INHIBITORS OF 11-BETA-HYDROXY STEROID DEHYDROGENASE
TYPE 1
RELATED APPLICATIONS
This application claims priority to Swedish application number 0103913-0, filed on November 22, 2001, Swedish application number 0104051-8, filed on November 30, 2001, Swedish application number 0103915-5, filed on November 22, 2001, U.S. provisional application number 60/348,468, filed on January 14, 2002, U.S. provisional application number 60/348,340, filed on January 14, 2002, the contents of which are incorporated herein by reference.
TECHNICAL FIELD
The present invention relates to novel compounds, to pharmaceutical compositions comprising the compounds, as well as to the use of the compounds in medicine and for the preparation of a medicament which acts on the human 11-β-hydroxysteroid dehydrogenase type 1 enzyme (llβHSDl).
BACKGROUND
1. Glucorticoids, diabetes and hepatic glucose production
It has been known for more than half a century that glucocorticoids have a central role in diabetes, e.g. the removal of the pituitary or the adrenal gland from a diabetic animal alleviates the most severe symptoms of diabetes and lowers the concentration of glucose in the blood (Long, CD. and F.D.W. Leukins (1936) J. Exp. Med. 63: 465-490; Houssay, B.A.
(1942) Endocrinology 30: 884-892). It is also well established that glucocorticoids enable the effect of glucagon on the liver. The role of 11 βHSD 1 as an important regulator of local glucocorticoid effect and thus of hepatic glucose production is well substantiated (see e.g. Jamieson et al. (2000) J.
Endocrinol. 165: p. 685-692). The hepatic insulin sensitivity was improved in healthy human volunteers treated with the non-specific llβHSDl inhibitor carbenoxolone (Walker, B.R. et al. (1995) J. Clin. Endocrinol. Metab. 80: 3155-3159). Furthermore, the expected mechanism has been established by different experiments with mice and rats. These studies showed that the mRNA levels and activities of two key enzymes in hepatic glucose production were reduced, namely: the rate-limiting enzyme in gluconeogenesis, phosphoenolpyruvate carboxykinase (PEPCK), and glucose-6-phosphatase (G6Pase) catalyzing the last common step of gluconeogenesis and glycogenolysis. Finally, the blood glucose level and hepatic glucose production is reduced in mice having the 1 lβHSDl gene knocked-out. Data from this model also confirm that inhibition of 11 βHSD 1 will not cause hypoglycemia, as predicted since the basal levels of PEPCK and G6Pase are regulated independently of glucocorticoids (Kotelevtsev, Y. et al, (1997) Proc. Natl. Acad. Sci. USA 94: 14924-14929).
Arzneim.-Forsch./Drug Res; 44 (II), No. 7, 821-826, 1994, discloses the hypoglycemic compounds 4-(3-methyl-5-oxo-2-pyrazolin-l-yl)benzoic acid and 1- (mesitylen-2-sulfonyl)-lH-l,2,4-triazole. The structures of these compounds differ considerably from the structure of the compounds of the present invention, in that the latter are fhiophenes having an (hetero)arylsulfonamido substituent.
FR 2,384,498 discloses compounds having a high hypoglycemic effect. Therefore, treatment of hyperglycemia with these compounds may lead to hypoglycemia.
2. Possible reduction of obesity and obesity related cardiovascular risk factors
Obesity is an important factor in syndrome X as well as in the majority (> 80%) of type 2 diabetic, and omental fat appears to be of central importance. Abdominal obesity is closely associated with glucose intolerance, hyperinsulinemia, hypertriglyceridemia, and other factors of the so-called syndrome X (e.g. raised blood pressure, decreased levels of HDL and increased levels of VLDL) (Montague & O'Rahilly, Diabetes 49: 883-888, 2000). Inhibition of the enzyme in pre-adipocytes (stromal cells) has been shown to decrease the rate of differentiation into adipocytes. This is predicted to result in diminished expansion (possibly reduction) of the omental fat depot, i.e. reduced central obesity (Bujalska, I.J., S. Kumar, and P.M. Stewart (1997) Lancet 349: 1210-1213).
