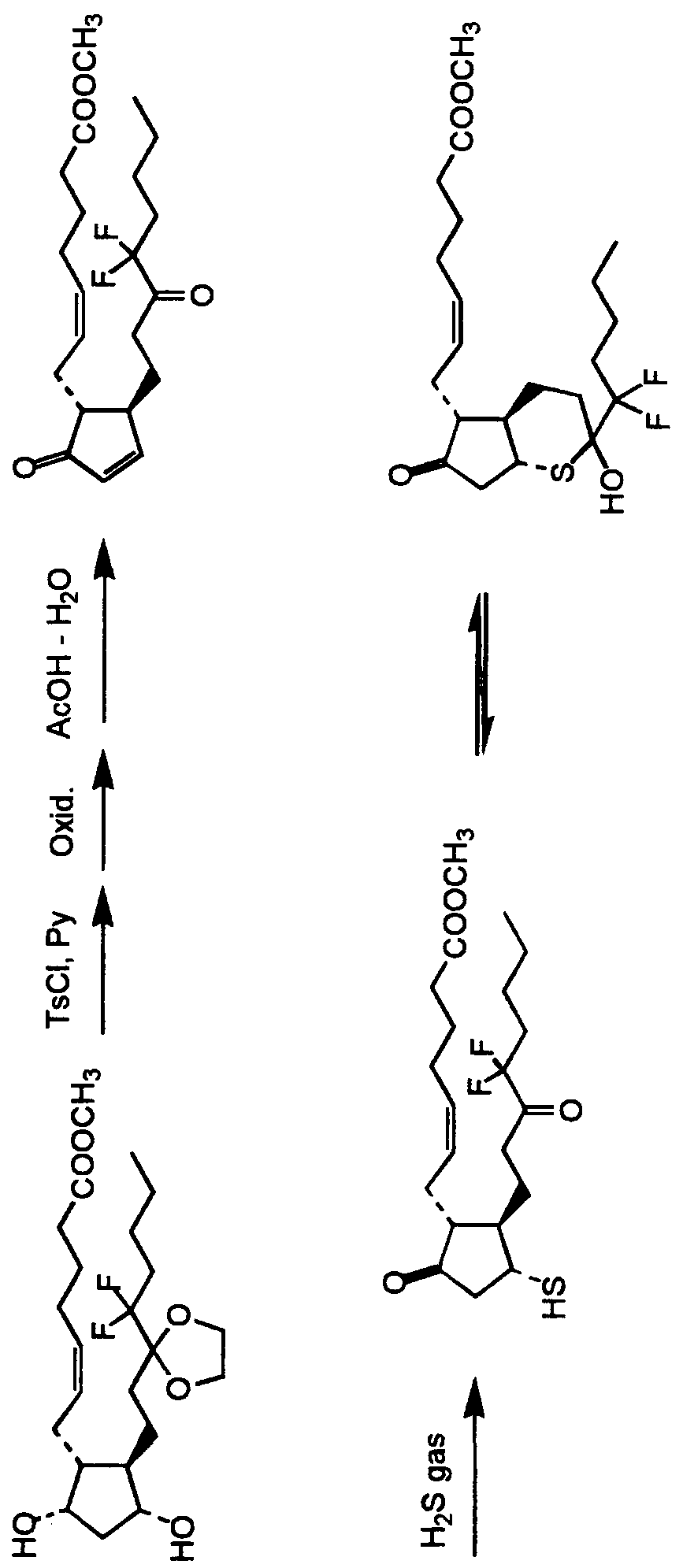
- the above described bi-cyclic compound may prepared according to the general process set forth below: Preparation of Isopropyl 7- [ ( IS, 3S, 6S, 7R) -3-heptyl-3- hydroxy-bi-cyclo [4.3.0] nonane-8-one-7-yl] hept-5-enoate and Isopropyl 7- [IS, 3R, 6S, 7R] -3-heptyl-3-hydroxy-bicyclo [4.3.0] nonane-8-one-7-yl] hept-5-enoate
- Isopropyl 7- (IR, 2R, 3R) -2- (3, 3-ethylenedioxydecyl) -3- iodemethyl-5-oxocyclopentyl] hept-5-enoate (6) (0.16g) was dissolved in a mixed solvent of acetic acid/water/tetrahydrofuran (3/1/1). The mixture was stirred for 20 hours at room temperature and for 2.5 hours at 50°C.
- the preparations in the present invention are not construed to be limited to them, and suitable means for protection, oxidation, reduction and the like may be employed.
- composition of the present invention comprises the above described bi-cyclic compound and a glyceride.
- examples of the glyceride used in the present invention include a glyceride of a saturated or unsaturated fatty acid which may have a branched chain.
