WO2002070589A2 - Preparation of polymer nanocomposites by dispersion destabilization - Google Patents
Preparation of polymer nanocomposites by dispersion destabilization Download PDFInfo
- Publication number
- WO2002070589A2 WO2002070589A2 PCT/US2002/006055 US0206055W WO02070589A2 WO 2002070589 A2 WO2002070589 A2 WO 2002070589A2 US 0206055 W US0206055 W US 0206055W WO 02070589 A2 WO02070589 A2 WO 02070589A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polymer
- clay
- dispersion
- clay mineral
- polymer dispersion
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/346—Clay
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y30/00—Nanotechnology for materials or surface science, e.g. nanocomposites
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/20—Compounding polymers with additives, e.g. colouring
- C08J3/205—Compounding polymers with additives, e.g. colouring in the presence of a continuous liquid phase
- C08J3/21—Compounding polymers with additives, e.g. colouring in the presence of a continuous liquid phase the polymer being premixed with a liquid phase
- C08J3/215—Compounding polymers with additives, e.g. colouring in the presence of a continuous liquid phase the polymer being premixed with a liquid phase at least one additive being also premixed with a liquid phase
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K2201/00—Specific properties of additives
- C08K2201/011—Nanostructured additives
Definitions
- the present invention generally relates to a polymer nanocomposite. More particularly, the invention relates to a polymer nanocomposite formed from a mixture of a clay dispersion with a polymer dispersion.
- Clay minerals such as montmorillonite are composed of silicate layers with a thickness of about 1 nanometer. Incorporation of such layered materials in polymers result in products which may frequently be referred to as nanocomposites. Incorporating clay minerals in a polymer matrix, however, may not always be a straightforward process. Incorporating clay minerals may require additional manufacturing steps or additional capital equipment costs, especially if melt processing may be required. Some have tried to improve the process of forming nanocomposites by increasing the compatibility between the clay minerals and the organic polymers.
- lipophilic compounds such as oniurn compounds
- a dispersion is produced having a layered silicate, a monomer, and a polymerization initiator.
- the monomer is polymerized to form the latex.
- This polymerization process results in a latex containing a layered material intercalated with a polymer.
- This method is disclosed in U.S. Patent No. 5,883,173 to Elspass et al., which is incorporated herein by reference.
- the approaches to preparing nanocomposites may prove difficult in controlling exfoliation and polymer molecular weight. Efficiency in emulsion polymerization may also be difficult to achieve.
- a clay mineral such as, but not limited to smectite clay minerals
- a clay mineral may be intercalated with a polymer by mixing a dispersion of a polymer in a liquid carrier and a dispersion of a clay mineral in a liquid carrier to form a dispersion mixture.
- the dispersion mixture may be treated with a flocculating agent.
- a dispersion of polymer in a liquid carrier may be prepared by any of the means available to those skilled in the art.
- the polymeric dispersion may be formulated by mixing a combination of a liquid carrier, a surfactant, and a polymer.
- a clay mineral dispersion may be produced by mixing a clay mineral with a liquid carrier such that the clay mineral is dispersed in the liquid carrier.
- a surfactant may be added when preparing a dispersion of a clay mineral in the liquid carrier.
- the two dispersions may be subsequently mixed together to produce a dispersion mixture of the polymer and the clay mineral.
- the dispersion mixture may then be flocculated by addition of a flocculating agent.
- flocculating agents may be, but are not limited to, inorganic salts, double-layered metal hydroxides, quaternary onium compounds, or an onium saturated clay mineral.
- An onium saturated clay mineral may be defined as a clay mineral that has been treated with a quaternary onium compound added in excess of that required to meet the Cation Exchange Capacity (CEC) of the clay mineral.
- CEC Cation Exchange Capacity
- a non-layered clay mineral may be substituted in the aforementioned compositions in place of a layered clay mineral.
- the flocculated nanocomposite material may be separated from the liquid carrier using techniques such as, but not limited to, filtration, centrifugation, or evaporation.
- the nanocomposite may be formulated, compounded, and processed for use in applications such as, but not limited to, plastic engineered parts, film, and fiber as well as rubber articles such as tires, belts, and hoses.
- Flocculation as defined herein , is the aggregation of colloidal particles suspended in water.
- Intercalation as defined herein is the movement of polymer between smectite layers, where the layers are separated, but the ordered relationship between the layers is maintained.
- the interlayer spacing can be measured by X-ray diffraction.
- Exfoliation as defined herein is the movement of polymer between the smectite layers, where the layers are separated and the ordered relationship between the layers is lost. In completely exfoliated examples, no X-ray diffraction results from the interlayer separations.
- Nanocomposite as defined herein is a composition comprising layered inorganic particles in a polymer matrix.
- a polymer dispersion may be prepared by dispersion of a polymer within a liquid carrier.
- the polymer dispersion may be prepared by adding an amount of polymer, up to about 80% by weight of polymer, to the liquid carrier.
- the liquid carrier may be either water, an organic solvent, or mixtures thereof.
- Polymers that may be used include, but are not limited to, the following examples: polyester, polyurethane, polyvinyl chloride, styrene-butadiene, acrylic rubber, chlorosulfonated polyethylene rubber, fluoroelastor ⁇ er, polyisoprene, polycarbonate resin, polyamide resin, polyolefin resin, thermoplastic resin or mixtures thereof.
- the polymer dispersion may be subjecting to a shearing process to fully disperse the polymeric material within the liquid carrier.
- a clay mineral dispersion may be prepared by adding from about 1% to about 10% by weight of a clay mineral to a liquid carrier.
- the liquid carrier may be either water, an organic solvent, or mixtures thereof.
- the clay mineral used may be naturally occurring or synthetic. Positively charged or negatively charged minerals may be used. Representative examples of negatively charged clay minerals useful in accordance with an embodiment may be as follows:
- R is selected from the group consisting of Na + , Li + , NH + , and mixtures thereof; Hectorite
- R is selected from the group consisting of Na + , Li + , NH 4 + , and mixtures thereof;
- Inorganic salts include, but are not limited to, any suitable Group I or Group II main group metal cation or any suitable transition metal cation that provides sufficient ionic charge to flocculate the dispersions. Any anion that provides sufficient solubility of the inorganic compound in the liquid carrier may be used. Examples of anions include, but are not limited to, chloride, bromide, iodide, sulfate, nitrate, perchlorate, chlorate, or phosphate. Examples of inorganic salts include, but are not limited to, calcium chloride, magnesium chloride, sodium chloride, potassium chloride, or lithium chloride. Mineral compounds include, but are not limited to, hydrotalcite.