Inhibition of 1 lβHSDl in mature adipocytes is expected to attenuate secretion of the plasminogen activator inhibitor 1 (PAI-1) - an independent cardiovascular risk factor (Halleux, CM. et al. (1999) J. Clin. Endocrinol. Metab. 84: 4097-4105). Furthermore, there is a clear correlation between glucocorticoid "activity" and cardiovascμlar risk factore suggesting that a reduction of the glucocorticoid effects would be beneficial (Walker, B.R. et al. (1998) Hypertension 31: 891-895; Fraser, R. et al. (1999) Hypertension 33: 1364-1368).
Adrenalectomy attenuates the effect of fasting to increase both food intake and hypothalamic neuropeptide Y expression. This supports the role of glucocorticoids in promoting food intake and suggests that inhibition of 11 βHSDl in the brain might increase satiety and therefore reduce food intake (Woods, S.C. et al. (1998) Science, 280: 1378-1383).
3. Possible beneficial effect on the pancreas
Inhibition of 11 βHSDl in isolated murine pancreatic β-cells improves the glucose- stimulated insulin secretion (Davani, B. et al. (2000) J. Biol. Chem. 2000 Nov 10; 275(45): 34841-4). Glucocorticoids were previously known to reduce pancreatic insulin release in vivo (Billaudel, B. and B.C.J. Sutter (1979) Horm. Metab. Res. 11: 555-560). Thus, inhibition of 11 βHSDl is predicted to yield other beneficial effects for diabetes treatment, besides effects on liver and fat.
4. Possible beneficial effects on cognition and dementia
Stress and glucocorticoids influence cognitive function (de Quervain, D.J.-F., B. Roozendaal, and J.L. McGaugh (1998) Nature 394: 787-790). The enzyme llβHSDl controls the level of glucocorticoid action in the brain and thus contributes to neurotoxicity (Rajan, V., C.R.W. Edwards, and J.R. Seckl, J. (1996) Neuroscience 16: 65-70; Seckl, J.R., Front. (2000) Neuroendocrinol. 18: 49-99). Unpublished results indicate significant memory improvement in rats treated with a non-specific llβHSDl inhibitor (J. Seckl, personal communication). Based the above and on the known effects of glucocorticoids in the brain, it may also be suggested that inhibiting 11 βHSD 1 in the brain may result in reduced anxiety (Tranche, F. et al. (1999) Nature Genetics 23: 99-103). Thus, taken together, the hypothesis
is that inhibition of 11 βHSDl in the human brain would prevent reactivation of cortisone into cortisol and protect against deleterious glucocorticoid-mediated effects on neuronal survival and other aspects of neuronal function, including cognitive impairment, depression, and increased appetite (previous section).
WO 98/27081 and WO 99/02502 disclose 5HT6 receptor antagonists for the treatment of CNS disorders. None of these compounds fall within formula (I) according to the present invention. Furthermore, nothing is said about the activity on 11 βHSDl .
5. Possible use of immuno-modulation using l lβHSDl inhibitors
The general perception is that glucocorticoids suppress the immune system. But in fact there is a dynamic interaction between the immune system and the HPA (hypothalamo- pituitary-adrenal) axis (Rook, G.A.W. (1999) Baillier's Clin. Endocrinol. Metab. 13: 576- 581). The balance between the cell-mediated response and humoral responses is modulated by glucocorticoids. A high glucocorticoid activity, such as at a state of stress, is associated with a humoral response. Thus, inhibition of the enzyme llβHSDl has been suggested as a means of shifting the response towards a cell-based reaction.
In certain disease states, including tuberculosis, lepra and psoriasis the immune reaction is normaly biased towards a humoral response when in fact the appropriate response would be cell based. Temporal inhibition of 11 βHSD 1 , local or systemic, might be used to push the immune system into the appropriate response (Mason, D. (1991) Immunology Today 12: 57-60; Rook et al., supra).
An analogous use of 11 βHSDl inhibition, in this case temporal, would be to booster the immune response in association with immunization to ensure that a cell based response would be obtained, when desired.