- Preferred fatty acid is a medium chain or higher chain fatty acid having at least C6, preferably C6-24 carbon atoms, for example caproic acid (C6) , caprylic ac ⁇ d(C8), capric ac ⁇ d(ClO), lauric ac ⁇ d(C12) and my ⁇ stic acid (C14), palmitic ac ⁇ d(Cl ⁇ ), palmitoleic ac ⁇ d(C16), stea ⁇ c ac ⁇ d(C18), oleic ac ⁇ d(C18), linoleic ac ⁇ d(C18), linolenic ac ⁇ d(C18), ricmolic ac ⁇ d(C18) and arachic ac ⁇ d(C20).
- 2 or more glycerides may be used as a mixture.
- Examples of the mixture of glycerides are mixture of caprylic acid triglyceride and capric acid triglyceride, vegetable oils such as castor oil, corn oil, olive oil, sesame oil, rape oil, salad oil, cottonseed oil, camellia oil, peanut o l, palm oil, sunflower oil.
- vegetable oils such as castor oil, corn oil, olive oil, sesame oil, rape oil, salad oil, cottonseed oil, camellia oil, peanut o l, palm oil, sunflower oil.
- the composition of the present invention may be generally prepared by dissolving or admixing the above- disclosed bi-cyclic compound m the glyceride.
- each of them may be dissolved in a solvent m which both of them are soluble respectively, and then the solutions may be combined.
- the solvent may be removed under vacuum.
- the amount of the glyceride relative to that of the bi-cyclic compound is not limited in so far as the object of the invention, that is, stabilization of the bi-cyclic compound is attained.
- 1-5,000,000 parts by weight, preferably, 5-1,000,000 parts by weight, and more preferably, 10-500,000 parts by weight of the glyceride may be employed per one part by weight of the bi-cyclic compound.
- composition of the present invention may comprise the other oil solvent.
- the other oil solvents may include mineral oils such as liquid paraffin and light liquid paraffin, tocopherol, and the like.
- the ratio of the glycerides to the other oil solvent is not limited.
- the glycerides may present in an amount that improve at least the stability of the bicyclic composition of the present invention.
- the ratio of the glycerides m total o l solvent is at least lv/v%, preferably not more than 5v/v% .
- the composition of the present invention is substantially free of water.
- substantially free of water means that the composition does not contain water that is intentionally added. It is understood that many materials contain water that is taken up from the atmosphere or is present as a coordination complex in its normal state. Water taken up by hygroscopic materials or present as a hydrate is permissibly present in the compositions of this embodiment. According to the embodiment, any water that is present in the composition should not be present in amounts such that the water will have a deleterious effect to the composition of the present invention.
- composition of the present invention may further contain physiologically acceptable additives which do not provide adverse effect to the stability of the compound of the formula (I) .
- the additives which may be employed in the present invention include, but not limited to, excipients, diluents, fillers, solvents, lubricants, adjuvants, binders, disintegrants, coatings, capuslating agents, ointment bases, suppository base, aerozoles, emulsifiers, dispersing agents, suspensions, viscosity increasing agents, isotonic agents, buffers, analgesic agents, preservatives, anti-oxidants, corrigents, flavors, colorants, and functional agents such as cyclodextrin, biologically degradable polymers.
- the details of the additives may be selected from those described in any of general textbooks in the pharmaceutical field. Further, the composition of the present invention may further contain another pharmaceutically active ingredient.
- composition of the present invention may be formulated by a conventional manner. They may be in the form suitable for oral administration, suppository, injection, or topical administration such as eye drops or ointments. Especially, compositions suitable for oral administration such as capsulated compositions and compositions suitable for topical administration such as eye drops are preferable.
- the above-described compounds 1 and 2 were dissolved in the medium chain fatty acid triglyceride (MCT) 31 at the amount shown in the table 1 below respectively.
- MCT medium chain fatty acid triglyceride
- Each of the solutions was placed in a container made of hard glass and stored at 40 °C.
- the time-course of the content of the compound 1 and 2 in the solutions were determined by HPLC method.
- the medium chain fatty acid triglyceride used herein was a mixture of caprylic acid triglyceride and capric acid triglyceride (85:15) .
- each of the compounds 1 and 2 was placed solely (without being dissolved m the solvent) m the container as above and stored at 40 °C to provide control study.
- the content of the compound was determined as follows (HPLC method) .
- the stored compounds 1 and 2, and standard compounds 1 and 2 were weighed precisely around 0.025g each, and exactly 5 ml aliquots of an internal standard solution were added to the respective weighed compounds. Then the test and standard preparations were obtained by adding acetonit ⁇ le (liquid chromatograph grade) to give the precise total amount of 10 ml each. Each lO ⁇ l of the test and standard preparations was loaded on liquid chromatograph and determined the content of the compound by internal standard method with one point calibration curve .
- an amount of the solution corresponding to 36 ⁇ g of the compound 1 or 2 was weighted precisely. Precisely 1.0 ml of an internal standard solution was added, and then ethyl acetate (liquid chromatograph grade) was added to give the total amount of 10 ml each. Each 0.1 ml of the solution was vacuum concentrated to dryness to give the test preparation. Each 18 mg of the respective standard compounds was weighted precisely and admixed with ethyl acetate (liquid chromatograph grade) to give the total amount of exactly 50 ml each.