- amine salts that may be used as a flocculant may include, but are not limited to, compounds having the following structure:
- R b R 2 , and R 3 may be independently hydrogen, alkyl, aryl, or alkylaryl groups.
- the alkyl, aryl, or alkylaryl groups may include carbon moieties of about 1 to about 20 carbon atoms and X may be chloride, bromide, iodide, nitrite, nitrate, hydroxide, sulfate, sulfite, phosphate or other suitable anionic substituents.
- the flocculating agent e.g., the onium compound
- the flocculating agent is thus added prior to forming the clay- polymer dispersion, rather than after.
- Mineral compounds may also be used as a flocculating agent.
- a polymeric dispersion and a clay mineral dispersion may be prepared as previously described.
- the clay mineral dispersion may be added to the polymeric dispersion without causing flocculation.
- the polymeric dispersion/clay mineral dispersion mixture may then be contacted with about 1 % to about 10% by weight of a mineral compound (e.g., hydrotalcite) to cause flocculation of the polymeric dispersion.
- a mineral compound e.g., hydrotalcite
- the clay mineral dispersion is added to the polymer dispersion to form a clay-polymer dispersion.
- the polymer dispersion may be added to the clay dispersion to form the clay-polymer dispersion.
- a polymer dispersion and a clay mineral dispersion may be prepared separately as described earlier. These dispersions may be mixed with sufficient shear to produce a clay-polymer dispersion as described earlier.
- To the clay-polymer dispersion, with mixing, may be added an inorganic salt at about 1% to about 20%) by weight of solution such that the clay-polymer dispersion may be flocculated.
- the produced nanocomposite may be processed as described in the previous sections.
- non-layered clay minerals may be utilized as well.
- non-layered clay minerals include, but are not limited to, sepiolite or attapulgite.
- X-ray diffraction may be used to determine the extent of exfoliation, or separation, of the mineral layers in the nanocomposite.
- the D 0 o ⁇ peak may be monitored and distance of the spacing between the platelets may be inferred.
- X-ray diffraction data may be used to determine intercalation and disorder of clay mineral particles incorporated within the polymer. Platelet spacing values may range from 1-2 A in an untreated clay mineral. In clays where the platelets are so well separated, such as in a nanocomposite, the D 0 o ⁇ peak may be absent in the x-ray scan.
- a surfactant may be added to the polymer dispersion to aid in dispersion of the polymer.
- Surfactants that may be used include amphoteric, anionic, cationic, and non-ionic.
- a surfactant may be added in an amount from about 1% to about 20% by weight of polymer.
- the nanocomposites herein described may be mixed with other materials to produce a number of different products or articles.
- the nanocomposite may be formulated, for example, into automobile tires.
- the nanocomposite may be added to impart improved performance of the automobile tire on ice by n ⁇ iimizing reinforcing performance of a tread rubber, while still improving the traction force by the elimination of hydroplaning and increasing the area of contact with a road surface.
- Paints may also be formulated with the nanocomposites described herein to improve desirable paint characteristics such as minimized sagging, luster, durability, thixotropy, and solids suspension.
- the nanocomposites described herein may be used in those specialty paint formulations especially designed to paint the polymeric • materials of automobile bumpers and the like.
- U.S. Patent Number 6,133,374 to Nam, et al. which is incorporated herein by reference, further describes the use of nanocomposites in paint formulations.
- the nanocomposites described herein may, for example, be used in melt extrusion of the nanocomposite into film.
- Formulation may be accomplished by feeding solid polymer to an extruder in which the polymer may be heated to a temperature above its melting point.
- a rotating screw pushes the polymer and the resulting viscous polymer melt through the extruder barrel into a die in which the polymer may be shaped to the desired form.
- the resulting extrudate may either be quenched or allowed to cool slowly to temperatures below the melting point, causing the extrudate to rigidity and assume the shape of the die orifice.
- a gapped coat hanger die may be used to lay a melt of modified polymerized organic system onto a roller.
- the film may then be fed through a nip roller and onto a take-up roll.
- Film may also be produced as a blown film by feeding the melt of the nanocomposites through an annular die, blowing air into the inside of the bubble, then collapsing the bubble and collecting on a roll-up spool.
- the film may be either a monolayer or multiple layer material.
- the nanocomposites may also improve the extrusion of the fibers similar to the elimination of melt fractures in commercial films.
- Injection molding processes may exhibit improvements in form release and more accurate replication of the molded product to the form.
- Blow molding processes may exhibit improved surface structure features.
- the following examples serve to illustrate methods of producing a nanocomposite by the previous embodiments. The examples should not be considered limiting.
- Example 2 was conducted as Example 1 except that Good-Rite SB-1168 (a latex polymer available from B.F. Goodrich) was utilized. The dispersion mixture flocculated. The D 00 ⁇ peak was present and indicated smectite clay platelet spacing of about 36 A.
- Good-Rite SB-1168 a latex polymer available from B.F. Goodrich
- Example 3 was conducted as Example 1 except that Good-Rite SB-1177 (a latex polymer available from B.F. Goodrich) was utilized. The dispersion mixture flocculated. The D 0 o ⁇ peak was present and indicated smectite clay platelet spacing of about 38 A.
- Good-Rite SB-1177 a latex polymer available from B.F. Goodrich
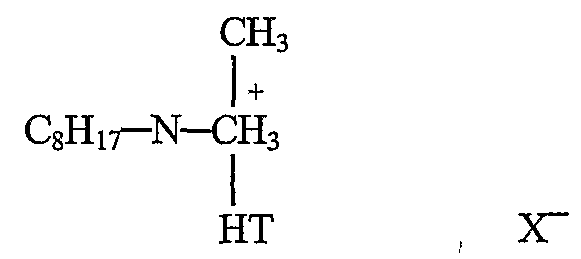
- EXAMPLE 4 A filtercake of an organoclay was prepared in the following way. A montmorillonite clay was exchanged with hydrogenated tallow alkyl(2-etl ⁇ ylhexyl)dimethyl ammonium metliylsulfate, (Arquad ® HTL8-MS, Akzo
- MER is a measure of milliequivalent ratio (MER), providing the relationship between the amount of a quaternary onium compound added to a clay based upon the cation exchange capacity of the clay.