6. Reduction of intraocular pressure
Recent data suggest that the levels of the glucocorticoid target receptors and the 1 lβHSD enzymes determines the susceptibility to glaucoma (Stokes, J. et al. (2000) Invest. Ophthalmol. 41: 1629-1638). Further, inhibition of llβHSDl was recently presented as a novel approach to lower the intraocular pressure (Walker E. A. et al, poster P3-698 at the Endocrine society meeting June 12-15, 1999, San Diego). Ingestion of carbenoxolone, a nonspecific inhibitor of 11 βHSDl, was shown to reduce the intraocular pressure by 20% in normal subjects. In the eye, expression of 11 βHSDl is confined to basal cells of the corneal epithelium and the non-pigmented epithelialium of the cornea (the site of aqueous production), to ciliary muscle and to the sphincter and dilator muscles of the iris. In contrast, the distant isoenzyme 1 lβHSD2 is highly expressed in the non-pigmented ciliary epithelium and corneal endothelium. None of the enzymes is found at the trabecular meshwork, the site of drainage. Thus, 11 βHSDl is suggested to have a role in aqueous production, rather than drainage, but it is presently unknown if this is by interfering with activation of the glucocorticoid or the mineralocorticoid receptor, or both.
7. Reduced osteoporosis
Glucocorticoids have an essential role in skeletal development and function but are detrimental in excess. Glucocorticoid-induced bone loss is derived, at least in part, via inhibition of bone formation, which includes suppression of osteoblast proliferation and collagen synthesis (Kim, C.H., S.L. Cheng, and G.S. Kim (1999) J. Endocrinol. 162: 371- 379). The negative effect on bone nodule formation could be blocked by the non-specific inhibitor carbenoxolone suggesting an important role of 11 βHSDl in the glucocorticoid effect (Bellows, C.G., A. Ciaccia, and J.N.M. Heersche, (1998) Bone 23: 119-125). Other data suggest a role of 11 βHSDl in providing sufficiently high levels of active glucocorticoid in osteoclasts, and thus in augmenting bone resorption (Cooper, M.S. et al. (2000) Bone 27: 375-381). Taken together, these different data suggest that inhibition of 1 lβHSDl may have beneficial effects against osteoporosis by more than one mechanism working in parallel.
8. Reduction of hypertension
Bile acids inhibit 1 lβ-hydroxysteroid dehydrogenase type 2. This results in a shift in the overall body balance in favour of cortisol over cortisone, as shown by studying the ratio of the urinary metabolites (Quattropani C, Vogt B, Odermatt A, Dick B, Frey BM, Frey FJ. 2001. J Clin Invest. Nov;108(9):1299-305. "Reduced activity of 1 lbeta-hydroxysteroid dehydrogenase in patients with cholestasis".). Reducing the activity of 1 lbHSDl in the liver by a selective inhibitor is predicted to reverse this imbalance, and acutely counter the symptoms such as hypertension, while awaiting surgical treatment removing the biliary obstruction.
WO 99/65884 discloses carbon substituted aminothiazole inhibitors of cyclin dependent kinases. These compounds may e.g. be used against cancer, inflammation and arthritis. US 5,856,347 discloses an antibacterial preparation or bactericide comprising 2- aminothiazole derivative and/or salt thereof. Further, US 5,403,857 discloses benzenesulfonamide derivatives having 5-lipoxygenase inhibitory activity. Additionally, tetrahydrothiazolo[5,4-c]pyridines are disclosed in: Analgesic tetrahydrothiazolo[5,4- yridines. Fr. Addn. (1969), 18 pp, Addn. to Fr. 1498465. CODEN: FAXXA3; FR 94123 19690704 CAN 72:100685 AN 1970:100685 CAPLUS and 4,5,6,7-Tetrahydrothiazolo[5,4- c]pyridines. Neth. Appl. (1967), 39 pp. CODEN: NAXXAN NL 6610324 19670124 CAN 68:49593, AN 1968: 49593 CAPLUS. However, none of the above disclosures discloses the compounds according to the present invention, or their use for the treatment of diabetes, obesity, glaucoma, osteoporosis, cognitive disorders, immune disorders, depression, and hypertension.
WO 98/16520 discloses compounds inhibiting matrix metalloproteinases (MMPs) and TNF-α converting enzyme (TACE). EP 0 749 964 Al and US 5,962,490 disclose compounds having an endothelin receptor antagonist activity. WO 00/02851 discloses compounds associated with a disturbed cGMP balance. None of these compounds fall within formula (I) according to the present invention. Furthermore, nothing is said about the activity on l lβHSDl. US 5,783,697 discloses thiophene derivatives as inhibitors of PGE2 and LTB4.
