WO2001032013A1 - Method and device for attracting insects - Google Patents
Method and device for attracting insects Download PDFInfo
- Publication number
- WO2001032013A1 WO2001032013A1 PCT/US2000/013477 US0013477W WO0132013A1 WO 2001032013 A1 WO2001032013 A1 WO 2001032013A1 US 0013477 W US0013477 W US 0013477W WO 0132013 A1 WO0132013 A1 WO 0132013A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- com
- soil
- termites
- larvae
- graph
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01M—CATCHING, TRAPPING OR SCARING OF ANIMALS; APPARATUS FOR THE DESTRUCTION OF NOXIOUS ANIMALS OR NOXIOUS PLANTS
- A01M1/00—Stationary means for catching or killing insects
- A01M1/10—Catching insects by using Traps
- A01M1/103—Catching insects by using Traps for crawling insects
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01M—CATCHING, TRAPPING OR SCARING OF ANIMALS; APPARATUS FOR THE DESTRUCTION OF NOXIOUS ANIMALS OR NOXIOUS PLANTS
- A01M1/00—Stationary means for catching or killing insects
- A01M1/02—Stationary means for catching or killing insects with devices or substances, e.g. food, pheronones attracting the insects
- A01M1/023—Attracting insects by the simulation of a living being, i.e. emission of carbon dioxide, heat, sound waves or vibrations
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01M—CATCHING, TRAPPING OR SCARING OF ANIMALS; APPARATUS FOR THE DESTRUCTION OF NOXIOUS ANIMALS OR NOXIOUS PLANTS
- A01M1/00—Stationary means for catching or killing insects
- A01M1/02—Stationary means for catching or killing insects with devices or substances, e.g. food, pheronones attracting the insects
- A01M1/026—Stationary means for catching or killing insects with devices or substances, e.g. food, pheronones attracting the insects combined with devices for monitoring insect presence, e.g. termites
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01M—CATCHING, TRAPPING OR SCARING OF ANIMALS; APPARATUS FOR THE DESTRUCTION OF NOXIOUS ANIMALS OR NOXIOUS PLANTS
- A01M1/00—Stationary means for catching or killing insects
- A01M1/20—Poisoning, narcotising, or burning insects
- A01M1/2005—Poisoning insects using bait stations
- A01M1/2011—Poisoning insects using bait stations for crawling insects
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
- A01N59/04—Carbon disulfide; Carbon monoxide; Carbon dioxide
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N63/00—Biocides, pest repellants or attractants, or plant growth regulators containing microorganisms, viruses, microbial fungi, animals or substances produced by, or obtained from, microorganisms, viruses, microbial fungi or animals, e.g. enzymes or fermentates
- A01N63/10—Animals; Substances produced thereby or obtained therefrom
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N63/00—Biocides, pest repellants or attractants, or plant growth regulators containing microorganisms, viruses, microbial fungi, animals or substances produced by, or obtained from, microorganisms, viruses, microbial fungi or animals, e.g. enzymes or fermentates
- A01N63/30—Microbial fungi; Substances produced thereby or obtained therefrom
- A01N63/32—Yeast
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N65/00—Biocides, pest repellants or attractants, or plant growth regulators containing material from algae, lichens, bryophyta, multi-cellular fungi or plants, or extracts thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N65/00—Biocides, pest repellants or attractants, or plant growth regulators containing material from algae, lichens, bryophyta, multi-cellular fungi or plants, or extracts thereof
- A01N65/40—Liliopsida [monocotyledons]
- A01N65/44—Poaceae or Gramineae [Grass family], e.g. bamboo, lemon grass or citronella grass
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01M—CATCHING, TRAPPING OR SCARING OF ANIMALS; APPARATUS FOR THE DESTRUCTION OF NOXIOUS ANIMALS OR NOXIOUS PLANTS
- A01M2200/00—Kind of animal
- A01M2200/01—Insects
- A01M2200/011—Crawling insects
Definitions
- the present invention is directed to a method and device for attracting certain insects, and more particularly is directed to a method and device for attracting termites to ultimately trap or otherwise destroy such termites, as well as a method to reduce damage caused by corn root worms.
- Baits stations have been utilized in an attempt to attract termites and thereby trap and/or destroy the termites that enter into such bait stations. Bait stations are available in a variety of shapes, sizes and structures, but principally rely upon the attractiveness of a cellulase product, such as paper or wood, to attract termite populations.
- Another aspect of the present invention involves the reduction of damage to crops, particularly corn crops, caused by the corn root worm.
- the damages caused by such insects is estimated to be over one billion dollars in the U.S. alone.
- pesticides have been used in the past to remedy such problems, they have been largely ineffective and have proven to cause environmental problems and to be fairly expensive.
- the present inventors were the first to discover that root worm larvae navigate to food sources by detecting carbon dioxide. There is therefore a long felt, but unsolved need for a method and formulation capable of attracting corn root worms to avoid the significant damage done by such insects every year.
- the present invention is directed to a method and device for attracting certain insects, and in particular, boring insects such as termites and beetles.
- a separate aspect of the invention relates to a method and formulations for alleviating and/or reducing corn root worm damage.
- the method comprises the use of particular amounts of CO 2 as an attractant for such boring insects.
- the present invention includes not only the method for using particular novel formations, but the formulations themselves, as well as devices which incorporate such formulations for the trapping and/or destruction of boring insects.
- the present formulation generally have in common the ability to give off particular amounts of CO 2 found by the present inventors to be particularly attractive to boring insects such as termites.
- the present formulation comprises the generation of CO 2 in a concentration of from between about 2 mmol/mol to about 50 mmol/mol, more particularly in amounts greater than about 2 mmol/mol and less than about 20 mmol/mol, and even more preferably between about 5 and about 10 mmol/mol.
- Preferred CO 2 concentrations are at least above ambient concentrations.
- Such CO 2 concentrations can be generated using one or more of a biological generation source, a chemical generation source and a mechanical generation source.
- certain bacterial, fungal (e.g., yeast), algal and other microorganism formulations can be used that generate the above-referenced concentrations of CO 2 over a particular period of time.
- chemical reactions that generate CO 2 can be utilized to achieve such concentrations such as carbonate, calcium carbonate and various bicarbonate formulations as set forth and/or referred to herein.
- mechanical systems which incorporate the slow release of contained sources of CO 2 can be utilized to achieve desired objectives of the present invention.
- Combinations of the biological, chemical and mechanical methods and devices are also within the scope of the present invention. The detailed description of such embodiments can be found in the detailed description of the preferred embodiments, below.
- the novel method of the present invention comprises the generation of CO 2 in an amount within the above-specified ranges in order to attract boring insect populations.
- such method comprises positioning an enclosure containing one or more of the above-referenced biological, chemical and/or mechanical sources of CO 2 in an area sought to be protected from boring insects such as termites.
- Various controls with respect to CO 2 generation fall within the scope of the present invention, including temperature, light sensors, temporal adjustment mechanisms, etc., to achieve desired CO 2 emissions within appropriate concentration ranges at particular times of day and/or night, and/or at particular ambient temperatures at which insects may be most attracted to such sources, etc.
- various forms and structures are in contemplated including bait traps and stations similar to those commercially available. Still other embodiments, however, have a varied configuration as set forth in the figures.
- a separate aspect of the present invention involves the use of charred cellulose material, and in particular charred wood, as an attractant for boring insects such as termites. While not being bound by theory, the present inventors believe that charred wood provides an easier target material for boring insects and thus, over evolutionary time, such boring insects have evolved a particular attraction to charred cellulase as a feeding stimulant.
- a further aspect of the present invention includes the particular novel compositions and formulations found in charred wood that attracts such boring insects and the use of such compounds in the above-described method, devices and formulations for attracting and extermination of undesired insects such as boring beetles, termites, etc.
- CO 2 mimics include, but are not limited to, haloalkanes and alkylcarbonates.
- the various formulations of the present invention that comprise CO 2 or CO 2 mimics may further be combined with sources of insecticide, sources of food, feeding stimulants, or other materials that arrest and/or stimulate termite movement or behavior.
- the use of CO 2 or CO 2 mimics, alone or in combination with other components can be used to disrupt the orientation behavior of termites in a behavioral fashion, rather than as acting as a physiologically deleterious fumigant.
- CO 2 and CO 2 mimics can be used as co-attractants for termites along with other attractive materials that may have fundamentally different chemical compositions.
- the formulations of the present invention can be used to attract termites to termite traps, and further can be used to monitor the presence or abundance of particular termite species.
- manipulation of the amount of CO 2 generated can be adjusted to attract a particular species of termite, given the present inventors' appreciation and recognition that different CO 2 concentrations are more or less attractive to various species of termites.
- An extensive list of termite bait compounds that can be used in conjunction with the present invention to fashion appropriate formulations is shown in tables set forth below.
- a separate aspect of the present invention relates to a method and formulation for ameliorating the damage caused by corn root worms.
- the present inventors were the first to discover that corn root worms are capable of navigating to food sources by detecting carbon dioxide emitted from roots.
- the present invention is directed to various formulations found effective in attracting such root worms in a manner that protects growing crops from destruction by such insects.
- the present inventors are the first to discover an inexpensive and readily available material that, if applied properly, can be used to vastly reduce the damage caused by corn root worms.
- the present inventors are first to discover that spent grain and distillers grain can be used by farmers as a readily available and inexpensive source of a CO 2 evolving agent.
- the present invention not only encompasses, therefore, the method of applying such materials at particular times during the growing season, but also to machinery used to preferably administer such material.
- the present invention involves a new use for existing machinery used in planting and in fertilizer applications, such as a cone planter and starter fertilizer equipment, conventionally used for com planting and fertilization.
- Such existing machines can be further modified to achieve the desired objective of the present invention so that sources of CO 2 evolving substances can be precisely contacted with the soil to achieve the com root worm attractant objective.
- Com root worms can be attracted by use of biological, chemical and mechanical means, most preferably biological and chemical means as set forth herein as applicable to other boring insects, such as termites.
- CO 2 is an inexpensive, environmentally-friendly compound that is readily available and can be generated in a number of ways.
- Fig. 1 illustrates how a typical cone planter can be modified in order to place formulations of the present invention a desired distance from a particular com seed.
- Fig. 2 illustrates how a starter fertilizer attachment on a com planter can be utilized to properly place the formulations of the present invention within a desired distance from a com seed.
- Fig. 3 illustrates one embodiment of a jar trap for insects, including termites.
- DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS The present inventors incorporate by reference the following U.S. Patents in their entirety, such patents disclosing various compounds and formulations that are useful in conjunction with the present invention.
- U.S. Patent No. 5,338,551 to Lajoie U.S. Patent No. 5,342,630 to Jones
- U.S. Patent No. 5,346,704 to Lajoie U.S. Patent Nos. 5,389,386,
- One aspect of the present invention is directed to the alleviation of com root worm damage by providing a CO 2 evolving agent in a planted field so as to attract and/or otherwise confuse com root worms, thus reducing the damage caused by such root worm to com roots.
- biological, chemical and mechanical methods, as otherwise set forth herein can be used, biological and chemical formulations are particularly preferred. Indeed, the present inventors are first to appreciate the use of inexpensive and readily available materials to accomplish the objective of reducing com root worm damage done to com crops in the United States and elsewhere in the world.
- spent grain and/or distiller's grain can be used, easily obtainable from breweries and alcohol generation facilities, such materials being either generally plowed into fields at appropriate times during the planting, cultivation and/or growing season, and/or precisely located in such fields to achieve desired attractant functions.
- the present invention thus entails the first appreciation and recognition that by contacting (e.g., plowing) particular biological material, such as spent grain/distiller's grain into a field (e.g., com fields), at an appropriate time in the spring or early summer (or any other planting and/or cultivation period in more temperate climates) it is possible to ameliorate the destruction caused by com root worms.
- a field e.g., com fields
- CO 2 evolving agents charcoal, activated carbon and decolorizing carbon
- com cob grits can be used as an acceptable microbial substrate for the production of CO 2 .
- strips of biological and/or chemical CO 2 evolving material are contacted with fields between or adjacent to the rows of plants. This can be accomplished by using various existing machines such a cone planters or starter fertilizer equipment. Modifying such equipment to achieve the desired precise placement of CO 2 evolving materials is preferred and such modifications will be obvious to one of skill in the art given the general teachings and guidance of the present invention.
- Various biological sources for CO 2 evolving agents include ground germinated com, clean cracked com, malted barley, any other malted grain, com gluten feed, fungal organisms such as yeast, bacteria, such as S.
- cervisae sour dough bread starter
- algae and various other microorganisms that exist in soil.
- Various chemical CO 2 evolving agents can be used, such as those mentioned herein, preferably including carbonates, including inorganic carbonates such as calcium carbonate, bicarbonates and alkyl carbonates. Urea-based compounds can also be utilized.
- double or other multiple acting compounds such as double acting baking powder can be utilized. It is within the scope of the present invention to combine the chemical and biological CO 2 evolving agents in various formulations. For example, spent grain, preferably in a dried form, can be mixed with appropriate amounts of carbonates and/or bicarbonates and/or urea to form appropriate compounds for attracting com root worm larvae/insects.
- Another aspect of the present invention involves the new use of dried spent grain and/or distiller's grain.
- spent grain and distiller's grain is provided in a "wet" composition.
- Such a form is not suitable for commercial sale for use as a CO 2 evolving agent since in such a "wet" and/or moist state, the material will rot and will evolve CO 2 prior to the time that it is administered to the soil.
- one aspect of the present invention involves the manufacture of dry spent grain/distiller's grain having a long shelf life so that it can be sold and properly administered to fields so as to accomplish the CO 2 evolving objective of the present invention.
- co-attractants can be added to the present inventive formulations (e.g., phermones, etc.) to further enhance the attractive features of the present formulations.
- the formulations of the present invention are produced in either a solid or liquid form.
- the present invention is preferably in granular form of a nature and size that facilitates administration of such granules through existing insecticide administering equipment used in conventional farming operations. These include, but are not limited to a noble meter and a Winter-Steiger meter.
- liquid forms of the various formulations are contemplated which are believed to be easier to handle and to administer. For example, such liquids could be crop dusted and/or subject to chemigation, using center pivot irrigation systems.
- the present invention can be in the form of a gel or slurry for particular applications.
- one aspect of the present invention involves a method for applying CO 2 evolving agents at a particular advantageous distance from roots of plants to attract various insects (e.g., com root worms).
- insects e.g., com root worms
- the farther away a CO 2 agent is placed from a plant root the stronger (e.g., concentrated) the CO 2 evolving agent can be.
- the goal is to attract desired larvae/insects without causing damage to plant roots and thus, the distance and concentration parameters will vary depending upon the particular plant involved and the particular CO 2 evolving agent employed.
- the inventors are also the first to appreciate the generation and use of a compound that is useful not only to alleviate com root worm problems, but at the same time, provides advantageous fertilization to desired plants.