- the slurry was filtered and the filtercake was processed by milling before use. The solids content of the filtercake was 47.40%.
- To 100 grams of Good-Rite SB-1168 was added 12.66 grams of the processed filtercake.
- a high speed disperser was employed for dispersion of the filtercake into Good-Rite SB-1168.
- the dispersion mixture flocculated.
- the solids were isolated and an x-ray diffraction analysis was conducted on the resulting nanocomposite. The D 0 o ⁇ peak was present and indicated smectite clay platelet spacing of about 28 A.
- EXAMPLE 5 A mixture of 97 g of deionized water and 3 grams of hydrotalcite were subj ected to a high energy disperser.
- the dispersion mixture flocculated.
- the solids were prepared for x-ray diffraction analysis.
- the Dooi peak indicated spacing of about 39.9 A.
- EXAMPLE 7 130 grams of a 3% aqueous montmorillonite slurry was added with stirring to 100 grams of Goodyear LPF-
- a 3% solids aqueous Cloisite ® dispersion was prepared with an excess of hydrogenated tallow alkyl(2- ethylhexyl)dimefhyl ammonium methylsulfate, (Arquad ® HTL8-MS, Akzo Chemical) to produce a 125 MER organoclay.
- the organoclay was passed through a Manton-Gaulin homogenizer at a setting of about 4,500 psig.
- To 100 grams of Goodyear LPF-6758 was added, with stirring, 180.7 grams of the aqueous Cloisite ® dispersion.
- the dispersion mixture flocculated.
- the solids were prepared for x-ray diffraction analysis.
- the D 0 o ⁇ peak indicated spacing of the clay platelets of about 39.2 A.
- EXAMPLE 9 A 3.06%) by weight aqueous organoclay slurry was made and passed through the Manton-Gaulin homogenizer at a setting of about 4,500 psig.
- the organoclay was a 125 MER organoclay with hydrogenated tallow alkyl(2-ethylhexyl)dimethyl ammonium methylsulfate, (Arquad ® HTL8-MS, Akzo Chemical).
- LPF-6758 was added, slowly and with stirring, 181.2 grams of the organoclay dispersion. A flock formed after 3 minutes of shearing.
- the solids produced were prepared for x-ray diffraction analysis.
- the D 0 o ⁇ peak indicated spacing of the clay platelets of about 37.1 A.
- EXAMPLE 10 About 50 grams of the nanocomposite produced in Example 9 was fed into a Brabender mixer over a period of two minutes and mixed for an additional 5 minutes at 150° C and at 60 rpm. The Brabender torque was measured during the mixing. The torque initially increased to about 36.1 Nm, and remained at that level during the 5 minute mixing. The material was removed and hot pressed for 5 minutes at 150° C. An x-ray diffraction analysis of the material was run. The D 0 o ⁇ peak was absent, indicating high exfoliation of the clay in the polymer.
- Example 12 is a control for comparison purposes. It is an aqueous dispersion 125 MER hydrogenated tallow alkyl(2-ethylhexyl)dimetl ⁇ yl ammonium methylsulfate, (Arquad ® HTL8-MS, Akzo Chemical) treated montmorillonite clay. An x-ray diffraction analysis of the material was run. The Dooi peak indicated spacing of the clay platelets of about 25.6 A.
- Example 13 is a control for comparison purposes. It is a dry dispersion 125 MER of hydrogenated tallow alkyl(2-ethylhexyl)dimethyl ammonium methylsulfate, (Arquad ® HTL8-MS, Akzo Chemical) treated montmorillonite clay. An x-ray diffraction analysis of the material was run. The D 0 o ⁇ peak indicated spacing of the clay platelets of about 23.7 A.
- Example 14 is a dried hydrotalcite. An x-ray diffraction analysis of the material was run. The D 00 ⁇ peak indicated spacing of the hydrotalcite platelets of about 8.6 A.
- MMT montmorillonite
- HTL8 hydrogenated tallow alkyl(2-ethylhexyl)dimetl ⁇ yl ammonium methylsulfate, (Arquad ® HTL8-MS,
- Example 5 the latex was added to the mineral. In all other examples, the mineral was added to the latex.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE60211129T DE60211129T2 (en) | 2001-03-02 | 2002-02-28 | PREPARATION OF POLYMERNANO FIBER MATERIALS BY DISPERSION DETESTILIZATION |
| CA002439632A CA2439632A1 (en) | 2001-03-02 | 2002-02-28 | Preparation of polymer nanocomposites by dispersion destabilization |
| EP02731107A EP1366109B1 (en) | 2001-03-02 | 2002-02-28 | Preparation of polymer nanocomposites by dispersion destabilization |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US27327101P | 2001-03-02 | 2001-03-02 | |
| US60/273,271 | 2001-03-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2002070589A2 true WO2002070589A2 (en) | 2002-09-12 |
| WO2002070589A3 WO2002070589A3 (en) | 2003-05-01 |
Family
ID=23043243
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2002/006055 WO2002070589A2 (en) | 2001-03-02 | 2002-02-28 | Preparation of polymer nanocomposites by dispersion destabilization |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US6849680B2 (en) |
| EP (1) | EP1366109B1 (en) |
| AT (1) | ATE325155T1 (en) |
| CA (1) | CA2439632A1 (en) |
| DE (1) | DE60211129T2 (en) |
| WO (1) | WO2002070589A2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1471108A1 (en) * | 2003-04-25 | 2004-10-27 | Rohm And Haas Company | Composite compositions including polymeric nanoparticles and clay nanoparticles |
| WO2006085957A3 (en) * | 2004-07-06 | 2007-03-22 | Exxonmobil Chem Patents Inc | Polymeric nanocomposites and processes for making the same |
| EP3500770B1 (en) | 2016-08-17 | 2022-10-05 | ContiTech Luftfedersysteme GmbH | Article, particularly an air spring bellows, a metal-rubber element, or a vibration damper |