- 0.1 ml of fluorescent labeling reagent and 0.85 ml of fluorescent labeling catalyst were added respectively, and the mixture was stirred and reacted at room temperature more than 30 minutes.
- 0.05 ml aliquots of acetonitrile (liquid chromatograph grade) containing 2% acetic acid were added to the reaction mixtures respectively, stirred and then stand for more than 30 minutes to provide test and standard solutions.
- test and standard solutions were loaded on liquid chromatograph and determined the content of the respective compounds by internal standard method with one point calibration curve.
- the above-described compound 1 was dissolved m various solvents at the amount shown in the table 2 below respectively. Each of the solutions was placed in a container made of low-density polyethylene (LDPE) , hard glass or stainless steel and stored at 40 ° C . The content of the compound 1 m the solutions after four weeks were determined by HPLC method according to the above described (2) of Example 1 except for using composition shown m table 2 below. The results are shown in Table 2 below.
- LDPE low-density polyethylene
- MCT mixture of caprylic acid triglyceride and capric acid triglyceride (85 : 15)
Abstract
Description
Claims
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2002112984A RU2695274C3 (en) | 1999-10-15 | 2000-10-13 | NEW COMPOSITIONS AND METHODS FOR THEIR STABILIZATION |
| BRPI0014869A BRPI0014869B8 (en) | 1999-10-15 | 2000-10-13 | bicyclic prostaglandins, their stabilized compositions, as well as a process for their stabilization |
| DK04005836T DK1426361T4 (en) | 1999-10-15 | 2000-10-13 | Novel compounds containing a bi-cyclic compound and a glyceride |
| HU1300369A HU230484B1 (en) | 1999-10-15 | 2000-10-13 | Novel composition containing a bi-cyclic compound and a glyceride |
| AU76856/00A AU780342B2 (en) | 1999-10-15 | 2000-10-13 | Novel composition and method for stabilizing the same |
| AT00966462T ATE267189T1 (en) | 1999-10-15 | 2000-10-13 | BIZYCLIC COMPOUNDS COMPOSITION AND METHOD FOR STABILIZING THEM |
| HU0203746A HU229547B1 (en) | 1999-10-15 | 2000-10-13 | Novel composition and method for stabilizing the same |
| CA002385732A CA2385732C (en) | 1999-10-15 | 2000-10-13 | Bicyclic compounds composition and method for stabilizing the same |
| NZ518020A NZ518020A (en) | 1999-10-15 | 2000-10-13 | Novel composition and method for stabilizing the same |
| MXPA02003756A MXPA02003756A (en) | 1999-10-15 | 2000-10-13 | Bicyclic compounds composition and method for stabilizing the same. |
| JP2001530318A JP4332316B2 (en) | 1999-10-15 | 2000-10-13 | Bicyclic compound composition and method for stabilizing the same |
| IL14880300A IL148803A0 (en) | 1999-10-15 | 2000-10-13 | Novel composition and method for stabilizing the same |
| DE60010913T DE60010913T2 (en) | 1999-10-15 | 2000-10-13 | Bizyclic compounds, composition and process for their stabilization |
| EP00966462A EP1220849B1 (en) | 1999-10-15 | 2000-10-13 | Bicyclic compounds composition and method for stabilizing the same |
| IL148803A IL148803A (en) | 1999-10-15 | 2002-03-21 | Composition and method for stabilizing the same |
| NO20021736A NO330258B1 (en) | 1999-10-15 | 2002-04-12 | Pharmaceutical preparations, bicyclic compounds and use of a glyceride for stabilizing a pharmaceutical preparation |
| HK02107337.5A HK1045693B (en) | 1999-10-15 | 2002-10-07 | Bicyclic compounds composition and method for stabilizing the same |
| AU2004242503A AU2004242503B2 (en) | 1999-10-15 | 2004-12-24 | Novel composition and method for stabilizing the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15954999P | 1999-10-15 | 1999-10-15 | |
| US60/159,549 | 1999-10-15 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2001027099A2 true WO2001027099A2 (en) | 2001-04-19 |
| WO2001027099A3 WO2001027099A3 (en) | 2001-10-25 |
Family
ID=22573018
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2000/007109 WO2001027099A2 (en) | 1999-10-15 | 2000-10-13 | Bicyclic compounds composition and method for stabilizing the same |