- ammonium bicarbonate for example, not only is CO 2 generated which attracts com root worm larvae, such compound also acts to provide needed nutrients and fertilizer to com plants.
- Another aspect of the present invention relates to the use of charred cellulose material, such as wood, to attract various insects, such as boring insects, and in particular, termites.
- charred wood as a bait for termites, including the role of burned wood as a source of volatile and non- volatile attractants and as a source of feeding stimulants for termites.
- activated carbon decolorizing carbon and co cob grits can be used as the attractant/CO 2 evolving agent.
- any form of bumed or charred natural materials or artificial materials may be used, preferably bumed cellulosic matrix/burned polymeric matrix.
- the pyrolysis products of burning are similar for such materials as wood, paper, cardboard, fabric, textiles, wool, silk, bone, hair, hom, claws, or any other natural products, and the pyrolysis products of artificial polymers mimic the pyrolysis products of natural materials in many instances.
- Examples of behavioral manipulation of termite species include, but are not limited to, the following:
- charred wood, products of charred wood, or other bumed materials (a) to attract termites to traps for monitoring the presence or abundance of termite species; (b) to attract termites to sources of insecticides, insect growth regulators, or other toxic or physiologically active materials; (c) as feeding stimulants for termites, to induce them to feed on sources of insecticides, insect growth regulators, or other toxic or physiologically active materials; (d) to attract termites to sources of food, feeding stimulants, or other materials that arrest termite movement; (e) to disrupt the orientation behavior of termites behaviorally rather than acting as a physiologically deleterious fumigant; (f) as co-attractants for termites along with other attractive materials that may have fundamentally different chemistry; and (g) for the behavioral manipulation of any termite species, including use of such bumed materials as attractants or feeding stimulants for termites.
- Still other aspects of the present invention relate to the use of compounds that are chemically isolated from bumed wood or other bumed materials: (a) as attractants for termites; (b) as feeding stimulants for termites; and (c) for use in disrupting termite behavior in any way.
- jars having appropriately sized holes therein are utilized within which are stored attractant material.
- the physical configuration of such jars can be greatly varied, however, a shorter, squatter configuration is particularly preferred.
- apertures in the jars are preferably spaced about the circumference of the jar, and more preferably, evenly spaced throughout the surface area of the jar's sides.
- An important aspect of the present invention is the total area of apertures with respect to the jar's surface.
- no more than about 10% of the surface area of the jar comprises apertures, and more preferably, less than about 5% of the surface area of the jar.
- the limited access of termites to the interior of the jar is believed to be advantageous given that termites seek such relatively small openings, potentially due to the higher concentrations of CO 2 emitting from such orifices.
- the physical configuration of such bait traps is typically that of "jars", such jars constmcted of any suitable material including plastic, glass, ceramic, metal, etc. In general, the larger the volume of the bait trap, the better.
- the diameter of the bait jar used is about 90 mm, with a height of about 100 mm and has hole diameters of approximately 3 mm wherein at least about 50 holes are evenly distributed over the entire circumference of the jar.
- the attractant material of the present invention is provided.
- the present invention comprises the addition of soil to bait traps as the attractant material.
- Soil which may include sand, gravel, pebbles, dirt, as well as other constituents, is freely attainable and especially when used in conjunction with conventional bait traps having cellulose products therein, the addition of soil is found to provide impressive and unexpected attractant results.
- citric acid combined with sodium bicarbonate is particularly preferred, especially in a pelletized form.
- "fizzies” have been found to be particularly advantageous as a termite control attractant when added to soil having a moisture content of at least about 10% and more preferably about 20% of moisture.
- Other compounds can be added to the present formulations to achieve either attractant or destruction ability of the formulation.
- various poisons can be mixed with the CO 2 bait traps of the present invention.
- any insecticide or insect growth regulator can be used in conjunction with a CO 2 evolving source.
- Examples of such compounds include hexaflurone and hydramethylnon.
- various phermones can also be utilized for particular insect species sought to be attracted, such phermones added with the formulations of the present invention.
- suitable bait traps are positioned away from building structures or other wooden edifices sought to be protected.
- the devices should have an effective life of several weeks, preferably several months, and as much as a year or more.
- the attractant compounds and formulations of the present invention are generally referred to herein as "attracticides".
- Yet another aspect of the present invention involves the manufacture of building materials so as to make such materials less susceptible to termite damage.
- conventional foam panels used in insulation materials emit carbon dioxide.
- the elimination of carbon dioxide in the manufacture of such foam materials by, for example, use of other non-CO 2 containing gases, provides a method to produce termite resistant building and/or insulation materials.
- Further aspects of the present invention also include methods to seal existing structures that are prone to emit CO 2 concentrations in amounts found attractive to various boring insects. For example, creating substantially air-tight seals around conventional CO 2 based foam products is effective in reducing the attractant quality of such materials to boring insects such as termites.
- CO 2 emitting insulation and building materials to avoid possible destruction by boring insects attracted to CO 2 emitting substances.
- CO 2 emitting concentrations should be reduced to below the dose found in soils so as to eliminate any source of CO 2 that may attract insects.
- Preferred formulations of the present invention are in pelleted form to achieve slow release of CO 2 at the above-described concentrations.
- Example 1 - Composition of Formulation 1 (Dried Spent Brewer's Grain): Spent brewer's grain obtained from a local brewery was spread out on trays and allowed to air dry overnight. The dried spent brewer's grain was then added to soil that contained 20% moisture (12 g dried spent brewer's grain per 100 g moist soil).
- Jar traps were constructed from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 1 in ajar trap. Unbaited traps were filled with 300 g of soil (20% moisture). A disk of cardboard (8 cm diameter) was placed in the top of each trap (baited and unbaited), covered with a thin layer of soil, and the lid was then screwed onto the trap.
- Field sites fence posts infested with termites (Reticulitermes tibialis) were used for field trapping experiments at three different ranches in Colorado (Fort Collins, Nunn, and Akron). Each infested fence post was used as a point source for an experiment. Six traps, three baited and three unbaited, were placed in the ground evenly around the fence post at a distance of 1 meter. The traps were placed in the ground at a depth of 20 to 25 cm and covered completely with soil. Traps were checked weekly for the presence of termites. Traps were checked for feeding damage on the cardboard disks. Cardboard disks were taken back to the laboratory, where each piece was carefully washed and spread out to dry. The amount of cardboard eaten was determined by scanning the pieces with a desktop scanner and calculating the area by using a computer graphics program (Adobe Photoshop). The experiment was continued for six weeks at each location.
- Example 2 Composition of Formulation 2 (Dried Ground Germinated Corn Seeds):
- Com seeds were soaked in soapy water overnight, rinsed well and germinated in a covered plastic tub containing moist germination paper. After 3 days of germination, the germinating com was ground to meal using a kitchen food processor, then spread out on trays and allowed to air dry overnight. Dried, ground, germinated com seed (12 g per 100 g soil) was added to soil that contained 20% moisture.
- Jar traps were constmcted from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 2 in ajar trap. Unbaited traps were filled with 300 g soil (20% moisture). A disk of cardboard (8 cm diameter) was placed in the top of each trap (baited and unbaited), covered with a thin layer of soil, and the lid was then screwed onto the trap.
- Field sites fence posts infested with termites (Reticulitermes tibialis) were used for field trapping experiments at three different ranches in Colorado (Fort Collins, Nunn, and Akron). Each infested fence post was used as a point source for an experiment. Six traps, three baited and three unbaited, were placed in the ground evenly around the fence post at a distance of 1 meter. The traps were placed in the ground at a depth of 20 to 25 cm and covered completely with soil. Traps were checked weekly for the presence of termites. Traps were checked for feeding damage on the cardboard disks. Cardboard disks were taken back to the laboratory, where each piece was carefully washed and spread out to dry. The amount of cardboard eaten was determined by scanning the pieces with a desktop scanner and calculating the area by using a computer graphics program (Adobe Photoshop). The experiment was continued for six weeks at each location.
- Example 3 - Composition of Formulation 3 (Whole Dry Malted Barley): Whole dry malted barley was obtained from a local brewer's store. The whole dry malted barley was then added to soil that contained 20% moisture (12 g whole dry malted barley per 100 g moist soil).
- Jar traps were constmcted from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 3 in ajar trap. Unbaited traps were filled with 300 g of soil (20% moisture). A disk of cardboard (8 cm diameter) was placed in the top of each trap (baited and unbaited), covered with a thin layer of soil, and the lid was then screwed onto the trap.
- Formulation 4 Composition of Formulation 4 (Coated Sucrose Pellets): Sucrose pellets with a light wax coating were obtained from a local supplier (Sprinkle Decorations, Wilton Enterprises, Woodridge, IL). The sucrose pellets with a light wax coating were then added to soil that contained 20% moisture (12 g per 100 g moist soil).
- Jar traps were constmcted from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 4 in a jar trap. Unbaited traps were filled with 300 g of soil (20% moisture). A disk of cardboard (8 cm diameter) was placed in the top of each trap (baited and unbaited), covered with a thin layer of soil, and the lid was then screwed onto the trap.
- Field sites fence posts infested with termites (Reticulitermes tibialis) were used for field trapping experiments at three different ranches in Colorado (Fort Collins, Nunn, and Akron). Each infested fence post was used as a point source for an experiment. Six traps, three baited and three unbaited, were placed in the ground evenly around the fence post at a distance of 1 meter. The traps were placed in the ground at a depth of 20 to 25 cm and covered completely with soil. Traps were checked weekly for the presence of termites. Traps were checked for feeding damage on the cardboard disks. Cardboard disks were taken back to the laboratory, where each piece was carefully washed and spread out to dry. The amount of cardboard eaten was determined by scanning the pieces with a desktop scanner and calculating the area by using a computer graphics program (Adobe Photoshop). The experiment was continued for six weeks at each location. Results:
- Jar traps were constmcted from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 1 in a jar trap. Unbaited traps were filled with 300 g of soil (20% moisture).
- Field sites fence posts infested with termites (Reticulitermes tibialis) were used for field trapping experiments at three different ranches in Colorado (Fort Collins, Nunn, and Akron). Each infested fence post was used as a point source for an experiment. Six traps, three baited and three unbaited, were placed in the ground evenly around the fence post at a distance of 2 meters. The traps were placed in the ground at a depth of 20 to 25 cm and covered completely with soil. Traps were checked weekly for the presence of termites. Traps were checked for feeding damage on the wood squares. Wood squares were taken back to the laboratory, washed with water, and spread out to dry. The dried wood squares were weighed to determine the amount that had been eaten. The experiment was continued for six weeks at each location.
- Example 6 Composition of Formulation 2 (Dried Ground Germinated Corn Seeds):
- Com seeds were soaked in soapy water overnight, rinsed well and germinated in a covered plastic tub containing moist germination paper. After 3 days of germination, the germinating com was ground to meal using a kitchen food processor, than spread out on trays and allowed to air dry overnight. Dried, ground, germinated com seed (12 g per 100 g soil) was added to soil that contained 20% moisture.
- Jar traps were constructed from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 2 in ajar trap. Unbaited traps were filled with 300 g soil (20% moisture). A pre- weighed square of Ponderosa pine (4 x 4 x 0.5 cm) was soaked in water for 15 minutes and was placed in the top of each trap (baited and unbaited), covered with a thin layer of soil, and the lid was then screwed onto the trap.
- Effervescent tablets comprised of 50:50 citric acid:sodium bicarbonate were obtained from a local grocery store (Fizzies brand drink tablets, Premiere Innovations, Pacific Palisades, CA 90272). Two tablets (3 g each) were added to soil (300 g) that contained 20% moisture.
- Jar traps were constmcted from 16 ounce polyethylene jars with plastic screw caps. Each jar was drilled with 36 evenly-spaced holes (3 mm diameter) to allow volatiles to diffuse out of the trap and to allow termites to enter. A cylindrical basket was constmcted for each cup trap from plastic fencing to facilitate removing the trap from the soil. Baited traps were prepared by placing 300 g of Formulation 5 in a jar trap. Control traps were filled only with 300 g soil (20% moisture). A square of Ponderosa pine (4 cm by 4 cm by 0.5 cm width) that had been pre- weighed was moistened by soaking it in water for 15 minutes, then placed in the top of each trap (baited and unbaited) just below the surface of the soil.
- Field sites fence posts infested with termites (Reticulitermes tibialis) were used for field trapping experiments at three different ranches in Colorado (Fort Collins, Nunn, and Akron). Each infested fence post was used as a point source for an experiment. Six traps, three baited and three unbaited, were placed in the ground evenly around the fence post at a distance of 2 meters. The traps were placed in the ground at a depth of 20 to 25 cm and covered completely with soil. Traps were checked weekly for the presence of termites. Traps were checked for feeding damage on the wood squares. Wood squares were taken back to the laboratory, washed with water, and spread out to dry. The dried wood squares were weighed to determine the amount that had been eaten. The experiment was continued for six weeks at each location.
- Bioassay apparatus The choice-test bioassay apparatus consisted of two traps, one filled with a CO 2 -generating formulation mixed in soil and the other filled with soil alone. Traps were constmcted from 1 ounce plastic nut cups with a 1 mm hole drilled in the top and three pin holes drilled at equal intervals around the cup
- the tub was placed on one shelf of a small wood shelf unit whose base is constructed of 10 cm thick foam rubber. After 15 minutes, the shell vial cap was gently tipped over, releasing the termites. A curtain was pulled in front of the shelves to provide dim lighting. After 24 hours, the tub was removed, each cup was gently disassembled and the termites counted. Termites were not reused in subsequent tests. All 12 formulations were tested using Reticulitermes tibialis (20 replicates) and 4 of the formulations were tested using Reticulitermes virginicus (10 replicates).
- 100 g soil is listed below. For each experiment, one cup was filled with 25 g moist soil (20% water). The other cup was filled with formulation/soil mixture (25 g total). A circle of corrugated cardboard (3 cm diameter) was moistened with water, blotted lightly and placed on top of soil. The lid was put on and the cups were inverted.