| EP3500771B1 (en) | 2016-08-17 | 2022-11-02 | ContiTech Elastomer-Beschichtungen GmbH | Rubber mixture and elastomeric article having flame-retardant properties |
Families Citing this family (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7084197B2 (en) * | 2001-06-29 | 2006-08-01 | Ciba Specialty Chemicals Corporation | Synergistic combinations of nano-scaled fillers and hindered amine light stabilizers |
| US20050245665A1 (en) * | 2001-08-17 | 2005-11-03 | Chenggang Chen | Method of forming nanocomposite materials |
| US6680016B2 (en) * | 2001-08-17 | 2004-01-20 | University Of Dayton | Method of forming conductive polymeric nanocomposite materials |
| US20050127329A1 (en) * | 2001-08-17 | 2005-06-16 | Chyi-Shan Wang | Method of forming nanocomposite materials |
| US20050272847A1 (en) * | 2001-08-17 | 2005-12-08 | Chyi-Shan Wang | Method of forming nanocomposite materials |
| US20060079623A1 (en) * | 2001-08-17 | 2006-04-13 | Chenggang Chen | Method of forming nanocomposite materials |
| JP4733978B2 (en) * | 2002-07-05 | 2011-07-27 | エクソンモービル・ケミカル・パテンツ・インク | Functionalized elastomeric nanocomposites |
| US8399551B2 (en) * | 2002-07-05 | 2013-03-19 | Exxonmobil Chemical Patents Inc. | Functionalized elastomer nanocomposite |
| AU2003301053A1 (en) * | 2002-12-18 | 2004-07-22 | Bridgestone Corporation | Method for clay exfoliation, compositions therefore, and modified rubber contaiing same |
| EP1581587B1 (en) * | 2003-01-08 | 2007-04-11 | Süd-Chemie Ag | Master batches based on pre-exfoliated nanoclays and the use of the same |
| CN1239587C (en) * | 2003-04-03 | 2006-02-01 | 中国石油化工股份有限公司 | Composite powder, its preparation method and application |
| JP2006527294A (en) | 2003-06-12 | 2006-11-30 | ジュート−ヒェミー アクチェンゲゼルシャフト | Process for producing nanocomposite additives with modified delamination in polymers |
| JP4139792B2 (en) * | 2003-09-12 | 2008-08-27 | ニッポン・ペイント(ユーエスエイ),インコーポレーテッド | Nanoclay-modified aqueous composition for coating plastics and method for producing the same |
| US7342065B2 (en) * | 2003-09-18 | 2008-03-11 | The Goodyear Tire & Rubber Company | Preparation of nanocomposite of elastomer and exfoliated clay platelets, rubber compositions comprised of said nanocomposite and articles of manufacture, including tires |
| CA2443059A1 (en) * | 2003-09-29 | 2005-03-29 | Le Groupe Lysac Inc. | Polysaccharide-clay superabsorbent nanocomposites |
| US8227527B2 (en) | 2003-12-23 | 2012-07-24 | Valorbec S.E.C. | Method and system for making high performance epoxies, and high performance epoxies obtained therewith |
| WO2005071023A1 (en) * | 2004-01-22 | 2005-08-04 | Nuplex Resins B.V. | Stain blocking water borne coating composition |
| CN2691904Y (en) * | 2004-04-01 | 2005-04-13 | 张凤云 | High elasticety aeration-free tyre for hand cart etc. |
| DE102004039451A1 (en) * | 2004-08-13 | 2006-03-02 | Süd-Chemie AG | Polymer blend of incompatible polymers |
| CN101103065B (en) * | 2005-01-14 | 2012-04-18 | 新加坡科技研究局 | Thermoplastic polymer based nanocomposites |
| US7572855B2 (en) * | 2005-01-28 | 2009-08-11 | Bridgestone Corporation | Nano-composite and compositions manufactured thereof |
| US7579398B2 (en) * | 2005-02-02 | 2009-08-25 | Bridgestone Corporation | Nano-composite and compositions therefrom |
| ES2554827T3 (en) | 2005-03-24 | 2015-12-23 | Xyleco, Inc. | Method for densifying fibrous material |
| KR20130118995A (en) * | 2005-09-30 | 2013-10-30 | 듀폰-미쯔이 플루오로케미칼 가부시끼가이샤 | A polymer composition with uniformly distributed nano-sized inorganic particles |
| KR100738830B1 (en) | 2005-10-26 | 2007-07-12 | 금호석유화학 주식회사 | Nanocomposite of styrene-butadiene copolymer and its preparation method |
| CA2632637A1 (en) * | 2005-12-06 | 2007-06-14 | Akzo Nobel N.V. | Nanocomposite material comprising rubber and modified layered double hydroxide, process for its preparation and use thereof |
| US7601772B2 (en) * | 2005-12-20 | 2009-10-13 | Bridgestone Corporation | Nano-composite and method thereof |
| US7687121B2 (en) * | 2006-01-20 | 2010-03-30 | Momentive Performance Materials Inc. | Insulated glass unit with sealant composition having reduced permeability to gas |
| US7531613B2 (en) * | 2006-01-20 | 2009-05-12 | Momentive Performance Materials Inc. | Inorganic-organic nanocomposite |
| US7935184B2 (en) * | 2006-06-19 | 2011-05-03 | Bridgestone Corporation | Method of preparing imidazolium surfactants |
| EP2049756A2 (en) * | 2006-07-21 | 2009-04-22 | Masonite Corporation | Nano-composite door facings, and related door assemblies and methods |
| US20080081182A1 (en) * | 2006-10-02 | 2008-04-03 | Pham Hoai Nam | Fluoropolymer blends with inorganic layered compounds |
| CA2857801C (en) | 2006-10-26 | 2014-12-30 | Xyleco, Inc. | Methods of processing biomass comprising electron-beam radiation |
| BRPI0808956B1 (en) * | 2007-03-16 | 2019-11-05 | National Oilwell Varco Denmark I/S | flexible tubing. |
| US9745453B2 (en) | 2007-06-01 | 2017-08-29 | Plantic Technologies Ltd. | Starch nanocomposite materials |
| US7867358B2 (en) | 2008-04-30 | 2011-01-11 | Xyleco, Inc. | Paper products and methods and systems for manufacturing such products |
| US8236535B2 (en) | 2008-04-30 | 2012-08-07 | Xyleco, Inc. | Processing biomass |
| US7928156B2 (en) * | 2008-06-27 | 2011-04-19 | Sabic Innovative Plastics Ip B.V. | Nanocomposite comprising exfoliated nanoclay-styrenic concentrate and methods of preparation |
| US20100081730A1 (en) * | 2008-09-26 | 2010-04-01 | Klaus Unseld | Process for production of clay nanocomposite |