Country Status (26)
| Country | Link |
|---|---|
| US (5) | US6583174B1 (en) |
| EP (2) | EP1220849B1 (en) |
| JP (2) | JP4332316B2 (en) |
| KR (1) | KR100830061B1 (en) |
| CN (1) | CN1268623C (en) |
| AR (3) | AR026046A1 (en) |
| AT (2) | ATE402925T2 (en) |
| AU (2) | AU780342B2 (en) |
| BR (1) | BRPI0014869B8 (en) |
| CA (1) | CA2385732C (en) |
| CZ (1) | CZ303625B6 (en) |
| DE (2) | DE60039727D1 (en) |
| DK (2) | DK1426361T4 (en) |
| ES (2) | ES2221859T3 (en) |
| HK (2) | HK1045693B (en) |
| HU (2) | HU229547B1 (en) |
| IL (2) | IL148803A0 (en) |
| MX (1) | MXPA02003756A (en) |
| NO (1) | NO330258B1 (en) |
| NZ (1) | NZ518020A (en) |
| PT (2) | PT1426361E (en) |
| RU (1) | RU2695274C3 (en) |
| TR (1) | TR200201032T2 (en) |
| TW (1) | TWI281918B (en) |
| WO (1) | WO2001027099A2 (en) |
| ZA (1) | ZA200202312B (en) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2842810A1 (en) * | 2002-07-25 | 2004-01-30 | Inst Nat Sciences Appliq | NOVEL DIFLUORINATED GEM COMPOUNDS, THEIR PREPARATION METHODS AND THEIR APPLICATIONS. |
| WO2007086541A2 (en) * | 2006-01-24 | 2007-08-02 | R-Tech Ueno, Ltd. | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| WO2009005172A1 (en) | 2007-07-03 | 2009-01-08 | Sucampo Ag | Pharmaceutical combination of nsaid and prostaglandin compound |
| EP2298314A1 (en) | 2001-11-14 | 2011-03-23 | Sucampo AG | Dosage unit comprising a prostaglandin analog for treating constipation |
| WO2011054087A1 (en) * | 2009-11-03 | 2011-05-12 | Bernard Charles Sherman | Stable pharmaceutical formulations comprising lubiprostone |
| US8026393B2 (en) | 2006-01-24 | 2011-09-27 | Sucampo Ag | Soft-gelatin capsule formulation |
| US8071613B2 (en) * | 2000-09-05 | 2011-12-06 | Sucampo Ag | Anti-constipation composition |
| WO2012102355A1 (en) * | 2011-01-27 | 2012-08-02 | 小野薬品工業株式会社 | Bicyclic compound and use thereof for medical purposes |
| US8614340B2 (en) | 2009-07-28 | 2013-12-24 | Ono Pharmaceuticals Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| EP2735566A1 (en) * | 2006-02-07 | 2014-05-28 | R-Tech Ueno, Ltd. | Method for preparing prostaglandin derivative |
| US8748482B2 (en) | 2008-04-01 | 2014-06-10 | Shanghai Techwell Biopharmaceutical Co., Ltd | Lubiprostone crystal, the use and the method for the preparation thereof |
| US9029574B2 (en) | 2011-01-27 | 2015-05-12 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US10561649B2 (en) | 2007-07-03 | 2020-02-18 | Sucampo Ag | Pharmaceutical combination of opioid and prostaglandin compound |
| US10899729B2 (en) | 2016-06-30 | 2021-01-26 | Ono Pharmaceutical Co.. Ltd. | Method for producing bicyclic compound |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1268623C (en) * | 1999-10-15 | 2006-08-09 | 苏坎波公司 | Novel composition and method or stabilizing same |
| DK1562604T3 (en) * | 2002-10-23 | 2012-04-02 | Sucampo Ag | Prostaglandin compounds for the treatment of obesity |
| CN1753680B (en) * | 2002-12-27 | 2010-12-08 | 苏坎波公司 | Derivatives of prostaglandins for treating abdominal discomfort |
| DE602004030245D1 (en) * | 2003-07-03 | 2011-01-05 | Sucampo Ag | PHARMACEUTICAL MAGENTATE-RESISTANT COATED COMPOSITION CONTAINING PROSTAGLANDIN ANALOGUE AS CHLORIDE CHANNEL OPENER |
| TWI348386B (en) * | 2003-08-12 | 2011-09-11 | R Tech Ueno Ltd | Composition and method for promoting hair growth |
| TWI387454B (en) | 2004-09-02 | 2013-03-01 | Sucampo Ag | Method and composition for treating gastrointestinal disorder |
| PT1871380E (en) * | 2005-04-12 | 2011-11-21 | Sucampo Ag | Combined use of prostaglandin compound and proton pump inhibitor for the treatment of gastrointestinal disorders |
| RU2506072C2 (en) * | 2007-07-19 | 2014-02-10 | Р-Тек Уено, Лтд. | Pharmaceutical composition containing 11-deoxy-prostaglandine compound and method for stabilising same |
| US20090082442A1 (en) * | 2007-09-26 | 2009-03-26 | Protia, Llc | Deuterium-enriched lubiprostone |
| US8569279B2 (en) * | 2009-05-27 | 2013-10-29 | Sucampo Ag | Method for modulating claudin mediated functions |