Nothing is said about the activity on llβHSDl.
EP 0 558 258, EP 0 569 193, and EP 1 069 114 disclose isoxazole derivatives as endothelin agonists and antagonists. Nothing is said about the activity on llβHSDl.
Consequently, there is a need of new compounds that are useful in the treatment of diabetes, obesity, glaucoma, osteoporosis, cognitive disorders, immune disorders, depression, and hypertension.
SUMMARY OF THE INVENTION
The compounds according to the present invention solves the above problems and embraces a novel class of compounds which has been developed and which inhibit the human 11-β-hydroxysteroid dehydrogenase type 1 enzyme (11-β-HSDi), and may therefore be of use in the treating disorders such as diabetes, obesity, glaucoma, osteoporosis, cognitive disorders, immune disorders, and hypertension.
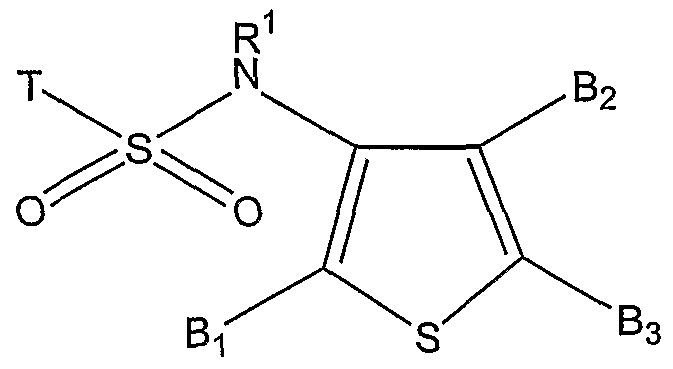
One object of the present invention is a compound of formula (I)
wherein:
T is an aryl ring or heteroaryl ring, optionally independently substituted by [R]n, wherein n is an integer 0-5, and R is hydrogen, aryl, heteroaryl, a heterocyclic ring, optionally halogenated C1-6-alkyl, optionally halogenated C1-6-alkoxy, C1-6-alkylsulfonyl, carboxy, cyano, nitro, halogen, amine which is optionally mono- or di-substituted, amide which is optionally mono- or di-substituted, aryloxy, arylsulfonyl, arylamino, wherein aryl, heteroaryl and aryloxy residues and heterocyclic rings can further be optionally substituted in one or more positions independently of each other by C1-6-acyl, C1-6-alkylthio, cyano, nitro, hydrogen, halogen, optionally halogenated Cι-6-alkyl, optionally halogenated C1-6-alkoxy,
amide which is optionally mono- or di-substituted, (benzoylamino)methyl, carboxy, 2- thienylmethylamino or ({[4-(2-ethoxy-2-oxoethyl)-l,3-thiazol-2-yl]amino}carbonyl); R1 is hydrogen or C1-6-alkyl;
Bi and B2 are B3 or Z, provided that Bi and B2 have different meanings, wherein: • Z is selected from an aryl ring or heteroaryl ring, which can further be optionally substituted in one or more positions independently of each other by hydrogen, C1-6-alkyl, halogenated Cι-6-alkyl, halogen, C1-6-alkoxy, nitro, C1-6-alkoxycarbonyl, C1-6- alkylsulfonyl, acetylamino or aryloxy, wherein the aryloxy can further be optionally substituted in one or more positions independently of each other by hydrogen and halogen; or is X-Y-R2, wherein
• X is CH2 or CO;
• Y is CH2, CO or a single bond;
• R2 is selected from C1-6-alkyl, azido, arylthio, heteroarylthio, halogen, hydroxymethyl, 2- hydroxyethylaminomethyl, methylsulfonyloxymethyl, 3-oxo-4- morpholinolinylmethylene, C1-6-alkoxycarbonyl, 5-methyl-l,3,4-oxadiazol-2-yl;