| US8475584B1 (en) | 2009-10-12 | 2013-07-02 | Raymond Lee Nip | Zinc clays, zinc organoclays, methods for making the same, and compositions containing the same |
| CA2986394C (en) | 2009-10-14 | 2020-04-28 | Xyleco, Inc. | Marking paper products |
| CA2883137C (en) | 2012-08-29 | 2020-03-10 | 7905122 Canada Inc. | Chromogenic absorbent material for animal litter and related chromogenic solution |
| EP3835428B1 (en) | 2014-02-27 | 2023-09-27 | 7905122 Canada Inc. | Chromogenic absorbent material for animal litter |
| WO2016007484A2 (en) | 2014-07-08 | 2016-01-14 | Xyleco, Inc. | Marking plastic-based products |
| JP6727195B2 (en) | 2014-10-01 | 2020-07-22 | 7905122 カナダ インコーポレイテッド | Process for the production of absorbent materials |
| KR101807019B1 (en) | 2015-12-15 | 2017-12-08 | 현대자동차 주식회사 | Process for preparing organic-inorganic hybrid porous insulation coating composition for enameled winding wires |
| WO2017106651A1 (en) * | 2015-12-17 | 2017-06-22 | Aramco Services Company | Targeting enhanced production through deep carbonate stimulation; stabilized acid emulsions containing insoluble solid materials with desired wetting properties |
| WO2017165953A1 (en) | 2016-04-01 | 2017-10-05 | 7905122 Canada Inc. | Water-absorbing material and uses thereof |
| US10626314B1 (en) | 2016-07-11 | 2020-04-21 | Byk-Chemie, Gmbh | Additive for drilling fluids |
| CN109021250B (en) * | 2018-06-12 | 2020-04-07 | 江南大学 | Preparation of waterborne polyurethane modified montmorillonite nano composite emulsion |
| TW202112669A (en) * | 2019-08-22 | 2021-04-01 | 多能顧問股份有限公司 | Metallic oxide/nsp nano-composite and method for producing the same |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5464472A (en) * | 1993-06-26 | 1995-11-07 | Hoechst Aktiengesellschaft | Fine aqueous dispersion of an organophilic sheet silicate |
| US5969029A (en) * | 1992-07-29 | 1999-10-19 | Sumitomo Chemical Company, Limited | Process for producing a gas barrier resin composition |
| EP1055706A1 (en) * | 1999-05-25 | 2000-11-29 | Rheox, Inc. | Smectite clay/organic chemical/polymer compositions useful as nanocomposites |
Family Cites Families (111)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2367384A (en) | 1942-09-22 | 1945-01-16 | Shell Dev | Method of removing oil from water |
| US2531427A (en) | 1946-05-03 | 1950-11-28 | Ernst A Hauser | Modified gel-forming clay and process of producing same |
| US2531440A (en) | 1947-03-29 | 1950-11-28 | Nat Lead Co | Lubricants |
| US2531396A (en) | 1947-03-29 | 1950-11-28 | Nat Lead Co | Elastomer reinforced with a modified clay |
| US2531812A (en) | 1948-01-16 | 1950-11-28 | Ernst A Hauser | Application of drilling fluids |
| US2552775A (en) | 1948-03-20 | 1951-05-15 | Union Oil Co | Drilling fluid |
| BE504196A (en) | 1950-06-24 | |||
| US2622987A (en) | 1951-04-10 | 1952-12-23 | Nat Lead Co | Coating composition and the vehicle therefor containing a compound of a clay and an onium base |
| US2750296A (en) | 1952-02-13 | 1956-06-12 | Sun Chemical Corp | Printing ink |
| US2767177A (en) | 1952-10-03 | 1956-10-16 | Gen Mills Inc | Complexes of bentonite, polyamine and monoquaternary ammonium compounds |
| US2739067A (en) | 1952-11-12 | 1956-03-20 | Nat Lead Co | Printing inks |
| US2754219A (en) | 1953-03-09 | 1956-07-10 | Huber Corp J M | Anti-misting printing inks |
| US2795545A (en) | 1953-04-14 | 1957-06-11 | Monsanto Chemicals | Organic materials |
| US2883356A (en) | 1953-05-27 | 1959-04-21 | Monsanto Chemicals | Composition containing a plastic material and a modified clay |
| US3136819A (en) | 1958-05-16 | 1964-06-09 | Armour & Co | Preparation of tertiary aliphatic methyl amines |
| US3027322A (en) | 1958-07-21 | 1962-03-27 | Nat Lead Co | Process of preparing a well drilling fluid |
| BE582883A (en) | 1958-10-28 | |||
| US3125547A (en) | 1961-02-09 | 1964-03-17 | Extrudable composition consisting of | |
| US3084117A (en) | 1961-04-04 | 1963-04-02 | Union Oil Co | Organoclay-polyolefin compositions |
| US3839389A (en) | 1963-12-06 | 1974-10-01 | Laporte Industries Ltd | Organophilic swelling clays |
| US3471439A (en) | 1966-03-10 | 1969-10-07 | Amicon Corp | Reinforcing filler |
| US3537994A (en) | 1967-07-25 | 1970-11-03 | Nat Lead Co | Organophilic clay greases |
| US3567680A (en) | 1968-05-03 | 1971-03-02 | Huber Corp J M | Surface modified pigments and methods for producing same and elastomers containing same |
| GB1313749A (en) | 1969-10-02 | 1973-04-18 | Canadian Patents Dev | Polymeric high performance composites |
| US3691070A (en) | 1970-04-27 | 1972-09-12 | Nat Lead Co | Employment of bentonite in brine muds |
| US3671190A (en) | 1970-11-10 | 1972-06-20 | Laporte Industries Ltd | Synthetic clay-like minerals of the smectite type and method of preparation |
| US3804656A (en) | 1972-02-22 | 1974-04-16 | Engelhard Min & Chem | Pigment dispersions and use thereof |
| US3843591A (en) | 1972-06-05 | 1974-10-22 | Monsanto Co | Reinforced polyamide compositions |
| US4049780A (en) | 1972-11-14 | 1977-09-20 | Laporte Industries Limited | Production of magnesium silicates |
| US3915867A (en) | 1973-04-24 | 1975-10-28 | Stepan Chemical Co | Domestic laundry fabric softener |
| US3951850A (en) | 1973-06-22 | 1976-04-20 | Clocker Edwin T | Conversion of clay to its colloidal form by hydrodynamic attrition |
| US4053493A (en) | 1973-10-01 | 1977-10-11 | Exxon Research & Engineering Co. | Layered tetraalkyl phosphonium clays |
| US4087365A (en) | 1974-01-28 | 1978-05-02 | American Colloid Company | Super-yield bentonite base drilling fluid |