| US20110034424A1 (en) * | 2009-06-30 | 2011-02-10 | Sucampo Ag | Method for the long term nsaid use |
| AU2010295464B2 (en) * | 2009-09-18 | 2015-11-26 | Merck Sharp & Dohme Corp. | Use of opioid receptor antagonist for gastrointestinal tract disorders |
| WO2014159679A1 (en) | 2013-03-12 | 2014-10-02 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Methods for using lubiprostone to absorb fluid from the subretinal space |
| WO2016161085A1 (en) | 2015-04-01 | 2016-10-06 | Cedars-Sinai Medical Center | Anti-methanogenic lovastatin analogs or derivatives and uses thereof |
| JP6957610B2 (en) | 2016-10-06 | 2021-11-02 | スキャンポ・アーゲーSucampo AG | Multilayer beads for pharmaceutical use |
| WO2019221215A1 (en) * | 2018-05-18 | 2019-11-21 | ニプロ株式会社 | Lubiprostone-containing particulate medicinal composition |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0347243A1 (en) * | 1988-06-17 | 1989-12-20 | The Wellcome Foundation Limited | Prostaglandin analogues for use in medicine |
| EP0353917A1 (en) * | 1988-07-29 | 1990-02-07 | Kabushiki Kaisha Ueno Seiyaku Oyo Kenkyujo | Precursor of prostaglandin and production thereof |
| EP0532218B1 (en) * | 1991-09-03 | 1998-03-25 | R-Tech Ueno Ltd. | Process for production of prostaglandin intermediates |
Family Cites Families (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US18202A (en) * | 1857-09-15 | Improvement in feathering paddle-wheels | ||
| JPS5720305B2 (en) | 1973-02-28 | 1982-04-27 | ||
| US4034003A (en) | 1974-04-11 | 1977-07-05 | Ono Pharmaceutical Co., Ltd. | 15-Cycloalkyl-prostaglandins |
| US3966962A (en) * | 1975-03-27 | 1976-06-29 | The Upjohn Company | Triacetin solutions of PGE-type compounds |
| JPS5350141A (en) * | 1976-10-18 | 1978-05-08 | Ono Pharmaceut Co Ltd | Stabilization of prostaglandin and prostaglandin analogues |
| JPS5919524B2 (en) | 1976-12-17 | 1984-05-07 | 中外製薬株式会社 | Stable 1α-hydroxyvitamin D-containing preparation |
| JPS5391110A (en) | 1977-01-20 | 1978-08-10 | Yamanouchi Pharmaceut Co Ltd | Novel drug composition for rectal infusion |
| CA1184906A (en) * | 1977-04-19 | 1985-04-02 | Roy A. Johnson | 9-deoxy-5,9-epoxy-prostaglandins |
| JPS6022708B2 (en) | 1977-07-14 | 1985-06-03 | 小野薬品工業株式会社 | Prostaglandin-like compounds |
| JPS5452717A (en) * | 1977-09-29 | 1979-04-25 | Chugai Pharmaceut Co Ltd | Stable oily pharmaceuticals containing 1alpha-hydroxyvitamin d |
| US4130566A (en) * | 1977-10-27 | 1978-12-19 | Sumitomo Chemical Company, Limited | Process for producing 5-carboxy-2-acetylthiophene |
| AT358530B (en) | 1978-04-25 | 1980-09-10 | Plc Pharma Licences | STABILIZATION PROCEDURE |
| JPS55136219A (en) | 1979-04-11 | 1980-10-23 | Sumitomo Chem Co Ltd | Diazepam oily drug |
| US4670569A (en) | 1981-04-02 | 1987-06-02 | G. D. Searle & Co. | 5-fluoro-PGI2 compounds |
| JPS59137413A (en) * | 1983-01-25 | 1984-08-07 | Teijin Ltd | Pharmaceutical composition containing 7- thiaprostaglandin e1 compound |
| US4579958A (en) | 1983-12-23 | 1986-04-01 | G. D. Searle & Co. | 5-fluoro-3-oxa-6,7-didehydro-PGI1 compounds |
| US4687864A (en) | 1983-12-23 | 1987-08-18 | G. D. Searle & Co. | 5-fluoro-3-oxa-prostacyclin compounds |
| US5428062A (en) | 1987-01-28 | 1995-06-27 | K.K. Ueno Seiyaku Oyo Kenkyujo | Prostaglandins E and anti ulcers containing same |
| US5166174A (en) | 1987-01-28 | 1992-11-24 | K.K. Ueno Seiyaku Oyo Kenkyujo | Prostaglandins E and anti-ulcers containing same |
| US5225439A (en) | 1987-01-28 | 1993-07-06 | K.K. Ueno Seiyaku Oyo Kenkyujo | Prostaglandins E and anti ulcers containing same |
| US4758880A (en) | 1987-03-27 | 1988-07-19 | The Grass Valley Group, Inc. | Video matte generator |
| KR920003601B1 (en) | 1987-09-03 | 1992-05-04 | 유니버시티 어브 죠지아 리서취 화운데이션 인코포레이티드 | Ocular cyclosporin composition |
| DE3873007T2 (en) | 1987-10-02 | 1992-12-03 | Ueno Seiyaku Oyo Kenkyujo Kk | KATHARTICA. |
| US5317032A (en) * | 1987-10-02 | 1994-05-31 | Kabushiki Kaisha Ueno Seiyaku Oyo Kenkyujo | Prostaglandin cathartic |
| JP2597649B2 (en) † | 1988-05-11 | 1997-04-09 | 株式会社上野製薬応用研究所 | Trachea / bronchodilator |