NR3R4, wherein R3 and R4 are each independently selected from hydrogen, C1-6-alkyl, optionally halogenated C1-6-alkylsulfonyl, C1-6-alkoxy, 2-methoxyethyl, 2-hydroxyethyl, 1-methylimidazolylsulfonyl, C1-6-acyl, cyclohexylmethyl, cyclopropanecarbonyl, aryl, optionally halogenated arylsulfonyl, furylcarbonyl, tetrahydro-2-furanylmethyl, N- carbethoxypiperidyl, or C1-6-alkyl substituted with one or more aryl, heterocyclic or heteroaryl, or
NR3R4 represent together heterocyclic systems which can be imidazole, piperidine, pyrrolidine, piperazine, morpholine, oxazepine, oxazole, thiomorpholine, 1,1- dioxidothiomorpholine, 2-(3,4-dihydro-2(lH)isoquinolinyl), (lS,4S)-2-oxa-5- azabicyclo[2.2. l]hept-5-yl, which heterocyclic systems can be optionally substituted by
C1-6-alkyl, C1-6-acyl, hydroxy, oxo, t-butoxycarbonyl;
OCONR3R4, wherein R3 and R4 are each independently selected from hydrogen, C1-6- alkyl or form together with the N-atom to which they are attached morpholinyl; R5O, wherein R5 is hydrogen, optionally halogenated C1-6-alkyl, aryl, heteroaryl, Cι-6- acyl, C1-6-alkylsulfonyl, arylcarbonyl, heteroarylcarbonyl, 2-carbomethoxyphenyl;
• B3 is hydrogen, C1-6-alkyl or dimethylaminomethyl;
or a salt, hydrate or solvate thereof; with the proviso that when:
Z is X-Y-R2, wherein X is CO and Y is a single bond, then R2 is not methyl, chloro, hydroxy, optionally halogenated C1-6-alkoxy, aryloxy, heteroaryloxy, amino, and phenylamino;
Z is X-Y-R2, wherein X is CH2 and Y is a single bond, then R2 is not methoxy.
It is preferred that:
T is selected from 5-chloro-l,3-dimethyl-lH-pyrazol-4-yl; 4-chloro-2,3,l- benzoxadiazolyl; 5-(dimethylamino)-l-naphthyl; l-methylimidazol-4-yl; 1-naphthyl; 2- naphthyl; 8-quinolinyl; thienyl substituted with one or more of (benzoylamino)methyl, bromo, chloro, 3- isoxazolyl, 2-(methylsulfanyl)-4-pyrimidinyl, 1 -methyl-5-(trifluoromethyl)pyrazol-3-yl, phenylsulfonyl, pyridyl; phenyl substituted with one or more of acetylamino, 3-acetylaminophenyl, 3- acetylphenyl, benzeneamino, l,3-benzodioxol-5-yl, 2-benzofuryl, benzylamino, 3,5- bis(trifluoromethyl)phenyl, bromo, butoxy, carboxy, chloro, 4-carboxyphenyl, 3-chloro-2- cyanophenoxy, 4-chlorophenyl, 5-chloro-2-thienyl, cyano, 3,4-dichlorophenyl, ({[4-(2- ethoxy-2-oxoethyl)-l, 3 -thiazol-2-yl] amino} carbonyl), fluoro, 5-fluoro-2-methoxyphenyl, 2- furyl, hydrogen, iodo, isopropyl, methanesulfonyl, methoxy, methyl, 4-methyl-l -piperazinyl, 4-methyl-l -piperidinyl, 4-methylsulfanylphenyl, 5-methyl-2-thienyl, 4-morpholinyl, nitro, 3- nitrophenyl, phenoxy, phenyl, n-propyl, 4-pyridyl, 3-pyridylmethylamino, 1-pyrrolidinyl, 2- thienyl, 3-thienyl, 2-thienylmethylamino, trifluoromethoxy, 4-trifluoromethoxyphenyl, trifluoromethyl; or