| US3988287A (en) | 1974-02-09 | 1976-10-26 | Teijin Limited | Polyamide compositions |
| US4221697A (en) | 1974-05-29 | 1980-09-09 | Imperial Chemical Industries Limited | Composite materials |
| US3974125A (en) | 1974-09-27 | 1976-08-10 | Exxon Research And Engineering Company | Higher dialkyl dimethyl ammonium clay gelling agents for unsaturated polyester compositions |
| DE2450673B2 (en) | 1974-10-25 | 1981-04-30 | Basf Ag, 6700 Ludwigshafen | Process for the continuous production of polyamides |
| US3977894A (en) | 1975-09-19 | 1976-08-31 | Nl Industries, Inc. | Rheological agent for non-aqueous fluid systems |
| US4240951A (en) | 1975-12-23 | 1980-12-23 | Yara Engineering Corporation | Rheological control of polyester-styrene resin compositions |
| US4040974A (en) | 1976-04-26 | 1977-08-09 | N L Industries, Inc. | Synthesized gellants containing smectite-type clay and process for producing same |
| US4105578A (en) | 1976-12-10 | 1978-08-08 | N L Industries, Inc. | Organophilic clay having enhanced dispersibility |
| US4081496A (en) | 1977-06-27 | 1978-03-28 | N L Industries, Inc. | Thixotropic polyester compositions containing an organophilic clay gellant |
| US4116866A (en) | 1977-07-01 | 1978-09-26 | N L Industries, Inc. | Organophilic clay gellant |
| US4291154A (en) | 1978-05-22 | 1981-09-22 | Blount David H | Process for the production of polyamide silicate resinous product |
| US4315828A (en) | 1978-03-10 | 1982-02-16 | Max L. Wymore | Water based window glass and chrome cleaner composition |
| US4216135A (en) | 1978-03-27 | 1980-08-05 | Nl Industries, Inc. | Organophilic clays and thixotropic polyester compositions containing the same |
| EP0010929B1 (en) | 1978-10-27 | 1983-02-02 | Toray Industries, Inc. | Highly rigid polyamide composition and a method for its manufacture |
| DE2928603A1 (en) | 1979-07-14 | 1981-02-05 | Hoechst Ag | QUATERNAIRE AMMONIUM COMPOUNDS, THEIR PRODUCTION AND THE USE THEREOF AS SOFT SOFTENER |
| DE3066683D1 (en) | 1979-07-26 | 1984-03-29 | Ici Plc | A dispersible pigment composition, its preparation and use in the coloration of thermoplastic materials and paints |
| US4314919A (en) | 1980-03-12 | 1982-02-09 | Engelhard Minerals & Chemicals Corporation | Method of thickening liquid polyester system with clay |
| US4444714A (en) | 1980-08-26 | 1984-04-24 | American Organics Corporation | Method of coloring resin products |
| US4341565A (en) | 1980-08-26 | 1982-07-27 | American Organics Corporation | Liquid colorant composition |
| US4386010A (en) | 1980-09-02 | 1983-05-31 | Engelhard Corporation | Treated attapulgite clay composition |
| US4569923A (en) | 1980-10-03 | 1986-02-11 | Southern Clay Products, Inc. | Process for manufacturing organoclays having enhanced gelling properties |
| US4412018A (en) | 1980-11-17 | 1983-10-25 | Nl Industries, Inc. | Organophilic clay complexes, their preparation and compositions comprising said complexes |
| US4434075A (en) | 1981-10-19 | 1984-02-28 | Nl Industries, Inc. | Anionically modified organophilic clays and their preparation |
| US4410364A (en) | 1980-11-17 | 1983-10-18 | Nl Industries, Inc. | Printing ink compositions |
| US4434076A (en) | 1981-10-19 | 1984-02-28 | Nl Industries, Inc. | Clay cation complexes and their use to increase viscosity of liquid organic systems |
| US4391637A (en) | 1981-10-19 | 1983-07-05 | Nl Industries, Inc. | Rheological additive for non-aqueous fluid systems |
| US4450095A (en) | 1980-11-17 | 1984-05-22 | Nl Industries, Inc. | Organophilic clay gellant having enhanced dispersibility |
| JPS5790050A (en) | 1980-11-26 | 1982-06-04 | Toyota Central Res & Dev Lab Inc | Preparation of composite material consisting of clay mineral and organic polymer |
| GB2092600A (en) | 1981-02-11 | 1982-08-18 | Du Pont | Mineral reinforced polyamides |
| US4464274A (en) | 1981-08-13 | 1984-08-07 | Venture Innovations, Inc. | Organophilic clay suspending agents |
| US4382868A (en) | 1981-08-13 | 1983-05-10 | Venture Innovations, Inc. | Organophilic clay gellants |
| US4473477A (en) | 1981-09-30 | 1984-09-25 | Radecca, Inc. | Method of organic waste disposal |
| US4470912A (en) | 1981-09-30 | 1984-09-11 | Radecca, Inc. | Method of breaking emulsions |
| US4517094A (en) | 1981-09-30 | 1985-05-14 | Radecca, Inc. | Process for treating organics contaminated water |
| US4462470A (en) | 1981-10-08 | 1984-07-31 | American Colloid Company | Extrusion of bentonite clay for fluid loss reduction in drilling fluids |
| US4465542A (en) | 1982-02-19 | 1984-08-14 | Mitsui Petrochemical Industries, Ltd. | Adhesive composition |
| US4431755A (en) | 1982-07-16 | 1984-02-14 | Standard Oil Company (Indiana) | Rubber composition comprising phyllosilicate minerals, silanes, and quaternary ammonium salts |
| US4528104A (en) | 1982-08-19 | 1985-07-09 | Nl Industries, Inc. | Oil based packer fluids |
| US4549966A (en) | 1982-09-20 | 1985-10-29 | Radecca, Inc. | Method of removing organic contaminants from aqueous compositions |
| JPS5956443A (en) | 1982-09-24 | 1984-03-31 | Mitsubishi Gas Chem Co Inc | Resin composition for molding material |
| US4455382A (en) | 1983-01-27 | 1984-06-19 | Corning Glass Works | Organic-inorganic composites of neutralized polyelectrolyte complexes |
| US4454237A (en) | 1983-01-27 | 1984-06-12 | Corning Glass Works | Organic-inorganic composites containing synthetic mica |
| US4480060A (en) | 1983-01-27 | 1984-10-30 | Corning Glass Works | Mica-resin composite material |