| JP2579193B2 (en) | 1988-07-19 | 1997-02-05 | 小野薬品工業株式会社 | 16,16-difluoro-15-oxo-15-deoxy PGE derivative |
| JP2794433B2 (en) | 1989-02-02 | 1998-09-03 | 丸善製薬株式会社 | Licorice hydrophobic flavonoid preparation |
| CA2030344C (en) * | 1989-11-22 | 2000-04-18 | Ryuji Ueno | Treatment of pulmonary dysfunction with 15-keto-prostaglandin compounds |
| US5254588A (en) * | 1989-11-22 | 1993-10-19 | Kabushikikaisha Ueno Seiyaku Oyo Kenkyujo | Treatment of pulmonary dysfunction with 15-ketoprostaglandin compounds |
| US5256696A (en) * | 1989-11-22 | 1993-10-26 | Kabushikikaisha Ueno Seiyaku Oyo Kenkyujo | Treatment of cardiac dysfunction with 15-ketoprostaglandin compounds |
| CA2030346C (en) * | 1989-11-22 | 2000-04-11 | Ryuji Ueno | Treatment of cardiac dysfunction with 15-keto-prostaglandin compounds |
| CA2030345C (en) * | 1989-11-22 | 1998-12-08 | Ryuji Ueno | Use of 15-keto-prostaglandin compound for improvement of encephalic function |
| CA2041417C (en) * | 1990-05-01 | 2002-05-21 | Ryuji Ueno | Treatment of pancreatic disease with 15-keto-prostaglandin compounds |
| JPH0446122A (en) | 1990-06-13 | 1992-02-17 | Nippon Iyakuhin Kogyo Kk | Active type vitamin d3s-containing pharmaceutical |
| CA2046069C (en) | 1990-07-10 | 2002-04-09 | Ryuji Ueno | Treatment of inflammatory diseases with 15-keto-prostaglandin compounds |
| JP2938579B2 (en) | 1990-12-15 | 1999-08-23 | 株式会社上野製薬応用研究所 | GI wall protectant |
| JP2746800B2 (en) * | 1991-09-03 | 1998-05-06 | 株式会社アールテック・ウエノ | Preparation of prostaglandin intermediate |
| US5474979A (en) | 1994-05-17 | 1995-12-12 | Allergan, Inc. | Nonirritating emulsions for sensitive tissue |
| CA2150287C (en) * | 1994-06-03 | 2004-08-10 | Ryuji Ueno | Agent for treating hepato-biliary diseases |
| US5981607A (en) | 1998-01-20 | 1999-11-09 | Allergan | Emulsion eye drop for alleviation of dry eye related symptoms in dry eye patients and/or contact lens wearers |
| CN1268623C (en) * | 1999-10-15 | 2006-08-09 | 苏坎波公司 | Novel composition and method or stabilizing same |
| US6414016B1 (en) | 2000-09-05 | 2002-07-02 | Sucampo, A.G. | Anti-constipation composition |
| JP4210631B2 (en) | 2004-06-24 | 2009-01-21 | 株式会社日立製作所 | Brake control device |
-
2000
- 2000-10-13 CN CNB008143137A patent/CN1268623C/en not_active Ceased
- 2000-10-13 PT PT04005836T patent/PT1426361E/en unknown
- 2000-10-13 DE DE60039727T patent/DE60039727D1/en not_active Expired - Lifetime
- 2000-10-13 AT AT04005836T patent/ATE402925T2/en active
- 2000-10-13 HU HU0203746A patent/HU229547B1/en not_active IP Right Cessation
- 2000-10-13 PT PT00966462T patent/PT1220849E/en unknown
- 2000-10-13 IL IL14880300A patent/IL148803A0/en active IP Right Grant
- 2000-10-13 AT AT00966462T patent/ATE267189T1/en active
- 2000-10-13 WO PCT/JP2000/007109 patent/WO2001027099A2/en active IP Right Grant
- 2000-10-13 BR BRPI0014869A patent/BRPI0014869B8/en not_active IP Right Cessation
- 2000-10-13 NZ NZ518020A patent/NZ518020A/en not_active IP Right Cessation
- 2000-10-13 EP EP00966462A patent/EP1220849B1/en not_active Expired - Lifetime
- 2000-10-13 AU AU76856/00A patent/AU780342B2/en not_active Expired
- 2000-10-13 MX MXPA02003756A patent/MXPA02003756A/en active IP Right Grant
- 2000-10-13 EP EP04005836.4A patent/EP1426361B2/en not_active Expired - Lifetime
- 2000-10-13 ES ES00966462T patent/ES2221859T3/en not_active Expired - Lifetime
- 2000-10-13 DK DK04005836T patent/DK1426361T4/en active
- 2000-10-13 HU HU1300369A patent/HU230484B1/en not_active IP Right Cessation
- 2000-10-13 TR TR2002/01032T patent/TR200201032T2/en unknown
- 2000-10-13 AR ARP000105407A patent/AR026046A1/en not_active Application Discontinuation
- 2000-10-13 DE DE60010913T patent/DE60010913T2/en not_active Expired - Lifetime
- 2000-10-13 TW TW089121423A patent/TWI281918B/en not_active IP Right Cessation
- 2000-10-13 JP JP2001530318A patent/JP4332316B2/en not_active Expired - Lifetime
- 2000-10-13 CZ CZ20021037A patent/CZ303625B6/en not_active IP Right Cessation
- 2000-10-13 ES ES04005836.4T patent/ES2309410T5/en not_active Expired - Lifetime