R1 is hydrogen or methyl; Bi and B2 are B3 or Z, provided that Bi and B2 have different meanings, wherein:
• Z is selected from l-benzothien-3-yl, 3-(2,5-dimethylfuryl), pyridinyl; thienyl optionally substituted with one or more of chloro, methylsulfonyl; phenyl optionally substituted with one or more of ethoxycarbonyl, nitro, fluoro, methyl, methoxy, acetylamino, chloro, 4-chlorophenoxy, trifluoromethyl; or is X-Y-R2, wherein • X is CH2 or CO;
• Y is CH2, CO or a single bond;
• R2 is selected from n-propyl, azido, bromo, chloro, 2-pyridinylsulfanyl, 3-oxo-4- morpholinolinylmethylene, ethoxycarbonyl, 5-methyl-l,3,4-oxadiazol-2-yl, hydroxymethyl, 2-hydroxyethylaminomethyl, methylsulfonyloxymethyl;
NR3R4, wherein R3 and R4 are each independently selected from acetyl, benzhydryl, l,3-benzodioxol-5-ylmethyl, benzyl, 3-chloro-2-methylphenylsulfonyl, cyclohexyl, cyclohexylmethyl, cyclopropanecarbonyl, ethyl, 2-furylcarbonyl, 2-furylmethyl, hydrogen, 2- hydroxyethyl, 2-(lH-indol-3-yl)ethyl, isopropyl, methoxy, 2-methoxyethyl, methyl, 4-(l- methylimidazolyl)sulfonyl, methylsulfonyl, phenyl, (lS)-phenylethyl, n-propyl, tetrahydro-2- furanylmethyl, trifluoromethylsulfonyl, N-carbethoxypiperidyl; or NR3R4 represent together 4-acetylpiperazinyl, 4-t-butoxycarbonylpiperazinyl, 2-(3,4- dihydro-2(lH)isoquinolinyl), (2R,6S)-2,6-dimethylmorpholinyl, (2R)-2,4-dimethyl-l- piperazinyl, 2-hydroxy-3-oxomorpholinyl, imidazolyl, 2-methyl-3-oxomorpholinyl, 4- methyl-2-oxopiperazinyl, 4-methylpiperazinyl, morpholinyl, (lS,4S)-2-oxa-5-aza- bicyclo[2.2.1]hept-5-yl, 2-oxoimidazolinyl, 3-oxomorpholinyl, 3-oxo-l,4-oxazepinyl, 2- oxooxazolinyl, piperazinyl; piperidinyl; pyrrolidinyl; pyrrolidonyl, thiomorpholinyl; 1,1- dioxido-thiomorpholinyl;
OCONR3R4, wherein R3 and R4 are each independently selected from ethyl, hydrogen or form together with the N-atom to which they are attached morpholinyl;
R5O, wherein R5 is acetyl, benzoyl, benzyl, ethyl, 2-fιuoroefhyl, 2-furylcarbonyl, hydrogen, isobutyryl, isopropyl, methyl, 2-carbomethoxyphenyl, methylsulfonyl, phenyl, n- propionyl, 3-pyridinyl, 2,2,2-trifluoroethyl;
• B3 is hydrogen, methyl or dimethylaminomethyl; with the proviso that when:
Z is X-Y-R2, wherein X is CO and Y is a single bond, then R2 is not chloro, hydroxy, benzyloxy, ethoxy, 2-fluoroethoxy, isopropyloxy, methoxy, 2-carbomethoxyphenoxy, phenoxy, 3-pyridinyloxy, 2,2,2-trifluoroethoxy, amino, and phenylamino; Z is X-Y-R2, wherein X is CH2 and Y is a single bond, then R2 is not methoxy.