| US4473675A (en) | 1983-02-01 | 1984-09-25 | Southern Clay Products, Inc. | Thixotropic cross-linkable unsaturated polyester compositions and method of production |
| US4508628A (en) | 1983-05-19 | 1985-04-02 | O'brien-Goins-Simpson & Associates | Fast drilling invert emulsion drilling fluids |
| JPS601256A (en) | 1983-06-19 | 1985-01-07 | Nippon Steel Chem Co Ltd | Polyamide resin composition |
| US4552712A (en) | 1983-06-28 | 1985-11-12 | Union Carbide Corporation | Process for reducing surface melt fracture during extrusion of ethylene polymers |
| US4664842A (en) | 1983-12-13 | 1987-05-12 | Southern Clay Products, Inc. | Process for manufacturing organoclays having enhanced gelling properties |
| US4558075A (en) | 1984-03-30 | 1985-12-10 | Ppg Industries, Inc. | High-solids coating composition for improved rheology control containing organo-modified clay |
| US4620993A (en) | 1984-03-30 | 1986-11-04 | Ppg Industries, Inc. | Color plus clear coating system utilizing organo-modified clay in combination with organic polymer microparticles |
| TR22515A (en) | 1984-04-27 | 1987-09-17 | English Clays Lovering Pochin | PREPARING AN ORGANO-HAIR EASILY TO DISPERSION IN AN ORGANIC VASAT |
| NL8401545A (en) | 1984-05-14 | 1985-12-02 | Gen Electric | POLYMER MIXTURE CONTAINING A POLYPHENYLENE ETHER AND A POLYAMIDE. |
| US4600515A (en) | 1984-09-12 | 1986-07-15 | National Starch And Chemical Corporation | Fluid loss control agents for drilling fluids containing divalent cations |
| US4789403A (en) * | 1986-07-22 | 1988-12-06 | E.C.C. America Inc. | Surface modified layered lattice silicate pigments |
| US4690868A (en) | 1985-02-08 | 1987-09-01 | E.C.C. America Inc. | Process for surface treating clay minerals and resultant products |
| DE3520314A1 (en) | 1985-06-07 | 1986-12-11 | Hoechst Ag, 6230 Frankfurt | GEL-FORMING ORGANOPHILIC LAYERED SILICATE, METHOD FOR THE PRODUCTION AND USE THEREOF |
| US4640716A (en) | 1985-06-26 | 1987-02-03 | Engelhard Corporation | High bulking pigment and method of making same |
| US4631091A (en) | 1985-08-13 | 1986-12-23 | English China Clays Lovering Pochin & Co. Ltd. | Method for improving the dispersibility of organoclays |
| US4683259A (en) * | 1985-08-13 | 1987-07-28 | Ecc International Limited | Resin compositions comprising organoclays of improved dispersibility |
| US4695402A (en) | 1985-08-20 | 1987-09-22 | Nl Chemicals, Inc. | Organophilic clay gellants and process for preparation |
| US4739007A (en) | 1985-09-30 | 1988-04-19 | Kabushiki Kaisha Toyota Chou Kenkyusho | Composite material and process for manufacturing same |
| GB8531254D0 (en) | 1985-12-19 | 1986-01-29 | English Clays Lovering Pochin | Organophilic compositions |
| DE3789518T2 (en) | 1986-01-31 | 1994-10-27 | Toray Industries | Laminated film and antistatic laminated film. |
| US4753974A (en) | 1986-12-12 | 1988-06-28 | E C.C. International Limited | Dispersible organoclay for unsaturated polyester resins |
| US4775586A (en) | 1987-02-17 | 1988-10-04 | Armstrong World Industries, Inc. | Paper, paper products, films composites and other silicate-polymer, construction materials |
| US4804703A (en) | 1987-07-12 | 1989-02-14 | E. I. Du Pont De Nemours And Company | Mineral reinforced nylon compositions for blowmolding |
| JPH0747644B2 (en) * | 1989-05-19 | 1995-05-24 | 宇部興産株式会社 | Polyamide composite material and method for producing the same |
| US5266538A (en) * | 1990-12-21 | 1993-11-30 | Southern Clay Products, Inc. | Method for preparing high solids bentonite slurries |
| CA2115255A1 (en) * | 1991-08-12 | 1993-03-04 | Macrae Maxfield | Melt process formation of polymer nanocomposite of exfoliated layered material |
| US5429999A (en) * | 1991-11-14 | 1995-07-04 | Rheox, Inc. | Organoclay compositions containing two or more cations and one or more organic anions, their preparation and use in non-aqueous systems |
| US5955535A (en) * | 1993-11-29 | 1999-09-21 | Cornell Research Foundation, Inc. | Method for preparing silicate-polymer composite |
| US5554670A (en) * | 1994-09-12 | 1996-09-10 | Cornell Research Foundation, Inc. | Method of preparing layered silicate-epoxy nanocomposites |
| KR100453938B1 (en) * | 1995-11-07 | 2005-01-31 | 사우던 클레이 프로덕츠, 아이엔시. | An organic clay composition for gelling an unsaturated polyester resin |
| US6287992B1 (en) * | 1998-04-20 | 2001-09-11 | The Dow Chemical Company | Polymer composite and a method for its preparation |
| US6380295B1 (en) * | 1998-04-22 | 2002-04-30 | Rheox Inc. | Clay/organic chemical compositions useful as additives to polymer, plastic and resin matrices to produce nanocomposites and nanocomposites containing such compositions |
| US6271298B1 (en) * | 1999-04-28 | 2001-08-07 | Southern Clay Products, Inc. | Process for treating smectite clays to facilitate exfoliation |
| US6521678B1 (en) * | 2000-11-21 | 2003-02-18 | Argonne National Laboratory | Process for the preparation of organoclays |
| US6858665B2 (en) * | 2001-07-02 | 2005-02-22 | The Goodyear Tire & Rubber Company | Preparation of elastomer with exfoliated clay and article with composition thereof |
-
2002
- 2002-02-28 AT AT02731107T patent/ATE325155T1/en not_active IP Right Cessation
- 2002-02-28 EP EP02731107A patent/EP1366109B1/en not_active Expired - Lifetime
- 2002-02-28 WO PCT/US2002/006055 patent/WO2002070589A2/en active IP Right Grant
- 2002-02-28 CA CA002439632A patent/CA2439632A1/en not_active Abandoned
- 2002-02-28 DE DE60211129T patent/DE60211129T2/en not_active Expired - Fee Related