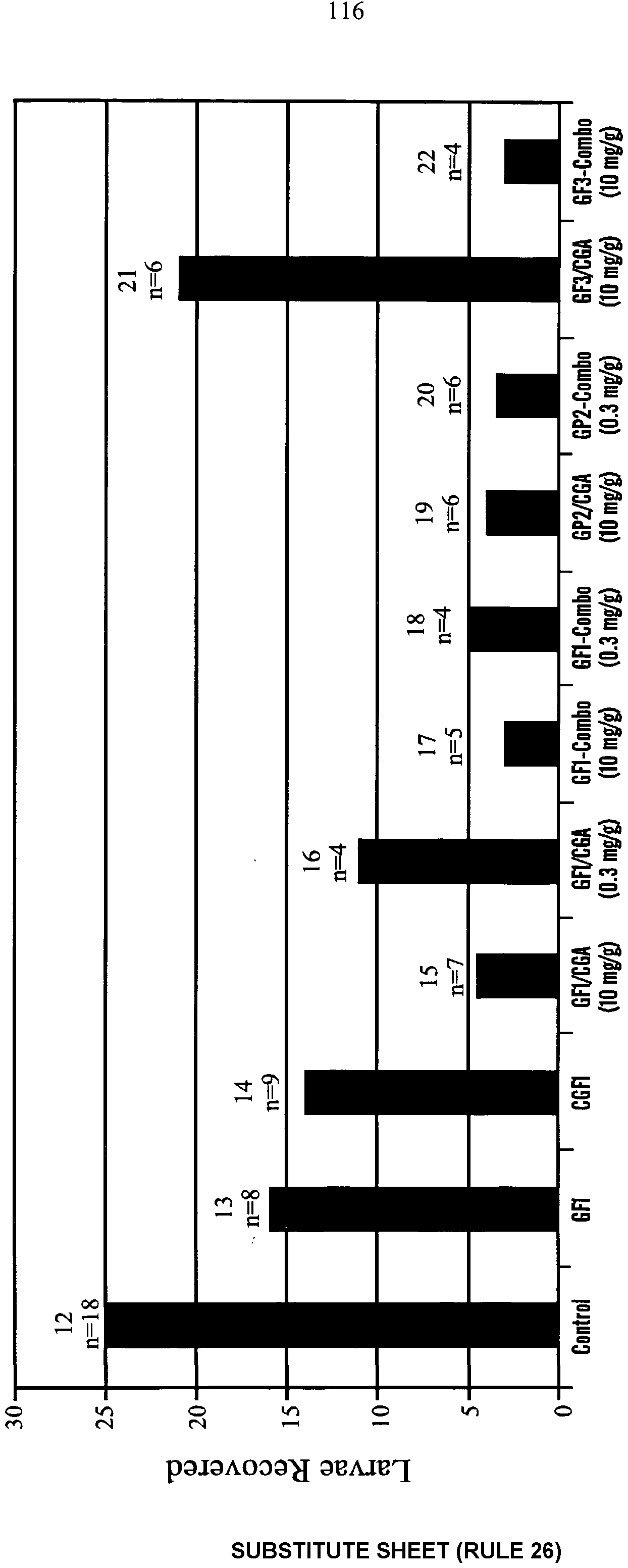
- Formulation 1 Dried Spent Grain (0.5 g per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for both species of termites (Graph 8). The average CO 2 concentration at the start of the bioassay was 6.48 mmol per mol (Graph 8).
- Formulation 2 Dried Ground Germinated Com Seeds (0.5 g per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). Slightly more termites were recovered from the treated cups than the controls in tests with Reticulitermes virginicus. The average CO 2 concentration at the start of the bioassay was 5.55 mmol per mol (Graph 8).
- Formulation 3 Whole, malted barley (0.5 g per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). Slightly more termites were recovered from the treated cups than the controls in tests with Reticulitermes virginicus. The average CO 2 concentration at the start of the bioassay was 3.7 mmol per mol (Graph 8).
- Formulation 4 Sucrose pellets with a light wax coating (0.5 g per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). The average CO 2 concentration at the start of the bioassay was 5.22 mmol per mol (Graph 8).
- Formulation 5 Effervescent tablets (Fizzies brand drink tablets, 0.25 g per 25 g soil): There was no significant difference in the number of termites recovered from the treatment and the control for Reticulitermes tibialis (Graph 8). The average CO 2 concentration at the start of the bioassay was 38.19 mmol per mol (Graph 8).
- Formulation 6 Yeast Granules (made from com flour, com syrup, NYPD nutrient broth and baker's yeast, 0.5 g granules per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). There was no significant difference in the number of termites recovered from the treatment and the control for Reticulitermes virginicus. The average CO 2 concentration at the start of the bioassay was 5.60 mmol per mol (Graph 8).
- Formulation 7 Dry Baker's Yeast (0.25 g granules per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). The average CO 2 concentration at the start of the bioassay was
- Formulation 8 Potassium Bicarbonate, Fine Granules (0.25 g granules per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). The average CO 2 concentration at the start of the bioassay was 16.71 mmol per mol (Graph 8).
- Formulation 9 Clean Cracked Com (sold as livestock feed) (0.5 g granules per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis (Graph 8). The average CO 2 concentration at the start of the bioassay was 4.21 mmol per mol (Graph 8).
- Formulation 10 Ground Dry Com Seed (0.5 g granules per 25 g soil): Significantly more termites were recovered from the treated cups than the controls for Reticulitermes tibialis. The average CO 2 concentration at the start of the bioassay was 4.48 mmol per mol (Graph 8).
- Formulation 11 Ground Malted Barley (0.5 g granules per 25 g soil): There was no significant difference in the number of termites recovered from the treatment and the control for Reticulitermes tibialis (Graph 8). The average CO 2 concentration at the start of the bioassay was 8.31 mmol per mol (Graph 8).
- Formulation 12 Baking Powder/Com Syrup Granules (0.5 g granules per 25 g soil): These granules were made from double-acting baking powder and com syrup. Significantly more termites were recovered from the treated cups than the controls for
- Example 9 Composition of Formulation 1: Dried spent brewer's grain was obtained from a local brewery, and was spread out and allowed to air dry overnight. Dried spent grain (12 g per 100 g soil) was added to soil that contained 20% moisture.
- Control traps contained perforated plastic sleeves filled with a strip of Dow Sentricon Wood and 150 g soil (20% moisture).
- Example 10 Composition of Formulation 2: Com seeds were soaked in soapy water overnight, rinsed well and germinated in a covered plastic tub containing moist germination paper. After 3 days of germination, the germinating com was ground to meal using a kitchen food processor, then spread out on trays and allowed to air dry overnight. Dried ground germinated corn seed (12 g per 100 g soil) was added to soil that contained 20% moisture.
- Baited 70 traps were prepared by placing a strip of Dow Sentricon Wood (18 cm by 2.5 cm by 0.5 cm) inside a perforated plastic sleeve, then adding 150 g of Formulation 2. This thinner strip of
- Bait Station Control traps contained perforated plastic sleeves filled with a strip of Dow Sentricon Wood and 150 g soil (20% moisture).
- Control traps contained perforated plastic sleeves filled with a strip of Dow Sentricon Wood and 150 g soil (20% moisture).
- Field sites fence posts infested with termites (Reticulitermes tibialis) were used for field trapping experiments at two ranches in Colorado (Fort Collins and Nunn). Each infested fence post was used as a point source for an experiment. Six traps were placed in the ground evenly around each infested fence post at a distance of 1 meter: 1. Two baited traps, containing bait plus soil, with 1 strip of Dow wood
- the choice-test bioassay apparatus was constmcted from a glass T-tube (5 mm inside diameter, 5 mm stem, with each branch
- CO 2 concentrations reached equilibrium after 15 min.
- the CO 2 concentration of the syringes was determined by using GC-MS-SIM analysis (see below) before each bioassay. Bioassays were conducted with both Reticulitermes tibialis and
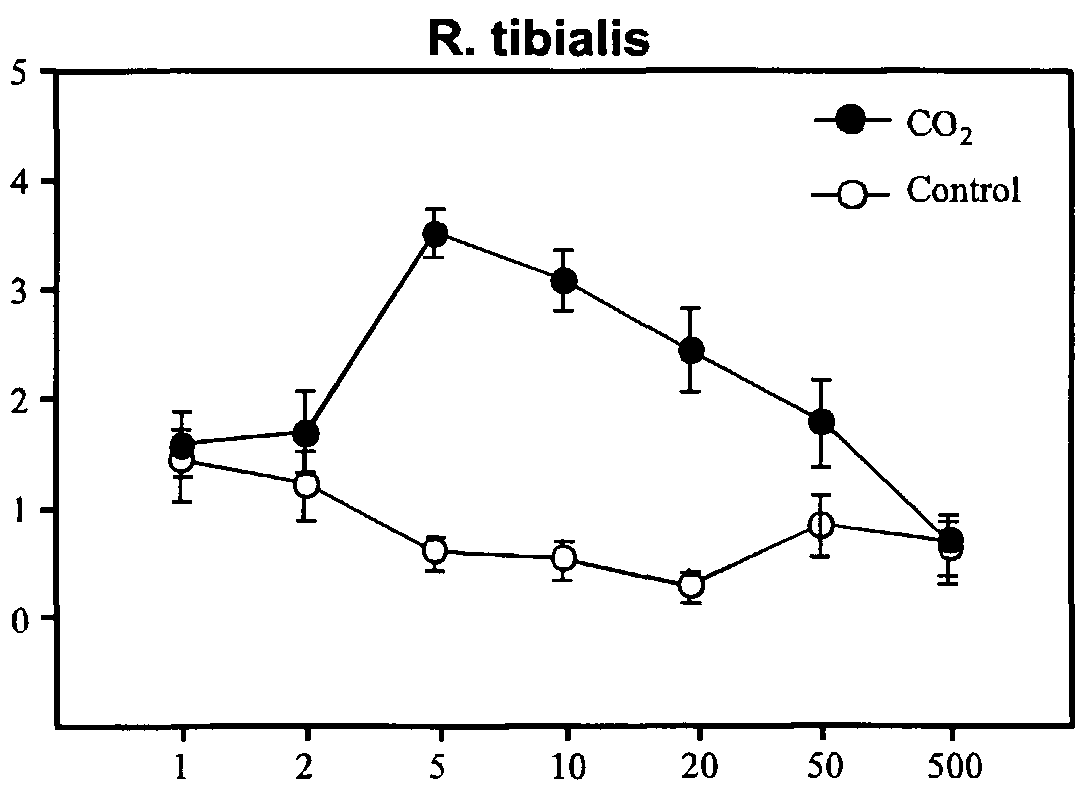
- Reticulitermes ⁇ avipes for 1, 2, 5, 10, 20, 50 and 500 mmol per mol concentrations of CO 2 and with Reticulitermes virginicus for 5, 10, 20, and 50 mmol per mol.
- the T-tube apparatus was assembled and clamped horizontally on top of a block of foam rubber (12 by 12 cm) with a wire bent into a U-shape.
- the syringe pump was turned on, and after 3 min of pumping, the cellophane seal was removed from the holding container and the entrance to the holding container was gently connected to the central arm of the T-tube, allowing termites to crawl out and enter the apparatus. Bioassays were conducted for 15 min, after which the number of termites in each pitfall was recorded.
- CO 2 measurements Gas chromatography-mass spectrometry in selected ion monitoring mode (GC-MS-SIM) at m/e 44 was used to determine CO 2 concentrations.
- GC-MS-SIM Gas chromatography-mass spectrometry in selected ion monitoring mode
- a 10- mmmol/mol mixture of CO 2 (a 300-ml glass bottle into which 3 ml of CO 2 was injected) was used as a standard to calculate the CO 2 concentrations of the unknown samples.
- Reticulitermes virginicus was attracted to 5, 10, 20 and 50 mmol per mol CO 2 .
- R. virginicus demonstrated best attraction to 5 mmol per mol CO 2 (example 12, page 4).
- Each infested fence post was used as a point source for an experiment.
- Six traps were placed in the soil evenly around a wood stmcture at a distance of 1 meter. For each experiment three of the traps contained 2 charred wood strips and three of the traps (controls) contained 2 uncharred wood strips. Traps were checked weekly for the presence of termites and feeding damage on the wood, for a period of 7 weeks.
- Treated (Charred) Wood A strip of Dow Wood (18 x 2.5 x 1 cm) was removed from a new Dow Sentricon Bait Station and cut into two pieces (9 x 2.5 x 1 cm). The surfaces of one piece were charred using a laboratory torch (propane and oxygen) with a three inch outer flame cone and one inch inner flame cone. The strip of wood was held in the flame and removed just prior to the point of ignition. All surfaces of the charred Dow Sentricon Wood strip were charred except for the top 1 cm of the wood strips. Prior to placing the strips in the bioassay device, the strips were moistened in separate water baths for several minutes. Charred and uncharred pieces of Ponderosa pine (2 x 4 x 7.5 cm) were tested in the same way.
- the lid was removed after one hour and the termites were released into the soil at the end of the tub opposite the wood bait.
- the lid was replaced on the tub, and the tub was placed in a dimly lighted area of the lab for one week. After one week the tub was inspected for termite activity near each piece of wood. After two weeks the tub was taken apart and the wood was cleaned and inspected for feeding damage.
- Example 15 Wood Impregnated with Aqueous Extract of Formulation 1:
- a plastic bowl with a snap-fit lid (Rubbermaid, 6 cup size) was filled with 24 ounces of water and 24 ounces of Formulation 1 (dried spent brewer's grain). This was mixed well and several pieces of Dow Wood (9 x 2.5 x 1 cm) were added to the bowl. The bowl was covered with the snap-fit lid and heated in a microwave oven for 2 minutes, which brought the liquid to a boil.
- the bowl was removed from the microwave oven, the contents of the bowl were stirred, the snap-fit lid was replaced on the bowl (with 4 small pin holes in lid for breathing), and the covered bowl was allowed to stand for 3 days. After 3 days, the pieces of wood were removed, rinsed sparingly with water to remove physical debris, and placed on paper towels to dry for 2 days. The extract- impregnated pieces of wood were moistened before placement in the bioassay.
- End-to-End Choice Test Bioassay A rectangular plastic tub (15 x 10 x 30 cm) was evenly partitioned into three separate sections, with two partitions made from the cut ends of another tub hot melt glued into the main tub. The partitions were drilled with fourteen 1/8 inch holes such that the holes were all below the soil surface and evenly arranged top-to-bottom and side-to-side. The tub was filled with 6 lbs. of soil (20% moisture by weight), evenly in the three sections. This amount of soil allowed for a level of soil 2.5 cm from the top of the tub. Two pieces of wood, one treated and one untreated, were placed at opposite ends of the tub, 0.5 cm from the end of the tub and 10 cm from the partition.
- the treated and untreated wood pieces were set upright, one at each end of the tub, and inserted into the soil nearly touching the bottom of the tub, resulting in a thin layer of soil between each piece of wood and the bottom of the tub, and with the upper 4 cm of each wood piece extending above the surface of the soil.
- One hundred termites were held in a petri dish for one hour in the closed assay apparatus in order to become acclimated to their new environment.
- the lid was removed after one hour and the termites were released into the soil at the center of the tub.
- the lid was replaced on the tub, and the tub was placed in a dimly lighted area of the lab for one week. After one week the tub was inspected for termite activity near each piece of wood. After two weeks the tub was taken apart and the wood was cleaned and inspected for feeding damage. Results:
- Teflon tubing conveyed the odors to the two arms of the T-tube, at 1.0 ml/min into each arm.
- the body of the T-tube was mounted horizontally on a foam rubber block.
- a group of 5 termites was placed inside a small Teflon holding tube for 15 min. To allow them to acclimate to the bioassay environment (NMR caps with small holes in them to allow gas flow were used to plug the ends of the holding tube).