- 2000-10-13 DK DK00966462T patent/DK1220849T3/en active
- 2000-10-13 KR KR1020027004709A patent/KR100830061B1/en active Protection Beyond IP Right Term
- 2000-10-13 CA CA002385732A patent/CA2385732C/en not_active Expired - Lifetime
- 2000-10-13 RU RU2002112984A patent/RU2695274C3/en active Protection Beyond IP Right Term
- 2000-10-16 US US09/688,351 patent/US6583174B1/en not_active Expired - Lifetime
-
2002
- 2002-03-21 IL IL148803A patent/IL148803A/en unknown
- 2002-03-22 ZA ZA200202312A patent/ZA200202312B/en unknown
- 2002-04-12 NO NO20021736A patent/NO330258B1/en not_active IP Right Cessation
- 2002-10-07 HK HK02107337.5A patent/HK1045693B/en not_active IP Right Cessation
- 2002-10-07 HK HK04109640.1A patent/HK1066792A1/en not_active IP Right Cessation
-
2003
- 2003-03-10 US US10/383,581 patent/US7417067B2/en not_active Expired - Lifetime
-
2004
- 2004-12-24 AU AU2004242503A patent/AU2004242503B2/en not_active Expired
-
2008
- 2008-06-23 US US12/144,000 patent/US8088934B2/en not_active Expired - Lifetime
-
2009
- 2009-05-25 JP JP2009125293A patent/JP2009235079A/en active Pending
-
2011
- 2011-11-04 US US13/289,386 patent/US8097649B1/en not_active Expired - Lifetime
- 2011-12-27 US US13/337,391 patent/US20120095090A1/en not_active Abandoned
-
2013
- 2013-01-30 AR ARP130100275A patent/AR089848A2/en not_active Application Discontinuation
-
2017
- 2017-08-16 AR ARP170102288A patent/AR109354A2/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0347243A1 (en) * | 1988-06-17 | 1989-12-20 | The Wellcome Foundation Limited | Prostaglandin analogues for use in medicine |
| EP0353917A1 (en) * | 1988-07-29 | 1990-02-07 | Kabushiki Kaisha Ueno Seiyaku Oyo Kenkyujo | Precursor of prostaglandin and production thereof |
| EP0532218B1 (en) * | 1991-09-03 | 1998-03-25 | R-Tech Ueno Ltd. | Process for production of prostaglandin intermediates |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8071613B2 (en) * | 2000-09-05 | 2011-12-06 | Sucampo Ag | Anti-constipation composition |
| US8748454B2 (en) | 2000-09-05 | 2014-06-10 | Sucampo Ag | Anti-constipation composition |
| US8114890B1 (en) | 2000-09-05 | 2012-02-14 | Sucampo Ag | Anti-constipation composition |
| EP2298314A1 (en) | 2001-11-14 | 2011-03-23 | Sucampo AG | Dosage unit comprising a prostaglandin analog for treating constipation |
| WO2004014928A2 (en) * | 2002-07-25 | 2004-02-19 | Institut National Des Sciences Appliquees De Rouen (Insa) | Novel difluorinated gem compounds, preparation methods thereof and applications of same |
| WO2004014928A3 (en) * | 2002-07-25 | 2004-04-01 | Inst Nat Sciences Appliq | Novel difluorinated gem compounds, preparation methods thereof and applications of same |
| FR2842810A1 (en) * | 2002-07-25 | 2004-01-30 | Inst Nat Sciences Appliq | NOVEL DIFLUORINATED GEM COMPOUNDS, THEIR PREPARATION METHODS AND THEIR APPLICATIONS. |
| US8669284B2 (en) | 2006-01-24 | 2014-03-11 | Sucampo Ag | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| CN104983712A (en) * | 2006-01-24 | 2015-10-21 | 株式会社·R-技术上野 | Soft-gelatin capsule formulation |
| US8026393B2 (en) | 2006-01-24 | 2011-09-27 | Sucampo Ag | Soft-gelatin capsule formulation |
| US7985770B2 (en) | 2006-01-24 | 2011-07-26 | Sucampo Ag | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| KR101465269B1 (en) * | 2006-01-24 | 2014-11-26 | 가부시키가이샤 아루떼꾸 우에노 | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| US8779187B2 (en) | 2006-01-24 | 2014-07-15 | Sumcampo AG | Soft-gelatin capsule formulation |
| US8338639B2 (en) | 2006-01-24 | 2012-12-25 | Sucampo Ag | Soft-gelatin capsule formulation |
| WO2007086541A2 (en) * | 2006-01-24 | 2007-08-02 | R-Tech Ueno, Ltd. | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| EP2682130A1 (en) | 2006-01-24 | 2014-01-08 | R-Tech Ueno, Ltd. | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| WO2007086541A3 (en) * | 2006-01-24 | 2008-02-21 | R Tech Ueno Ltd | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
| EP2735566A1 (en) * | 2006-02-07 | 2014-05-28 | R-Tech Ueno, Ltd. | Method for preparing prostaglandin derivative |