- 2002-02-28 US US10/086,173 patent/US6849680B2/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5969029A (en) * | 1992-07-29 | 1999-10-19 | Sumitomo Chemical Company, Limited | Process for producing a gas barrier resin composition |
| US5464472A (en) * | 1993-06-26 | 1995-11-07 | Hoechst Aktiengesellschaft | Fine aqueous dispersion of an organophilic sheet silicate |
| EP1055706A1 (en) * | 1999-05-25 | 2000-11-29 | Rheox, Inc. | Smectite clay/organic chemical/polymer compositions useful as nanocomposites |
Non-Patent Citations (3)
| Title |
|---|
| C.O. ORIAKHI ET. AL.: "Poly(pyrrole) and poly(thiophene)/clay nanocomposites via latex-colloid interaction" MATERIALS RESEARCH BULLETIN, vol. 30, no. 6, 1995, pages 723-729, XP002211331 OREGON, USA * |
| DATABASE CHEMABS [Online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, USA; database accession no. 133:121513, XP002211332 * |
| See also references of EP1366109A2 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1471108A1 (en) * | 2003-04-25 | 2004-10-27 | Rohm And Haas Company | Composite compositions including polymeric nanoparticles and clay nanoparticles |
| US7244781B2 (en) | 2003-04-25 | 2007-07-17 | Rohm And Haas Company | Composite compositions including polymeric nanoparticles and clay nanoparticles |
| WO2006085957A3 (en) * | 2004-07-06 | 2007-03-22 | Exxonmobil Chem Patents Inc | Polymeric nanocomposites and processes for making the same |
| JP2008506009A (en) * | 2004-07-06 | 2008-02-28 | エクソンモービル・ケミカル・パテンツ・インク | Polymer nanocomposites |
| US8980978B2 (en) | 2004-07-06 | 2015-03-17 | Exxonmobil Chemical Patents Inc. | Polymeric nanocomposites and processes for making the same |
| EP3500770B1 (en) | 2016-08-17 | 2022-10-05 | ContiTech Luftfedersysteme GmbH | Article, particularly an air spring bellows, a metal-rubber element, or a vibration damper |
| EP3500771B1 (en) | 2016-08-17 | 2022-11-02 | ContiTech Elastomer-Beschichtungen GmbH | Rubber mixture and elastomeric article having flame-retardant properties |
Also Published As
| Publication number | Publication date |
|---|---|
| US6849680B2 (en) | 2005-02-01 |
| EP1366109B1 (en) | 2006-05-03 |
| DE60211129T2 (en) | 2007-05-03 |
| US20020165305A1 (en) | 2002-11-07 |
| CA2439632A1 (en) | 2002-09-12 |
| DE60211129D1 (en) | 2006-06-08 |
| WO2002070589A3 (en) | 2003-05-01 |
| ATE325155T1 (en) | 2006-06-15 |
| EP1366109A2 (en) | 2003-12-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6849680B2 (en) | Preparation of polymer nanocomposites by dispersion destabilization | |
| CA2255488C (en) | Clay/organic chemical compositions useful as additives to polymer, plastic and resin matrices to produce nanocomposites and nanocomposites containing such compositions | |
| US5880197A (en) | Intercalates and exfoliates formed with monomeric amines and amides: composite materials containing same and methods of modifying rheology therewith | |
| US20060199890A1 (en) | Nanocomposites including modified fillers | |
| US6730719B2 (en) | Process for treating smectite clays to facilitate exfoliation | |
| US6242500B1 (en) | Intercalates and exfoliates formed with long chain (C6+) or aromatic matrix polymer-compatible monomeric, oligomeric or polymeric intercalant compounds, and composite materials containing same | |
| US6521678B1 (en) | Process for the preparation of organoclays | |
| JP2009526904A (en) | Polymer composite, polymer nanocomposite and method | |
| CN101384673A (en) | Organoclay suitable for use in halogenated resin and composite systems thereof | |
| US20060199889A1 (en) | Silanated clay compositions and methods for making and using silanated clay compositions | |
| EP3360918A1 (en) | Polymer nanocomposite masterbatch, polymer nanocomposite and methods for preparation thereof | |
| US7160942B2 (en) | Polymer-phyllosilicate nanocomposites and their preparation | |
| US8012540B2 (en) | Aqueous emulsion comprising a functionalized polyolefin and carbon nanotubes | |
| AU2002303106A1 (en) | Preparation of polymer nanocomposites by dispersion destabilization | |
| Ma et al. | Manufacturing techniques of rubber nanocomposites | |
| Abdelaal et al. | An overview on polysulphone/clay nanocomposites | |
| Luecha et al. | A novel and facile nanoclay aerogel masterbatch toward exfoliated polymer-clay nanocomposites through a melt-mixing process | |
| US20110048282A1 (en) | Hybrid nanoparticles with controlled morphology and their use in thermoplastic polymer matrix nanocomposites | |
| Jayaraj et al. | Review on development of natural rubber/nanoclay nanocomposites | |
| Edraki et al. | Study on the Optical and Rheological properties of polymer-layered Silicate Nanocomposites | |
| EP1155066B1 (en) | Nanocomposite | |
| US7435773B1 (en) | Resin composite and method for producing the same | |
| ANAZY et al. | Effect of Clay Modification and Preparation Method on Crystalline Structure of Isotactic Polypropylene/Organoclay Nanocomposites | |
| WO2005030850A1 (en) | Process to obtain an intercalated or exfoliated polyester with clay hybrid nanocomposite material | |
| Makadia | Nanocomposites of polypropylene by polymer melt compounding approach |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2439632 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002731107 Country of ref document: EP Ref document number: 2002303106 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2002731107 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: JP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2002731107 Country of ref document: EP |