- the termite moved along the arm about 2 cm to here the dropped off at 45 degrees, and slid down the chute into the pitfall.
- the number of termites that was attracted to the CO 2 side of the bioassay was significantly greater than the number that moved to the control side.
- a behavioral bioassay was used to demonstrate that termites are attracted to CO 2 .
- the termites chose the 5 mmol/mol CO 2 side significantly more often.
- the bioassay apparatus was constmcted from a horizontal glass T-tube with the ends of the choice arms bent downward at 45 to provide pitfalls.
- a syringe pump was used to provide slow, consistent delivery of candidate compounds to the two sides of the choice-test.
- Termite collections Termites were collected at three different sites in Larimer County: Big Hill Overlook, Lone Pine Wildlife refuge, and Poudre Canyon in the early part of June 1997.
- the termites were captured in one of two ways. Big Hill termites were captured using traps consisting of a square wood frame (6x6') made of lxl untreated wood. In the center of the frame was a piece of doubly corrugated wood cut to fit the frame. The cardboard was held in by a wire mesh with 1/4 inch holes. The traps were left for two weeks, in a spot where termites were seen. The termites were then removed from the traps and placed in petri dishes (see below). The second method (Lone Pine, and Poudre Canyon) was to look under logs and rocks.
- Bioassay Apparatus The choice-test bioassay apparatus was constmcted from a glass T-tube (5 mm inside diameter, 5 mm stem, with each branch 4.5 cm long). Each branch of the 'T' was bent downward (2 cm from junction of the T) at a
- a 5 mm NMR cap (cat. no. 100-0050, Drummond Scientific, Broomall, PA) with a 1 mm pinhole in it was firmly pushed over the end of each bent branch.
- a 25 cm length of Teflon tubing (0.8 mm ID) was inserted (3 mm) into the pinhole of each NMR cap and the other end of the tubing was connected to a 35 ml polyethylene syringe (cat no. 106-0490, Sherwood Medical, St. Louis, MO).
- the two 35-ml polyethylene syringes used for each bioassay were connected to a syringe pump (Sage Model 355, Fisher Scientific, Pittsburgh, PA) which was adjusted to provide an airflow of 1.0 ml/min into each choice arm of the bioassay apparatus.
- Bioassay Procedure For bioassays, termite workers were collected using a camel-hair brush from a petri dish containing moist paper towels and cardboard, and were placed in a holding container constmcted from a 3 cm length of Teflon tubing (8 mm ID). The container was plugged at one end with a NMR cap with two holes (1 mm) drilled in the bottom. A second NMR cap with a 4 mm hole drilled through it was inserted backwards into the other end of the Teflon tube. The NMR cap was then sealed with a small square of cellophane held in place with a plastic tube (a piece of plastic soda straw) that fit snugly over the open end. Termites (5 workers) were placed in the container and the top was sealed. The container was placed on its side
- the T-tube apparatus was assembled and clamped horizontally on top of a block of foam rubber (12 cm x 12 cm) with a wire bent into a U-shape.
- the syringe pump was set to provide a flow of 1.0 mvmin from each syringe, and each syringe was connected with Teflon tubing to one choice arm of the T-tube.
- a flow meter was used to verify that the flow exiting the central arm of the T-tube was 2.0 ml/min, confirming the flow of volatiles through the apparatus, and verifying that there were no leaks in the connections. If the flow was inadequate, all connections were inspected and/or secured, and the flow was rechecked.
- a Hewlett-Packard Series II 5890 gas chromatograph interfaced with a Hewlett-Packard 5971 mass selective detector was operated in selected ion monitoring mode (SIM) for m/e 44 with a methyl silicone capillary column (30 m x 0.32 mm ID, RSL-150, Alltech, Inc.).
- SIM selected ion monitoring mode
- a 10 mmol/mol mixture of CO 2 (a 300 ml glass bottle into which 3 ml of CO 2 were injected) was used as a standard to calculate the CO 2 concentrations of the unknown samples.
- CO 2 Bioassay A 5 mmol/mol concentration of CO 2 was used to test termite attraction. A 35 ml polyethylene syringe was rinsed with distilled water to moisten the inside of the syringe, and partially filled (approximately 5 ml) with ambient air.
- CO 2 (100 microliters) was obtained with a glass syringe from a tank containing pure (1 00%) CO 2 and injected into the 35 ml polyethylene syringe. Ambient air was then drawn into the syringe to fill it to a total volume of 35 ml, mixing the air and CO 2 thoroughly by turbulence. The gas mixture in the syringe was allowed to equilibrate for 15 minutes, and GC-MS-SIM was used to verify the CO 2 concentration prior to each bioassay. A second 35 ml polyethylene syringe was filled with ambient air for a control, and the CO 2 concentration was measured using GC-MS-SIM.
- a behavioral bioassay was developed to test responses of newly hatched (neonate) larvae of western com root worm Diabrotica virgifera virgifera LeConte to volatile compounds from com plants, a major host for this insect.
- a glass Y-tube filled with glass beads was used to allow choice tests in a vertical direction and to reproduce the thigmotactic cues available to larvae in their natural soil environment.
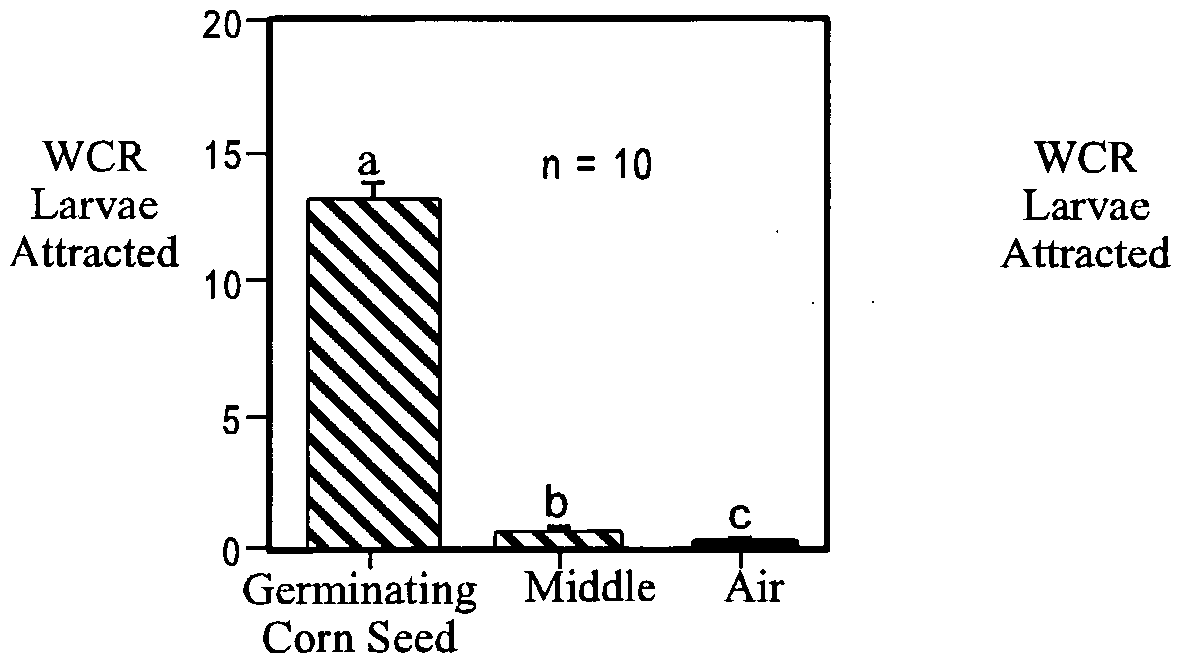
- a syringe pump was used to provide slow, consistent delivery of candidate compounds to the 2 sides of the apparatus. Significantly more larvae were attracted to the side containing a germinating com seed than to the side containing ambient air.
- the concentration of CO 2 in soil that contained no com was 1.38 ⁇ 0.03 mmol/mol and the concentration in ambient air was 0.94 ⁇ 0.01 mmol/mol.
- CO 2 carbon dioxide
- Stmad et al. (1986) first reported that western com root worm larvae are highly attracted to CO 2 , and subsequent investigators have confirmed this attraction (Hibbard and Bjostad 1988, MacDonald and Ellis 1990, Stmad and Dunn 1990, Jewett and Bjostad 1996).
- Carbon dioxide alone is attractive to a number of soil invertebrates, including insect larvae (Klingler 1957, 1958, 1959, 1961, 1965, 1966; Paim and Beckel 1963b; Stadler 1971, 1972; Meeking et al. 1974; Doane et al. 1975; Jones and Coaker 1977, 1979), insect adults (Paim and Beckel 1963a, b), mites (Moursi 1962, 1970), chilopods (Moursi 1970), nematodes (Johnson and Niglierchio 1961; Klingler 1961,
- the minimum concentration of CO 2 required for attraction of western com root worm larvae and the concentration for optimal attraction have not previously been determined.
- the objectives of the current study were to determine threshold concentrations of CO 2 for attraction of western com root worm larvae and to determine the range of concentrations attractive to the larvae. If western com root worm larvae are given a choice between a high and a low concentration of CO 2 , the difference in concentration required to elicit a significant difference in attraction would be expected to increase as both concentrations are increased, and we tested this hypothesis as well.
- the plants typically reached a shoot length of 1 cm and a root length of 6 cm.
- the choice-test bioassay apparatus (Graph 18-1 -A) was constmcted from a glass Y-tube filled with glass beads to simulate the thigmotactic cues of the soil environment that are ordinarily encountered by western com root worm larvae.
- the glass Y-tube was fabricated by a local glassblower (9.5 mm inside diameter, 60° angles, with each branch 3 cm long), and clamped to a ring stand with 2 branches of the "Y" facing down.
- a glass connection tube (4 cm long, 0.5 cm diameter) with a piece of vinyl screen (2.5-mm mesh) held over 1 end by a 0.5-cm section of Teflon tubing (6 mm inside diameter) was inserted snugly into the end of each of the arms of the Y-tube to support the glass beads.
- Glass beads (3 mm, cat. no. 11-312A, Fisher Scientific, Pittsburgh, PA) were poured into the top of the Y-tube, filling the entire apparatus to within 0.5 cm of the top (250 beads).
- a 5-mm NMR tube cap (cat. no.
- a 2nd piece of slender Teflon tubing was used to connect the shell vial to 1 arm of the bioassay apparatus.
- the 2 syringes used for each bioassay were connected to a syringe pump (Sage Model 355, Fisher Scientific, Pittsburgh, PA) that provided an airflow through each shell vial containing a candidate chemical treatment, and subsequently into a choice arm of the bioassay apparatus.
- a syringe pump Sage Model 355, Fisher Scientific, Pittsburgh, PA
- the shell vial containing either a carbonated water dilution or a com seed or cut com roots was left open for 5 min to allow the gas concentrations to reach equilibrium.
- the vial was capped, and the syringe pump was started, providing an airflow of 1.0 ml/min from each syringe.
- the Y-tube apparatus was assembled and filled with glass beads and the appropriate treatment and control sources (shell vials or syringes) were connected to the arms of the Y-tube.
- the syringe pump was set to provide a flow of 1 ml/min and turned on.
- a flow meter was used to verify that the flow exiting the top of the Y-tube was 2 ml/min, confirming the flow of volatiles through the apparatus and verifying that there were no leaks in the connections. If the flow was inadequate, all connections were inspected and secured, and the flow was rechecked.
- Germinating Corn Seed Versus Air Using the shell vial source technique, germinating com seeds were tested to determine whether larvae could detect volatile compounds produced by the growing seeds and follow them through a glass bead medium to the source. Individual washed com seeds were placed in glass shell vials (4 ml) with a moistened piece of filter paper inside. The vials were placed on moistened germination paper inside a covered plastic container (30 by 15 cm) and germinated for 3 d. A vial containing a single 3-d-old germinating seed was removed from the covered plastic container just before testing and connected to the bioassay apparatus. An empty shell vial was connected to the other side as a control. The CO 2 concentrations of the germinating com seeds and the control were determined by using GC-MS-SIM.
- Cut Corn Roots Versus Air In a companion experiment, cut com roots were tested to determine whether larvae were attracted to volatile compounds produced by the roots alone. Com roots (14.5 cm, 3 d old) were cut into 2—3 cm lengths and placed into 1 shell vial. The other shell vial (control side) contained ambient air. The CO 2 concentrations of the cut com roots and the control were determined by using GC-MS-SIM.
- Corn Headspace Bioassay Using the syringe source technique, the headspace over germinating com seedlings was tested to determine the larval response to com volatiles in the glass bead apparatus. Washed com seeds were spread on moistened germination paper inside a covered plastic container (30 by 15 cm) and germinated for 3 d to allow volatile com compounds to be produced. A 35- ml polyethylene syringe was filled with the headspace containing these volatile compounds by means of a 25 cm length of slender Teflon tubing inserted into a hole drilled into the cover. The control syringe was filled from an identical plastic container containing only moistened germination paper. The CO 2 concentrations of the syringes were determined by using GC-MS-SIM before each bioassay.
- Consistency of CO 2 Delivery The consistency of the CO 2 concentration delivered into the bioassay apparatus was measured using GC-MS-SIM.
- syringe sources a 35-ml polyethylene syringe was partially filled with ambient air (5 ml) and 80 ⁇ l of CO 2 (obtained with a glass syringe from a tank containing pure [100%] CO 2 ) was injected into the syringe. Ambient air was then drawn into the syringe to fill it, mixing the air and CO 2 thoroughly by turbulence at the same time.
- the syringes were allowed to equilibrate for 30 min before they were connected to the syringe pump (set at a flow of 1 ml/min). After 3 min of pumping, a 2- ⁇ l sample was taken from 5 cm inside a 20-cm length of Teflon tubing exiting from each syringe, by using a 10- ⁇ l (Hamilton) syringe. To test consistency of CO 2 release from the syringes, samples were taken at 0, 10, 20, and 30 min (following the initial
- CO 2 Bioassay In a preliminary experiment, a 10-mmol/mol concentration of CO 2 was used to test larval attraction. A 35-ml polyethylene syringe was rinsed with distilled water to moisten the inside of the syringe, and partially filled (5 ml) with ambient air. The CO 2 (350 ⁇ l) was obtained with a glass syringe from a tank containing pure (100%) CO 2 and injected into the 35-ml polyethylene syringe.
- Ambient air was then drawn into the syringe to fill it to a total volume of 35 ml, mixing the air and CO 2 thoroughly by turbulence.
- the gas mixture in the syringe was allowed to equilibrate for 15 min, and GC-MS-SIM was used to verify the CO 2 concentration before each bioassay.
- a 2nd 35-ml polyethylene syringe was filled with ambient air for a control, and the CO 2 concentration was measured using GC-
- CO 2 Selective Response Pairs of CO 2 mixtures were tested to determine if the larvae could detect small differences in CO 2 concentration.