| US10561649B2 (en) | 2007-07-03 | 2020-02-18 | Sucampo Ag | Pharmaceutical combination of opioid and prostaglandin compound |
| WO2009005172A1 (en) | 2007-07-03 | 2009-01-08 | Sucampo Ag | Pharmaceutical combination of nsaid and prostaglandin compound |
| EP2275419A4 (en) * | 2008-04-01 | 2016-03-30 | Shanghai Techwell Biopharm Co | A lubiprostone crystal, its preparation process and its use |
| US8748482B2 (en) | 2008-04-01 | 2014-06-10 | Shanghai Techwell Biopharmaceutical Co., Ltd | Lubiprostone crystal, the use and the method for the preparation thereof |
| US9889114B2 (en) | 2009-07-28 | 2018-02-13 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US8962868B2 (en) | 2009-07-28 | 2015-02-24 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US9388157B2 (en) | 2009-07-28 | 2016-07-12 | Ono Pharmaceuticals Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US10201520B2 (en) | 2009-07-28 | 2019-02-12 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US8614340B2 (en) | 2009-07-28 | 2013-12-24 | Ono Pharmaceuticals Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US10940134B2 (en) | 2009-07-28 | 2021-03-09 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US11617733B2 (en) | 2009-07-28 | 2023-04-04 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| WO2011054087A1 (en) * | 2009-11-03 | 2011-05-12 | Bernard Charles Sherman | Stable pharmaceutical formulations comprising lubiprostone |
| US8940790B2 (en) | 2009-11-03 | 2015-01-27 | Bernard Charles Sherman | Stable pharmaceutical formulations comprising lubiprostone |
| US9029574B2 (en) | 2011-01-27 | 2015-05-12 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| US9150537B2 (en) | 2011-01-27 | 2015-10-06 | Ono Pharmaceutical Co., Ltd. | Bicyclic compound and use thereof for medical purposes |
| WO2012102355A1 (en) * | 2011-01-27 | 2012-08-02 | 小野薬品工業株式会社 | Bicyclic compound and use thereof for medical purposes |
| US10899729B2 (en) | 2016-06-30 | 2021-01-26 | Ono Pharmaceutical Co.. Ltd. | Method for producing bicyclic compound |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1220849B1 (en) | Bicyclic compounds composition and method for stabilizing the same | |
| AU2008276840B2 (en) | Pharmaceutical composition comprising 11-deoxy-prostaglandin compound and method for stabilizing the compound | |
| US8669284B2 (en) | Pharmaceutical composition comprising a bi-cyclic compound and method for stabilizing the bi-cyclic compound |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 76856/00 Country of ref document: AU Ref document number: 2385732 Country of ref document: CA Ref document number: 148803 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV2002-1037 Country of ref document: CZ Ref document number: 2002/02312 Country of ref document: ZA Ref document number: 200202312 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 518020 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000966462 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IN/PCT/2002/516/CHE Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2001 530318 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020027004709 Country of ref document: KR Ref document number: PA/a/2002/003756 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 008143137 Country of ref document: CN Ref document number: 2002/01032 Country of ref document: TR |
|
| ENP | Entry into the national phase |
Ref document number: 2002 2002112984 Country of ref document: RU Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000966462 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020027004709 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: PV2002-1037 Country of ref document: CZ |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 518020 Country of ref document: NZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2000966462 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 518020 Country of ref document: NZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 76856/00 Country of ref document: AU |