- a syringe containing 1 mmol/mol CO 2 was connected to 1 arm of the Y-tube, and a syringe containing 1.5 mmol/mol CO 2 was connected to the opposite arm of the Y- tube.
- comparisons were made for 2 versus 2.5 mmol/mol, 5 versus 5.5 mmol/mol, 10 versus 10.5 mmol/mol, and 20 versus 20.5 mmol/mol CO 2 .
- the CO 2 concentration of the syringes was determined by using GC-MS-SIM analysis before each bioassay.
- Comparisons were made for 1 versus 1.25, 2 versus 2.25, 5 versus 5.25, 10 versus 10.25, and 20 versus 20.25 mmol/mol CO 2 , for 1 versus 1.125, 2 versus 2.125, 5 versus 5.125, 10 versus 10.125, and 20 versus 20.125 mmol/mol CO 2 , and also for 1 versus 1, 2 versus 2, 5 versus 5, 10 versus 10, and 20 versus 20 mmol/mol of CO 2 .
- each dilution of carbonated water (1 ml) was slowly dispensed into a shell vial (4 ml capacity) with a 1-ml Pasteur pipette. Distilled water (1 ml) was placed into a 2nd vial (control). The vials were left open for 5 min to allow the CO 2 gas concentration to reach equilibrium, then were connected to the bioassay apparatus.
- the CO 2 concentration in the headspace above the carbonated water dilutions in the shell vials was determined by using GC-MS-SIM.
- Shell Vial Control Bioassays Control tests with air on both sides of the Y- tube and with carbonated water on both sides of the Y-tube were conducted to determine if there was an intrinsic tendency for the larvae to move to 1 side or the other when chemical cues were absent, or when CO 2 was present.
- shell vials containing ambient air were connected to both arms of the Y-tube.
- Measurements of CO 2 were taken from the soil when the plants were 6—8 d old.
- a piece of metal wire (5.3 cm) was inserted into a glass tube (5 cm long, 1 mm inside diameter) so that the wire projected 3 mm from the end of the glass tube.
- the tube was inserted, wire first, 4 cm into the soil.
- the wire plug was removed from the glass tube, leaving a 3-mm gap in the soil just below the end of the glass tube.
- the needle of a 10- ⁇ l Hamilton syringe was inserted into the glass tube so that it projected 1 mm into the gap, and a 5- ⁇ l sample of soil headspace was removed. Samples were taken from different locations in the tub to minimize disturbance of the soil CO 2 concentrations.
- the CO 2 concentration of the soil headspace was determined by using GC-MS-SIM. Using the same method, samples were taken from control tubs containing soil alone.
- the CO 2 concentration of the headspace above germinating com roots was 2.97 ⁇ 0.15 mmol/mol, and the CO 2 concentration of the headspace on the control side was 0.99 ⁇ 0.08 mmol/mol (Graph 18-1-E).
- Corn Headspace Bioassay In bioassays with syringe sources, significantly more western com root worm larvae (P ⁇ 0.05) were attracted to the side containing the headspace over germinating com seeds than to the control side (Graph 18-2-B).
- the CO 2 concentration of the headspace above the germinating com seeds was 5.38 ⁇ 0.45 mmol/mol, and the CO 2 concentration of the headspace on the control side was 1.14 ⁇ 0.13 mmol/mol (Graph 18- 2-D).
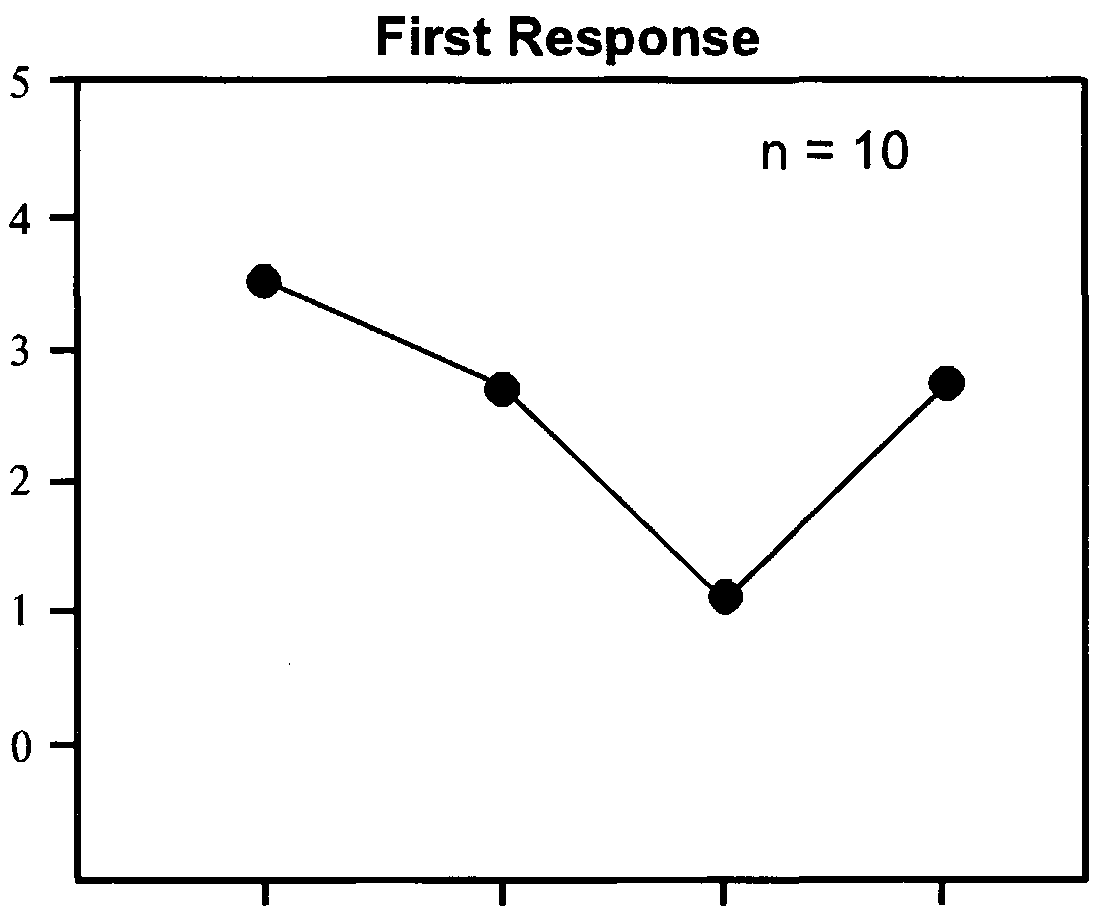
- CO 2 (Dose— Response). The larvae were attracted to a wide range of CO 2 concentrations. The lowest concentration of CO 2 that was attractive to the larvae (Graph 18-4) was 1.34 ⁇ 0.05 mmol/mol (10 ⁇ l of CO 2 added to syringe) (P ⁇ 0.05), where the control CO 2 concentration was 0.91 ⁇ 0.03. The highest dose to which the larvae were attracted was 85.60 ⁇ 1.20 mmol/mol (3 ml of CO 2 added to syringe). They were not attracted to 300 mmol/mol (10 ml of CO 2 added to syringe) or 900 mmol/mol (30 ml of CO 2 added to syringe) concentrations (Graph 18-4). CO 2 Selective Response. Significantly more larvae were attracted (Graph 18-4)
- the larvae responded optimally to the 10% dilution of carbonated water, and all concentrations (3, 10, 30, and 100%) greater than the 1% dilution were significantly more attractive (P ⁇ 0.05) than the control (distilled water).
- the CO 2 concentration of the control (distilled water) was 1.42 ⁇ 0.08 mmol/mol, and the concentration of the 1% dilution was 1.48 ⁇ 0.10 mmol/mol (Graph 18-6-B).
- the CO 2 concentration of the 3% dilution was 1.91 ⁇ 0.09 mmol/mol, and the 10% dilution produced 2.55 ⁇ 0.12 mmol/mol of CO 2 .
- the 30% dilution produced 6.06 ⁇ 0.36 mmol/mol of CO 2 , and the 100% carbonated water produced 24.49 ⁇ 0.22 mmol/mol of CO 2 .
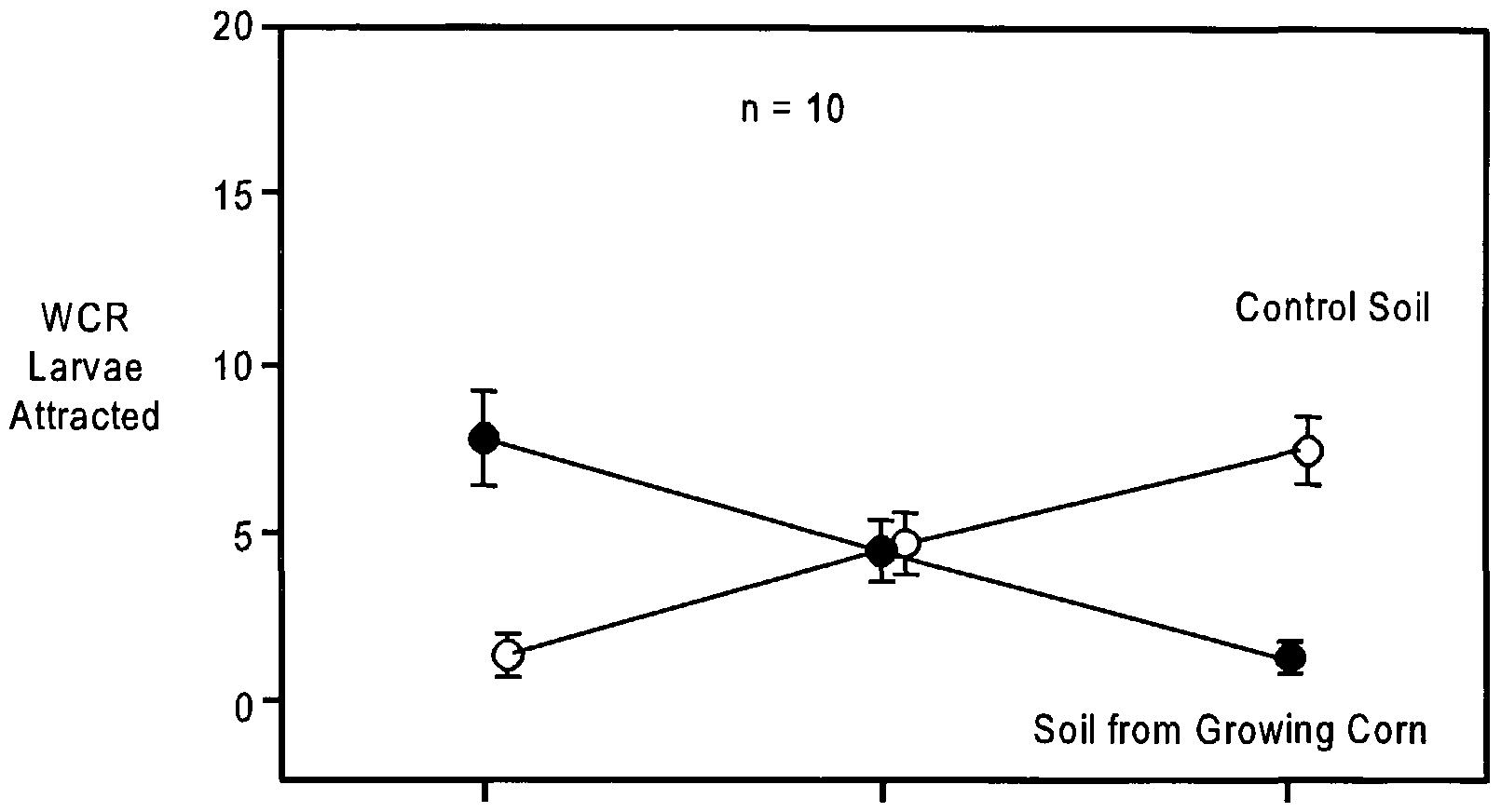
- CO 2 Analysis of Corn Plants in Soil The CO 2 concentration in the soil atmosphere in tubs containing 8-d-old growing com plants was 4.36 ⁇ 0.31 mmol/mol (measured by GC-MS-SIM). The concentration of CO 2 in tubs containing soil alone was 1.38 ⁇ 0.03 mmol/mol, and the concentration in the ambient air was 0.94 ⁇ 0.01 mmol/mol.
- the larvae exhibited a tendency to move downward. They moved in a downslope direction when placed on a flat, slightly tilted surface (petri dish), and also moved downward when they were allowed to move through a porous, soil-like medium such as glass beads.
- the larvae appeared to use thigmotactic cues to maneuver. When placed in the center of a small (5 cm) petri dish, the larvae quickly moved to the outside of the dish and continued to crawl around the circumference of the dish, keeping their bodies in contact with the outside edge at all times.
- the new bioassay design accommodates the small size of the neonate larvae, provides a choice in the vertical direction, and uses glass beads to simulate the thigmotactic cues that are ordinarily encountered by western com root worm larvae in their natural soil environment.
- the glass bead apparatus also can be adapted to facilitate the testing of a variety of chemical sources.
- gaseous mixtures of CO 2 were shown to attract newly hatched western com root worm larvae in this behavioral bioassay, and the headspace above diluted carbonated water also was found to be attractive.
- Neonate larvae exhibited a positive chemotactic response to CO 2 in the glass bead bioassay similar to that demonstrated previously using other bioassay designs (Stmad et al. 1986, Hibbard and Bjostad 1988, MacDonald and Ellis 1990, Jewett and Bjostad 1996).
- the larvae were able to detect and were attracted to levels of CO 2 as small as 1.34 ⁇ 0.05 mmol/mol when the control (ambient air) contained 0.91 ⁇ 0.03 mmol/mol.
- the larval response to CO 2 increased with each increase in the amount of CO 2 added to the syringe mixtures (1, 3, 10, .... ⁇ l of CO 2 ) (Graph 18-4) when the control side contained 1.00 ⁇ 0.09 mmol/mol of CO 2 .
- the attractive range of concentrations was from 1.34 ⁇ 0.05 to 85.6 ⁇ 1.20 mmol/mol.
- the most attractive concentrations of CO 2 were 2.51 ⁇ 0.13 mmol/mol (30 ⁇ l of CO 2 added to the syringe), and 4.20 ⁇ 0.21 mmol/mol (100 ⁇ l added to the syringe).
- Western com root worm larvae may use their ability to detect differences in concentration to orient directly to the root of the com plant and avoid losing valuable time searching the entire area in which the roots are growing.
- Sources of CO 2 include carbonated water.
- Sufficient CO 2 gradients can be produced by granules of potassium bicarbonate coformulated with an acid and a pesticide that are broadcast or incorporated into the soil.
- Graph 18-1 (A) Glass bead bioassay apparatus with candidate chemical cues in shell vials. (B) Choice test bioassay with a germinating com seed versus air. (C) Choice test bioassay with cut com roots (0.34 g) versus air. (D) CO 2 concentrations (measured with GC-MS-SIM) of germinating com seed and air in shell vials. (E) Concentrations of CO 2 (measured with GC-MS-SIM) of cut com roots and air in shell vials. Significant differences (p ⁇ 0.05) are indicated by different lower case letters.
- Graph 18-2 (A) Glass bead bioassay apparatus with candidate chemical cues in syringes. (B) Choice test bioassay with headspace over germinating com seeds versus air. (C) Choice test bioassay with CO 2 (10 mmol/mol) versus air. (D) Concentrations of CO 2 (measured with GC-MS-SIM) of headspace over germinating com seeds and air in syringes. (E) Concentrations of CO 2 (measured with GC-MS- SIM) of CO 2 (10 mmol/mol) and ambient air in syringes. Significant differences (p ⁇ 0.05) are indicated by different lower case letters. Bars represent standard errors. WCR, western com root worm.
- Graph 18-7 (A) Choice-test bioassay with syringe sources containing the headspace from different dilutions of carbonated water. (B) CO 2 concentrations (measured with SIM-GC-MS) from the headspace over each dilution of carbonated water. Significant differences (p ⁇ 0.05) in attraction to a particular dose of CO 2 and its corresponding control are indicated by different lower case letters. Bars represent standard errors (many are too small to be visible).
- larvae chose equally between the 2 sides of the bioassay when volatile compounds from corn were present on 1 side and an equivalent concentration of CO 2 was present on the other side. When given a choice between corn volatiles and a higher concentration of CO 2 , the larvae chose the CO 2 side significantly more often.
- the headspace from germinating corn seeds was collected and continuously injected into 1 side of the bioassay apparatus, and a defined concentration of CO 2 was continuously injected into the other side.
- We tested the possibility that compounds of limited volatility may be involved in larval attraction by preparing glass beads coated directly with volatiles produced by germinating corn seeds, and also by testing soil that was removed from corn roots.
- the Y-tube accommodates the geotropic tendency of the larvae by allowing them to make a choice between the downward arms, and the glass beads reproduce the thigmotactic cues available to larvae in their natural soil environment.
- a syringe pump is used to provide slow, consistent delivery of candidate compounds to the 2 sides of the apparatus.
- the glass bead apparatus can be adapted to facilitate the testing of a variety of chemical sources.
- Soil Soil was obtained from a local agricultural research farm whose history was known, and where no com had been grown for 5 years.
- Bioassay Procedure All bioassays were choice tests conducted using a vertical glass "Y" tube apparatus filled with 3-mm glass beads (Bernklau and Bjostad 1998) (Graph 19-1-A). Volatile compounds were prepared in 35-ml polyethylene syringes (cat no. 106-0490, Sherwood Medical, St. Louis, MO) and a syringe pump (Sage Model 355, Fisher Scientific, Pittsburgh, PA) was used to provide, slow (1 ml per min) consistent delivery of the compounds into each choice arm of the bioassay apparatus. Twenty newly-hatched larvae (less thanl2-h-old) were used for each bioassay. Non-diapausing larvae were used for all experiments unless otherwise indicated below. For each choice test a minimum of 10 replicates were conducted.
- a 2nd 35-ml polyethylene syringe was partially filled (approximately 5 ml) from a tank containing pure (100%) CO 2 using a glass syringe. Headspace from a plastic tub containing only moist germination paper was drawn into the syringe to fill it, mixing the air and CO 2 thoroughly at the same time. The gas mixtures in the polyethylene syringes were allowed to equilibrate for 15 min, and GC-MS-SIM was used to verify the CO 2 concentrations in both syringes prior to each bioassay.
- Corn Headspace Versus CO 2 with Diapausing Larvae The larvae used in our studies were from a colony of nondiapausing western com root worm that has been maintained in our laboratory since 1986. We wished to determine if diapausing western com root worm larvae would respond differently to com volatiles than the colony larvae. Using the same method described above, the headspace over germinating com seeds was tested in a choice test against a series of CO 2 concentrations with western com root worm larvae from a diapausing strain. Corn Headspace-Coated Glass Beads versus CO 2 . In the previous experiments, com volatiles were introduced into the bottom of the Y-tube and carried through the glass beads by the airstream from the syringe pump.
- Teflon tubing was inserted into the hole in the NMR cap and the other end was connected to a 35-ml polyethylene syringe.
- the plunger was slowly drawn out, pulling the com headspace through the glass beads and filling the syringe.
- the glass tube was then removed from the com tub, the top was capped with a rubber stopper, and the bottom was sealed with a metal plug inserted into the hole in the NMR cap.
- a 35-ml polyethylene syringe was filled with 1 of 3 concentrations of CO 2 , as described previously (ambient CO 2 , CO 2 matching the concentration in the headspace over the germinating com seeds, or twice the concentration of CO 2 in the com headspace).
- the gas mixture from 1 of the syringes was pushed through a glass test tube filled with glass beads through a 25-cm length of Teflon tubing inserted into a hole in the rubber stopper capping the top.
- the hole in the NMR cap was sealed with a wire plug.
- the glass tubes containing com headspace or 1 of the CO 2 controls were uncapped and inserted into the ends of the Y-tube so that the tops were even with the junction of the ⁇ With this arrangement, com compounds of limited volatility were available to the larvae at the choice point.
- the rest of the Y-tube was filled to within 0.5 cm of the top with untreated glass beads.
- the syringe containing com headspace and the 2nd syringe containing a CO 2 mixture were connected to the ends of the Y-tube with 25-cm lengths of Teflon tubing inserted into the hole in the NMR cap.
- a 2nd 35-ml polyethylene syringe was filled (as described above) with 1 of 3 concentrations of CO 2 (ambient CO 2 , CO 2 matching the concentration in the headspace over the damaged com seeds, or twice the concentration of CO 2 in the soil headspace).
- the gas mixtures in the polyethylene syringes were allowed to equilibrate for 15 min, and GC-MS-SIM was used to verify the CO 2 concentration in both syringes prior to each bioassay.
- Soil Bioassay A variation of the bioassay apparatus containing soil was used to test larval attraction to com compounds of limited volatility that might be present in soil in which com is grown. Washed, soaked com seeds (9) were planted in a plastic tub (11 cm high, 7 cm diameter) in soil that had been sifted through a 0.32 mm mesh and through a 5 mm mesh screen (W.S. Tyler Inc., Mentor, Ohio 44060). An equal amount of soil was added to a 2nd tub as a control. Both tubs were uncovered after 3 d and the soil was used for bioassays when the com plants were 8 d old.
- the com plants were removed from the soil and the soil was examined under a microscope to remove any pieces of com roots that might remain.
- the bottom of a glass test tube (4 cm long, 8 mm diameter, with a 1.5 mm hole in the bottom) was lined with a square (1 by 1 cm) of organza cloth and the tube was filled with the soil.
- a Teflon connector was snugly fitted over the bottom end of the tube and a NMR cap
- a 2nd glass test tube was prepared, using soil from the control tub. The 2 glass tubes were inserted snugly inside the glass Y-tube so that the tops were even with the junction of the 'Y', and the rest of the Y-tube was filled to within 1 cm of the top with soil from the com tub.
- a 35-ml polyethylene syringe was filled with the headspace containing the com volatiles from the damaged com, and a 2nd 35-ml polyethylene syringe was filled with 1 of 3 concentrations of CO 2 (ambient CO 2 , CO 2 matching the concentration in the headspace over the damaged com seeds, or twice the concentration of CO 2 in the com headspace).
- the gas mixtures in the polyethylene syringes were allowed to equilibrate for 15 min, and GC-MS-SIM was used to verify the CO 2 concentration in both syringes prior to each bioassay.
- Corn Surface Extracts Surface extracts of germinating com seeds were tested for larval attraction. Germinating com seeds (3-d-old, 50 grams dry wt as determined at the end of the experiment) were firmly packed into a glass tube (30 cm long, 30 mm diameter, tapering to 12 mm diameter) and diethyl ether (glass-distilled) was dribbled through the seedlings until 8 ml of extract had been collected. The extract was concentrated to 2 ml by evaporation with a gentle stream of nitrogen. Different aliquots of the extract (0.003, 0.03, 0.1, 0.3, 3.0, and 30 gram equivalents com) were applied to a strip of filter paper (Whatman no.
- Germinating com seeds (3-d-old, 50 grams dry wt as determined at the end of the experiment) were packed into a glass tube (30 cm by 30 mml, tapering to 12 mm).
- a strip of filter paper (0.5 by 2 cm) and a boiling chip were placed in the bottom of a glass sample tube (12 mm by 35 cm, closed at the bottom) and the sample tube was attached to the bottom of the seed- holding tube with a Teflon connector.
- a strip of filter paper and a boiling chip were placed in an empty sample tube. Both sample tubes were immersed in a liquid nitrogen bath (3.5 liters).
- Petri Dish Bioassay The attraction of western com root worm larvae to volatile compounds other than CO 2 was previously reported by our laboratory on the basis of experiments conducted using a petri dish bioassay apparatus (Hibbard and Bjostad 1988, 1989; Bjostad and Hibbard 1992). The results we have now obtained using the Y-tube apparatus conflict with these reports, and we conducted experiments using the petri dish bioassay apparatus to re-investigate the results reported previously (Hibbard and Bjostad 1988). Three plastic petri dishes (5 cm diameter) were connected with 2-cm lengths of Teflon tubing (10 mm diameter) inserted into holes in their sides (Graph 19-6-A). Holes were cut with a brass tube attached to a soldering iron.
- the bottoms of the 2 end dishes had 12 mm holes melted through their centers.
- the apparatus was supported on a ring stand. Cryogenic collections of com seedlings were prepared as described above, except that no filter paper strip was placed in the bottom of the collection tube.
- the tube had warmed to room temperature, it was flushed for 10 sec with 100% CO 2 from a tank at 4 psi, then inverted for 30 sec.
- an empty sample tube was similarly flushed with CO 2 for 10 sec and inverted for 30 sec. Immediately after inversion for 30 sec, each tube was capped and allowed to sit for 15 min to allow the CO 2 to equilibrate.
- the petri dish apparatus was assembled and a bubble level was used to insure that the apparatus was not tilted to 1 side or the other.
- GC-MS-SIM measurements indicated that the CO 2 concentrations in the tubes were equal (measured through pinholes in the caps from within 5 cm of the top of the tubes) both tubes were connected with a Teflon connector to the holes in the bottom of the end dishes of the bioassay apparatus.
- the covers were placed on all 3 dishes and the apparatus was allowed to sit for 5 min to allow volatile compounds to begin diffusing. After 5 min, 10 2nd-instar western com root worm larvae were placed in the center of the middle
- the petri dish bioassay was analyzed similarly, except that 7 orthogonal comparisons were made, comprising the 7 bioassay intervals, for both the CO 2 data and the behavioral data. Means and standard errors are expressed as mean + standard error in the text that follows.
- Soil Bioassay The larvae chose the soil from growing com roots significantly more often (P ⁇ 0.05) (Graph 19-4- A) when the syringe on the com side contained a higher concentration of CO 2 than the control side (Graph 19-4-B). There was no significant difference between the number of larvae that chose the com headspace and larvae that chose the control when the CO 2 concentrations were the same. Larvae chose the control side more often when the control contained twice the concentration of CO 2 as the treatment side.
- Petri Dish Bioassay There was no significant difference between the number of larvae that chose the cryogenic collection of com volatiles and larvae that chose the control (P > 0.05) in the petri dish bioassay (Graph 19-6-B). During the 30 min that the bioassay was run, there was no significant difference between the CO 2 concentration on the com side and the control side inside the petri dish apparatus (Graph 19-6-C).
- One aspect of the present invention is the use of CO 2 to attract soil organisms (insects, nematodes, mites) away from their host plants or to confuse the organisms so that they are unable to locate the host plants.
- One source of CO 2 that might be used is carbonated water. When used to irrigate the soil, carbonated water has been demonstrated to enrich the soil and increase the health and production of certain crops.
- Sources of CO 2 can also be used to attract soil-dwelling organisms to pesticide granules or to pellets containing a biocontrol agent. Under field conditions, sufficient CO 2 gradients can be produced by granules of potassium bicarbonate co-formulated with an acid and a pesticide that are broadcast or incorporated into the soil.
- Organic sources can be used to achieve a slow release of CO 2 for control of soil organisms using various approaches.
- One approach is the co-encapsulation of yeast and a nutrient substrate with calcium alginate, or with k-carrageenan, which is less expensive than calcium alginate.
- Calcium alginate co-encapsulation is relatively new technique in the fermentation industry that is currently used as a means for storage and dispersal of microorganisms, and has the potential to be employed in a variety of applications.
- Starch granules can also be used as formulations for microbial pesticides, and it is possible to incorporate chemical or biological sources of CO 2 into these granules to attract and kill soil pests.
- FIG. 19-1 Figure Legends Graph 19-1.
- A Glass bead bioassay apparatus with candidate chemical cues in syringes.
- B Choice test bioassay with syringe sources containing the headspace from germinating com seedlings versus 3 different concentrations of CO 2 alone with larvae from a non-diapausing strain of western com root worm.
- C CO 2 concentrations (measured with GC-MS-SIM) of headspace over germinating com seeds and CO 2 mixtures in syringes for the choice tests with larvae from a non- diapausing strain of western com root worm.
- A Choice test bioassay with syringe sources containing the headspace from germinating com seedlings versus 3 concentrations of CO 2 alone and the glass beads on the treatment side coated with the volatiles from the com headspace.
- B CO 2 concentrations (measured with GC-MS-SIM) of the headspace over germinating com seeds and the CO 2 mixtures in syringes. Significant differences (P ⁇ 0.05) are indicated by different lower case letters. Bars represent standard errors.
- Graph 19-5 (A) Choice test bioassay with syringe sources containing the headspace from germinating com seedlings that have been fed upon by western com root worm larvae versus 3 different concentrations of CO 2 alone. (B) CO 2 concentrations (measured with GC-MS-SIM) of headspace over western com root worm-damaged com seedlings and CO 2 mixtures in the syringes. Significant differences (P ⁇ 0.05) are indicated by different lower case letters. Bars represent standard errors. Graph 19-6. (A) 3-petri-dish choice test bioassay apparatus.
- the bioassay comprises a laboratory test conducted in tubs (30 x 15 x 8 cm) filled with 3000 grams of soil. A single corn seed is placed 2 cm from one end of the tub. After the corn plant is 4 days old, 30 neonate Western Corn root worm larvae are introduced into the soil in the end of the tub opposite the corn seedings. Larvae are given 24 hours to attempt to locate and attack the corn plant. Larvae are recovered from the corn plant.
- a measured amount (0.25 gram of prepared granule is mixed into 1.0 kg. of soil (20% moisture). This soil is used to fill the center 1/3 of the bioassay tub.
- Graph 22 shows a similar trial conducted with CO 2 and feeding stimulants in combination with various rates (10 mg. and 0.3 mg/g.) of CGA-29334 (Thiamethoxam).
- Graph 23 shows a larval bioassay in soil tubs using various increasing amounts of Thiamethoxam (e.g., CGA-293343).
- Other forms of attractants in addition to the CO 2 releasing agents as disclosed herein, can also be used in combination with insecticides and CO 2 attracting agents.
- Such compounds include, for example, pheromones, generally known by those of skill in the art, which can be used in combination with the present invention and therefore forms part of the present invention.
- GRAPH 20 examples include, for example, pheromones, generally known by those of skill in the art, which can be used in combination with the present invention and therefore forms part of the present invention. GRAPH 20
- Granular formulation (1 gram) mixed into soil in center 1/3 of tub Rates of CGA-293343 are listed as (mg/g) - milligrams (A/1) per 1 gram granules
- compositions according to the invention can be prepared by a) mixing at least one pesticidally active compound, at least one signal substance, at least one liquid UV absorber which is only sparingly miscible with water and, if appropriate, additives, b) dispersing the resulting mixture in a solution of at least one surfactant and water and c) treating the resulting emulsion with an aqueous solution or dispersion of at least one curable polymer which, after drying of the formulation, has sponge-like structure or forms a porous film, and if appropriate, with additives.
- compositions according to the invention comprise one or more pesticidally active compounds. These are to be understood as meaning all customary substances which are suitable for combating harmful insects and undesirable representatives of the order Acarina.
- the following are preferably suitable, carbamates, organophosphorous compounds, nitrophenols and their derivatives, nitromethylenes, nicotinoids, formamidines, ureas, phenylbenzoylureas, pyrethroids, chlorinated hydrocarbons and Bazillus thuringensis preparations.
- A azinphos M, azocyclotin, • Bacillus thuringiensis, bendiocarb, benfuracarb, bensultap, betacyfluthrin, bifenthrin, BPMC, brofenprox, bromophos A, bufencarb, buprofezin, butocarboxin, butylpyridaben, • cadusafos, carbaryl, carbofuran, carbophenothion, carbosulfan, cartap,
- compositions according to the invention comprise can be all customary substances which have an attractant action on the pests to be combated and which alter the behaviour of these pests.
- Signal substances which are preferably suitable are pheromones, kairomones and attractants. Examples of such materials which may be mentioned are the following substances.
- compositions according to the invention comprise at least one liquid UV absorber which is only sparingly miscible with water.
- Suitable polymers which the compositions according to the invention comprise are all customary water-soluble or water-dispersible polymers or copolymers which, after application of the compositions, dry as a film to form a sponge-like, porous matrix.
- Polymers which are preferably suitable are those which can be synthesized by means of anionic or non-ionic polymerization of suitable monomers, for example by emulsion polymerization or bead polymerization with the aid of free-radical initiators or other initiator systems.
- Other polymers which can preferably be employed are those based on natural rubber latices.
- polyvinyl acetate Movable Low Density Polyethylene
- Movable Low Density Polyethylene polyvinyl acetate
- Movkol K® polyvinyl alcohols with various degrees of hydrolyzation
- Carbopol® polyvinylpyrrolidones
- Carbopol® polyvinylpyrrolidones
- acrylate-, polyol- or polyester-based varnish- system binders which are soluble or dispersible in water
- Desmophen®, Roskydal®, Bayhydrol® furthermore copolymers of two or more monomers such as acrylic acid, methacrylic acid, itaconic acid, maleic acid, fumaric acid, maleic anhydride, vinylpyrrolidone, ethylenically unsaturated monomers such as ethylene, butadiene, isoprene, chloroprene, styrene, divinylbenzene, alph, -methylstyrene or p
- the polymers When the formulations dry, the polymers form a matrix which is not or only sparingly soluble, or sparingly dispensible, in water.
- Polymers which have a low glass transition temperature are preferably employed, in particular those substances with a glass transition temperature of below 50°C.
- the compositions according to the invention comprise one or more surfactants. Suitable surface-active substances are all those which are conventionally used in the production of plant treatment products.
- non-ionic and anionic emulsifiers such as polyoxyethylene fatty acid esters, polyoxyethylene fatty alcohol ethers, alkylaryl polyglycol ethers, fatty amine ethoxylates, alkylsulphonates, alkyl sulphates, alkylarylsulphonates, aryl sulphates and silicone surfactants.
- surface-active substances are listed in McCutcheon's "Emulsifiers and Detergents" 1982 North America Edit. MC Publishing Co., 175 Rock Road, Glen rock, NJ. 07 452, USA.
- Suitable additives which may be present in the compositions according to the invention are all those which can conventionally be used as additives in plant treatment products. These include colourants, antioxidants, thickeners, filters, antifreeze agents and solvents.
- Suitable colorants are soluble dyes or sparingly soluble colour pigments, such as, for example, titanium dioxide, pigment-grade colour black or zinc oxide.
- Suitable antioxidants are all substances which can conventionally be employed for this purpose in plant treatment products. Sterically hindered phenols and alkyl-substituted hydroxyanisoles and hydroxytoluenes are preferred.
- Thickeners which are suitable are all substances which can conventionally be employed for this purpose in plant treatment products.
- the following are preferably suitable: organic polymers, such as partially or fully neutralized polyacrylic acids (Carbopol®), polyethylene glycols (Polyox®), polyvinyl alcohols and non-ionically or ionically modified celluloses (Tylose®), xanthan-based thixotropic thickeners (Kelzan®), and also inorganic disperse thickeners, such as precipitated or pyrogenic silicas, kaolins, bentonites and aluminum/silicon mixed oxides.
- organic polymers such as partially or fully neutralized polyacrylic acids (Carbopol®), polyethylene glycols (Polyox®), polyvinyl alcohols and non-ionically or ionically modified celluloses (Tylose®), xanthan-based thixotropic thickeners (Kelzan®), and also inorganic disperse thickeners, such as precipitated
- Suitable antifreeze agents are all substances which can conventionally be employed for this purpose in plant treatment products.
- Urea, glycerol or propylene glycol area all preferably suitable.
- Fillers are, again, all inert materials which can conventionally be employed for this purpose in plant treatment products.
- the following are preferably suitable: rockmeals, calcium carbonate, quartzmeal and aluminum/silicon mixed oxides or mixed hydroxides.
- Solvents which are suitable are all inert organic solvents which can conventionally be employed for this purpose in plant treatment products.
- glycols such as propylene glycol and polyethylene glycols of different molecular weight ketones, such as methyl isobutyl ketone, methyl isopropyl ketone and cyclohexanone
- amides such as dimethylformamide or diethylformamide
- N,N-dialkylated carboxamides for example Hallcomid®
- alkyl lactams such as substituted pyrrolidones (for example N-methylpyrrolidone and Surfadone®) and caprolactams (for example Azone®)
- aromatic hydrocarbons such as xylene and aromatic distillation fractions (for example Solvesso®)
- esters such as propylene glycol monomethyl ether acetate, dibutyl adipate and
- the solvents can be employed in the form of individual components or of mixtures. Particularly preferred are those which are miscible with the UV stabilizer and which are not unduly volatile.
- concentration of individual components in the compositions according to the invention can be varied within a substantial range. After deducting the water present in the compositions according to the invention, the concentrations
- • of pesticidally active compounds are generally between 0.1 and 10% by weight, preferably between 1 and 4% by weight.
- of signal substances are generally between 0.01 and 1% by weight, preferably 0.05 and 0.3% by weight.
- UV absorbers are generally between 10 and 80% by weight, preferably between 40 and 70% by weight.
- of polymers of generally between 10 and 90% by weight, preferably between 25 and 75% by weight.
- • of surface-active substances of generally between 0.1 and 4% by weight, preferably between 0.2 and 2% by weight, and of additives are generally between 0 and 50% by weight, preferably between 0 and 25% by weight.
- compositions according to the invention also comprise water.
- the water content prior to drying varies within a substantial range. It is generally between 50 and 90% by weight.
- a procedure is generally followed in which a premix is first prepared by dissolving or dispersing at least one pesticidally active compound and at least one signal substance in at least one liquid UV absorber with stirring at temperatures between 20°C and 50°C, preferably at room temperature, and, if appropriate, additives are added.
- the premix is then dispersed into a stirred solution of at least one surfactant in water at temperatures between 20°C. and 50°C, preferably at room temperature.
- the dispersing process is carried out in such a way that an oil-in-water emulsion is formed in which the mean particle size is generally between 0.5 and 50 ⁇ m.
- the resulting emulsion is subsequently treated with a solution or dispersion at least one polymer in water and, if appropriate, additives, at temperatures between 20°C. and 50°C, preferably at room temperature, with stirring.
- a different procedure may be followed when preparing the compositions according to the invention. In principle, it is possible to mix the components in any desired sequence. All stirring and mixing apparatus customary for the preparation of the compositions according to the invention is suitable.
- compositions according to the invention are obtained in a liquid or viscous state. After application, the viscous compositions dry and form a non-flowable, non-tacky coating with good adhesive properties which has a spongelike structure or forms a porous film. Once dry, the coating has a firm to rubber-like consistency.
- compositions according to the invention are highly suitable for combating harmful insects and undesirable representatives of the order Acarina, which are encountered in agriculture, in forestry and in horticulture including viticulture. They can be used against the pests mentioned below.
- Isopoda for example, Oniscus asellus, Armadillidium vulgare and Porcellio scaber.
- Diplopoda for example, Blaniulus guttulatus.
- Chilopoda for example, Geophilus carpophagus and Scutigera spec.
- Symphyla for example, Scutigerella immaculata.
- Thysanura for example, Lepisma saccharina.
- Collembola for example, Onychiurus armatus.
- Periplaneta americana Leucophaea maderae, Blattella germanica, Acheta domesticus, Gryllotalpa spp., Locusta migratoria migratorioides, Melanoplus differentialis and Schistocerca gregaria. From the order of the Dermaptera, for example, Forficula auricularia.
- Thysanoptera From the order of the Thysanoptera, for example, Hercinothrips femoralis and Thrips tabaci.
- From the order of the Homoptera for example, Aleurodes brassicae, Bemisia tabaci, Trialeurodes vaporariorum, Aphis gossypii, Brevicoryne brassicae, Cryptomyzus ribis, Aphis fabae, Doralis pomi, Eriosoma lanigerum, Hyalopterus arundinis, Macrosiphum avenae, Myzus spp., Phorodon humuli, Rhopalosiphum padi, Empoasca spp., Euscelis bilobatus, Nephotettix cincticeps, Lecanium corni, Saissetia oleae, Laodelphax striatellus, Nilaparvata lugens, Aonidiella aurantii, Aspidiotus hederae, Pseudococcus spp. and Psylla spp.
- Rhizopertha dominica Acanthoscelides obtectus, Bruchidius obtectus, Hylotrupes bajulus, Agelastica alni, Leptinotarsa decemlineata, Phaedon cochleariae, Diabrotica spp., Psylliodes chrysocephala, Epilachna varivestis, Atomaria spp., Oryzaephilus surinamensis, Anthonomus spp., Sitophilus spp., Otiorrhynchus sulcatus, Cosmopolites sordidus, Ceuthorrhynchus assimilis, Hypera postica, Dermestes spp.,
- Tannia spp. Bibio hortulanus, Oscinella frit, Phorbia spp., Pegomyia hyoscyami, Ceratitis capitata, Dacus oleae and Tipula paludosa.
- Acarina for example, Acarus siro, Argas spp., Ornithodoros spp., Dermanyssus gallinae, Eriophyes ribis, Phyllocoptruta oleivora, Boophilus spp., Rhipicephalus spp., Ambylomma spp., Hyalomma spp., Ixodes spp., Psoroptes spp., Chorioptes spp., Sarcoptes spp., Tarsonerous spp., Bryobia praetiosa,
- compositions according to the invention can be applied to the plants or areas under cultivation to be treated in the form of droplets, drop-like areas or thin defined layers by using conventional devices as they are known to those skilled in the art.
- Particularly suitable for the treatment of fruit-bearing trees or of vines is a process in which a defined mount of the formulations according to the invention is applied to the stem of the plants with the aid of dosing dispensers, pipettes or syringes, it also being possible for the application devices to be provided with a brushing device or with a surface nozzle to distribute the compositions over a substantial area to cover it entirely. It is also possible to spread the formulations according to the invention onto a solid carrier where they are allowed to harden, to cut up the resulting solid coatings and to attach the individual pieces at the sites where they are desired in each case.
- compositions according to the invention can be varied within a substantial range. They are generally on the order of magnitude which is conventionally chosen for the application of "attract-and-kill formulations".
- One new class of insecticides that is gaining significant recognition in the market place are the so-called “neonicotinoid” insecticides.
- Compounds of this class include, for example, the compounds imidacloprid, acetamiprid, and thiamethoxam that are described, for example, in U.S. patent nos. 4742060 and 5304566 and EP580553A2, respectively.
- A is 2-chloropyrid-5-yl, 2-methylpyrid-5-yl, l-oxido-3-pyridinio, 2-chloro-l-oxido- 5-pyridinio, 2,3-dichloro-l-oxido-5-pyridinio, tetrahydrofuran-3-yl, 5-methyl- tetrahydrofuran-3-yl or 2-chlorothiazol-5-yl group, R is hydrogen, C C 6 alkyl, phenyl-C,-C 4 alkyl, C 3 -C 6 cycloalkyl, C 2 -C 6 alkenyl or
- the compounds (I) may be in the form of tautomers. Accordingly, hereinbefore and hereinafter, where appropriate the compound compounds (I) are to be understood to include corresponding tautomers, even if the latter are not specifically mentioned in each case.
- Preferred compounds of the formula (I) are those wherein A is a pyrid-3-yl,
- -CH 2 -S-CH 2 - -CH 2 -NH-CH 2 -, -CH 2 -N(CH 3 )-CH 2 -, especially group -CH 2 -CH 2 - or -CH 2 -O-CH 2 -, particularly -CH 2 -O-CH 2 -; and X is N-NO 2 or N-CN, more especially N-NO 2 .
- an at least quaternary pesticidal composition comprising an insecticidally effective amount of a compound selected from the group consisting of: a compound of the formula
- the compounds (I) are capable of forming acid addition salts.
- Those salts are formed, for example, with strong inorganic acids, such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, nitrous acid, a phosphoric acid or a hydrohalic acid, with strong organic carboxylic acids, such as unsubstituted or substituted, for example halo-substituted, C,-C 4 alkanecarboxylic acids, for example acetic acid, saturated or unsaturated dicarboxylic acids, for example oxalic, malonic, succinic, maleic, fumaric or phthalic acid, hydroxycarboxylic acids, for example ascorbic, lactic, malic, tartaric or citric acid, or benzoic acid, or with organic sulfonic acids, such as unsubstituted or substituted, for example halo-substituted, C,-C 4 alkane- or aryl-sulfonic
- salts with bases are, for example, metal salts, such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower alkylamine, for example ethyl-, diethyl-, triethyl- or dimethyl-propyl-amine, or a mono-, di- or tri-hydroxy-lower alkylamine, for example mono-, di- or tri-ethanolamine.
- corresponding internal salts may also be formed.
- any reference hereinbefore or hereinafter to the free compounds of formula (I) or to their respective salts is to be understood as including also the corresponding salts or the free compounds of formula (I), where appropriate and expedient.
- the free form is generally preferred in each case.
- an attractant feeding stimulant for corn root worm with particular insecticides.
- an attractant feeding stimulant for corn room worm with a neonicotinoid class of insecticides, and even more preferably, thiamethoxam.
- the inventors are the first to appreciate that while commonly used corn root worm insecticides, (such as organophosphates/carbonates) repel the neonate corn root worm larvae, neonicotinoid compounds do not repel such larvae.
- the present invention provides for the attraction of corn root worms used in conjunction with a technology that terminates or kills such root worms and root worm larvaes, thus providing a fully functional root worm control method and compound whether applied as a seed treatment or as a granule which is applied to the soil at planting.
- other insecticide compounds that do not repel boring insects, such as the corn root worm, will also find application and are within the scope of the invention.
- formulations that can be utilized in conjunction with the present invention are those described in U.S. Patent No. 5,707,638, incorporated in its entirety herein by this reference.
- an insecticidal attract-and-kill formulation comprising the following is useful in combination with the CO 2 attractants mentioned herein: • 0.1 to 10% by weight of an insecticidally active compound selected from cyfluthrin, ⁇ -cyfluthrin and transfluthrin,
- a UV absorber selected from a benzotriazole, and a 2-hydroxy-4-methoxy-benzophenone and 2-(2-ethyl-hexyl)-2-cyano
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU52719/00A AU5271900A (en) | 1999-11-04 | 2000-05-17 | Method and device for attracting insects |
| JP2001534229A JP2003526341A (en) | 1999-11-04 | 2000-05-17 | Apparatus and method for attracting insects |
| BR0015510A BR0015510A (en) | 1999-11-04 | 2000-05-17 | Method and device for attracting insects |
| EP20000937570 EP1229786A1 (en) | 1999-11-04 | 2000-05-17 | Method and device for attracting insects |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| USPCT/US99/26074 | 1999-11-04 | ||
| PCT/US1999/026074 WO2000027187A2 (en) | 1998-11-06 | 1999-11-04 | Method and device for attracting insects |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2001032013A1 true WO2001032013A1 (en) | 2001-05-10 |
Family
ID=22273989
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2000/013477 WO2001032013A1 (en) | 1999-11-04 | 2000-05-17 | Method and device for attracting insects |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP1229786A1 (en) |
| JP (1) | JP2003526341A (en) |
| AU (1) | AU5271900A (en) |
| BR (1) | BR0015510A (en) |
| WO (1) | WO2001032013A1 (en) |
| ZA (1) | ZA200203664B (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7294343B2 (en) * | 2001-06-15 | 2007-11-13 | Grain Processing Corporation | Biodegradable sorbents |
| US7347994B2 (en) | 2002-09-13 | 2008-03-25 | Ica Trinova, Llc | Method and composition for attracting arthropods by volatilizing an acid |
| WO2010020477A2 (en) * | 2008-08-21 | 2010-02-25 | Syngenta Participations Ag | Crop safeners |
| CN103461297A (en) * | 2013-09-21 | 2013-12-25 | 汪盛明 | Preparation method of overlaid termite trapping device |
| WO2014041112A1 (en) | 2012-09-14 | 2014-03-20 | BIOCARE Gesellschaft für biologische Schutzmittel mbH | Attraction systems for pests and use thereof |
| DE102014103268A1 (en) | 2014-03-12 | 2015-09-17 | BIOCARE Gesellschaft für biologische Schutzmittel mbH | Locking system for flying insects and arachnids |
| WO2017060349A1 (en) * | 2015-10-07 | 2017-04-13 | Basf Se | Hollow wax capsules with reduced carbon dioxide release for attracting soil-dwelling pests |
| CN107410387A (en) * | 2017-07-27 | 2017-12-01 | 毕晓峰 | A kind of Chinese herbal medicine type termite-killing trapping gluing bait formulation, glued bait formulation plate and preparation method thereof |
| US9884875B2 (en) | 2010-08-10 | 2018-02-06 | Takeda Pharmaceutical Company Limited | Heterocyclic compound and use thereof |
| US11700846B2 (en) | 2016-07-12 | 2023-07-18 | Jeremy Eli Hirsch | Insecticide dispensing device and method |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20220008390A (en) * | 2017-04-14 | 2022-01-20 | 글렌 밥콕 | Carbon dioxide underwater delivery device and supplementation system |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US1599408A (en) * | 1925-05-04 | 1926-09-14 | Albert D Cardinet | Insect trap |
| US4506473A (en) * | 1983-11-14 | 1985-03-26 | John G. Mills, II | Carbon dioxide generator insect attractant |
| US5112843A (en) * | 1990-12-13 | 1992-05-12 | Colorado State University Research Foundation | Arthropodicidal use of 6-methoxy-2-benzoxazolinone to attract and control corn rootworm (Diabrotica) |
| US5189830A (en) * | 1991-08-26 | 1993-03-02 | Janette Zuckerman | Tick trap |
| US5329726A (en) * | 1992-09-08 | 1994-07-19 | Thorne Barbara L | System for termite detection and control |
| US5338551A (en) | 1992-07-02 | 1994-08-16 | Lajoie M Stephen | Polyfunctional agrochemical bicarbonate-containing compositions |
| US5342630A (en) | 1992-07-01 | 1994-08-30 | Church & Dwight Co., Inc. | Environmentally safe pesticide compositions |
| US5389386A (en) | 1994-06-30 | 1995-02-14 | Church & Dwight Co., Inc. | Method of controlling fungal disease in cultivated plants |
| US5415877A (en) | 1993-04-23 | 1995-05-16 | Church & Dwight Co., Inc. | Bicarbonate fungicide product with a combination of surfactant ingredients |
| US5424270A (en) | 1992-12-02 | 1995-06-13 | Church & Dwight Co., Inc. | Bicarbonate fungicide compositions containing spreader-sticker ingredients |
| US5425952A (en) | 1993-10-13 | 1995-06-20 | Church & Dwight Co., Inc. | Slow-release ammonium bicarbonate fungicide compositions |
| US5432146A (en) | 1992-12-02 | 1995-07-11 | Church & Dwight Co., Inc. | Free-flowing bicarbonate fungicide compositions |
| US5607684A (en) * | 1991-10-31 | 1997-03-04 | Micro Flo Company | Manufacture of bait with hot melt binder |
-
2000
- 2000-05-17 EP EP20000937570 patent/EP1229786A1/en not_active Withdrawn
- 2000-05-17 BR BR0015510A patent/BR0015510A/en not_active IP Right Cessation
- 2000-05-17 WO PCT/US2000/013477 patent/WO2001032013A1/en not_active Application Discontinuation
- 2000-05-17 JP JP2001534229A patent/JP2003526341A/en active Pending
- 2000-05-17 AU AU52719/00A patent/AU5271900A/en not_active Abandoned
-
2002
- 2002-05-08 ZA ZA200203664A patent/ZA200203664B/en unknown
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US1599408A (en) * | 1925-05-04 | 1926-09-14 | Albert D Cardinet | Insect trap |
| US4506473A (en) * | 1983-11-14 | 1985-03-26 | John G. Mills, II | Carbon dioxide generator insect attractant |
| US5112843A (en) * | 1990-12-13 | 1992-05-12 | Colorado State University Research Foundation | Arthropodicidal use of 6-methoxy-2-benzoxazolinone to attract and control corn rootworm (Diabrotica) |
| US5189830A (en) * | 1991-08-26 | 1993-03-02 | Janette Zuckerman | Tick trap |
| US5607684A (en) * | 1991-10-31 | 1997-03-04 | Micro Flo Company | Manufacture of bait with hot melt binder |
| US5342630A (en) | 1992-07-01 | 1994-08-30 | Church & Dwight Co., Inc. | Environmentally safe pesticide compositions |
| US5338551A (en) | 1992-07-02 | 1994-08-16 | Lajoie M Stephen | Polyfunctional agrochemical bicarbonate-containing compositions |
| US5346704A (en) | 1992-07-02 | 1994-09-13 | Church & Dwight Co., Inc. | Polyfunctional agrochemical bicarbonate-containing compositions |
| US5329726A (en) * | 1992-09-08 | 1994-07-19 | Thorne Barbara L | System for termite detection and control |
| US5424270A (en) | 1992-12-02 | 1995-06-13 | Church & Dwight Co., Inc. | Bicarbonate fungicide compositions containing spreader-sticker ingredients |
| US5432146A (en) | 1992-12-02 | 1995-07-11 | Church & Dwight Co., Inc. | Free-flowing bicarbonate fungicide compositions |
| US5415877A (en) | 1993-04-23 | 1995-05-16 | Church & Dwight Co., Inc. | Bicarbonate fungicide product with a combination of surfactant ingredients |
| US5425952A (en) | 1993-10-13 | 1995-06-20 | Church & Dwight Co., Inc. | Slow-release ammonium bicarbonate fungicide compositions |
| US5389386A (en) | 1994-06-30 | 1995-02-14 | Church & Dwight Co., Inc. | Method of controlling fungal disease in cultivated plants |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7294343B2 (en) * | 2001-06-15 | 2007-11-13 | Grain Processing Corporation | Biodegradable sorbents |
| US7922992B2 (en) | 2002-09-13 | 2011-04-12 | Ica Trinova, Llc | Composition and method for producing carbon dioxide |
| US7347994B2 (en) | 2002-09-13 | 2008-03-25 | Ica Trinova, Llc | Method and composition for attracting arthropods by volatilizing an acid |
| US8709396B2 (en) | 2002-09-13 | 2014-04-29 | Premark Feg L.L.C. | Method and composition for attracting arthropods by volatizing an acid |
| CN102131391A (en) * | 2008-08-21 | 2011-07-20 | 先正达参股股份有限公司 | Crop safeners |
| WO2010020477A3 (en) * | 2008-08-21 | 2010-09-16 | Syngenta Participations Ag | Crop safeners |
| EA018318B1 (en) * | 2008-08-21 | 2013-07-30 | Зингента Партисипейшнс Аг | Antidotes for agricultural crops |
| WO2010020477A2 (en) * | 2008-08-21 | 2010-02-25 | Syngenta Participations Ag | Crop safeners |
| US11279713B2 (en) | 2010-08-10 | 2022-03-22 | Takeda Pharmaceutical Company Limited | Heterocyclic compound and use thereof |
| US10654874B2 (en) | 2010-08-10 | 2020-05-19 | Takeda Pharmaceutical Company Limited | Heterocyclic compound and use thereof |
| US9884875B2 (en) | 2010-08-10 | 2018-02-06 | Takeda Pharmaceutical Company Limited | Heterocyclic compound and use thereof |
| US20170079258A1 (en) * | 2012-09-14 | 2017-03-23 | BIOCARE Gesellschaft für biologische Schutzmittel mbH | Attraction Systems for Pests and Use Thereof |
| DE102012108621A1 (en) | 2012-09-14 | 2014-03-20 | BIOCARE Gesellschaft für biologische Schutzmittel mbH | Locking systems for pests and their use |
| WO2014041112A1 (en) | 2012-09-14 | 2014-03-20 | BIOCARE Gesellschaft für biologische Schutzmittel mbH | Attraction systems for pests and use thereof |
| US11553703B2 (en) | 2012-09-14 | 2023-01-17 | Biocare Gesellschaft Fuer Biologische Schutzmittel Mbh | Attraction systems for pests and use thereof |
| CN103461297A (en) * | 2013-09-21 | 2013-12-25 | 汪盛明 | Preparation method of overlaid termite trapping device |
| WO2015136023A1 (en) * | 2014-03-12 | 2015-09-17 | Biocare Gesellschaft Fuer Biologische Schutzmittel Mbh | System for attracting flying insects and arachnids |
| DE102014103268A1 (en) | 2014-03-12 | 2015-09-17 | BIOCARE Gesellschaft für biologische Schutzmittel mbH | Locking system for flying insects and arachnids |
| AU2015228866B2 (en) * | 2014-03-12 | 2019-02-07 | Biocare Gesellschaft Fur Biologische Schutzmittel Mbh | System for attracting flying insects and arachnids |
| WO2017060349A1 (en) * | 2015-10-07 | 2017-04-13 | Basf Se | Hollow wax capsules with reduced carbon dioxide release for attracting soil-dwelling pests |
| US11700846B2 (en) | 2016-07-12 | 2023-07-18 | Jeremy Eli Hirsch | Insecticide dispensing device and method |
| CN107410387A (en) * | 2017-07-27 | 2017-12-01 | 毕晓峰 | A kind of Chinese herbal medicine type termite-killing trapping gluing bait formulation, glued bait formulation plate and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| BR0015510A (en) | 2004-02-25 |
| JP2003526341A (en) | 2003-09-09 |
| AU5271900A (en) | 2001-05-14 |
| EP1229786A1 (en) | 2002-08-14 |
| ZA200203664B (en) | 2003-08-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20050063956A1 (en) | Method and device for attracting insects | |
| US5707638A (en) | Insecticidal attract-and-kill formulations | |
| US6528049B2 (en) | Bisexual attractants, aggregants and arrestants for adults and larvae of codling moth and other species of lepidoptera | |
| US6395776B1 (en) | Pesticides | |
| US6978572B1 (en) | Method and device for attracting insects | |
| JPH11511152A (en) | Combinations of active compounds including pyrethroids and insect development inhibitors | |
| TW200932114A (en) | Pesticidal compound mixtures | |
| JP4846098B2 (en) | Use of spinosyns as soil killing insecticides | |
| WO2001032013A1 (en) | Method and device for attracting insects | |
| WO1996017515A1 (en) | An ant bait attractive to multiple species of ants | |
| US20230122932A1 (en) | New sex pheromone components for the fall armyworm, spodoptera frugiperda | |
| JPH11511151A (en) | Insecticidal composition | |
| US20070071783A1 (en) | Pheromone attractants for the green mirid | |
| US5798112A (en) | Phototoxic insecticidal composition and method for controlling insect populations | |
| CN112998024A (en) | Insecticidal composition containing fenoxycarb and application thereof | |
| MXPA98000914A (en) | Agents pestici | |
| KR20000016982A (en) | Composition for extermination of harmful arthropods |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ DE DK DM EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| ENP | Entry into the national phase |
Ref country code: JP Ref document number: 2001 534229 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002/03664 Country of ref document: ZA Ref document number: 200203664 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 52719/00 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000937570 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000937570 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2000937570 Country of ref document: EP |