When R1 is hydrogen or methyl, B3 is hydrogen, methyl or dimethylaminomethyl, and Z is X-Y-R2, wherein X is CO and Y is a single bond, then it is preferred that:
R2 is selected from n-propyl, azido, bromo, 2-pyridinylsulfanyl, 3-oxo-4- morpholinolinylmethylene, ethoxycarbonyl, 5-methyl-l,3,4-oxadiazol-2-yl, hydroxymethyl,
2-hydroxyethylaminomethyl, methylsulfonyloxymethyl; NR3R4, wherein: (i) R3 and R4 are either each independently selected from acetyl, benzhydryl, 1,3- benzodioxol-5-ylmethyl, benzyl, 3-chloro-2-methylphenylsulfonyl, cyclohexyl, cyclohexylmethyl, cyclopropanecarbonyl, ethyl, 2-furylcarbonyl, 2-furylmethyl, 2- hydroxyethyl, 2-(lH-indol-3-yl) ethyl, isopropyl, methoxy, 2-methoxyethyl, methyl, 4-(l-methylimidazolyl)sulfonyl, methylsulfonyl, (lS)-phenylethyl, n-propyl, tetrahydro-2-furanylmethyl, trifluoromethylsulfonyl, N-carbethoxypiperidyl; or
(ii) R3 is hydrogen and R4 is selected from acetyl, benzhydryl, l,3-benzodioxol-5- ylmethyl, benzyl, 3-chloro-2-methylphenylsulfonyl, cyclohexyl, cyclohexylmethyl, cyclopropanecarbonyl, ethyl, 2-furylcarbonyl, 2-furylmethyl, 2-hydroxyethyl, 2-(lH- indol-3-yl)ethyl, isopropyl, methoxy, 2-methoxyethyl, methyl, 4-(l- methylimidazolyl)sulfonyl, methylsulfonyl, (lS)-phenylethyl, n-propyl, tetrahydro-2- furanylmethyl, trifluoromethylsulfonyl, N-carbethoxypiperidyl; or
(iii) R3 is phenyl and R4 is selected from acetyl, benzhydryl, l,3-benzodioxol-5-ylmethyl, benzyl, 3-chloro-2-methylphenylsulfonyl, cyclohexyl, cyclohexylmethyl, cyclopropanecarbonyl, ethyl, 2-furylcarbonyl, 2-furylmethyl, 2-hydroxyethyl, 2-(lH- indol-3-yl)ethyl, isopropyl, methoxy, 2-methoxyethyl, methyl, 4-(l- methylimidazolyl)sulfonyl, methylsulfonyl, phenyl, (lS)-phenylethyl, n-propyl, tetrahydro-2-furanylmethyl, trifluoromethylsulfonyl, N-carbethoxypiperidyl; or NR3R4 represent together 4-acetylpiperazinyl, 4-t-butoxycarbonylpiperazinyl, 2-(3,4- dihydro-2(lH)isoquinolinyl), (2R,6S)-2,6-dimethylmorpholinyl, (2R)-2,4-dimethyl-l- piperazinyl, 2-hydroxy-3-oxomorpholinyl, imidazolyl, 2-methyl-3-oxomorpholinyl, 4- methyl-2-oxopiperazinyl, 4-methylpiperazinyl, morpholinyl, (lS,4S)-2-oxa-5-aza- bicyclo[2.2.1]hept-5-yl, 2-oxoimidazolinyl, 3-oxomorpholinyl, 3-oxo-l,4-oxazepinyl, 2- oxooxazolinyl, piperazinyl; piperidinyl; pyrrolidinyl; pyrrolidonyl, thiomorpholinyl; 1,1- dioxido-thiomorpholinyl; OCONR3R4, wherein R3 and R4 are each independently selected from ethyl, hydrogen or form together morphohnyl;
R5O, wherein R5 is acetyl, benzoyl, 2-furylcarbonyl, isobutyryl, methylsulfonyl, n- propionyl.
When R1 is hydrogen or methyl, B3 is hydrogen, methyl or dimethylaminomethyl, and Z is X-Y-R2, wherein X is CH2 and Y is a single bond, then it is preferred that: R2 is selected from n-propyl, azido, bromo, chloro, 2-pyridinylsulfanyl, 3-oxo-4- morpholinolinylmethylene, ethoxycarbonyl, 5-methyl-l,3,4-oxadiazol-2-yl, hydroxymethyl, 2-hydroxyethylaminomethyl, methylsulfonyloxymethyl;
NR3R4, wherein R3 and R4 are each independently selected from acetyl, benzhydryl, l,3-benzodioxol-5-ylmethyl, benzyl, 3-chloro-2-methylphenylsulfonyl, cyclohexyl, cyclohexylmethyl, cyclopropanecarbonyl, ethyl, 2-furylcarbonyl, 2-furylmethyl, hydrogen, 2- hydroxyethyl, 2-(lH-indol-3-yl)ethyl, isopropyl, methoxy, 2-methoxyethyl, methyl, 4-(l- methylimidazolyl)sulfonyl, methylsulfonyl, phenyl, (lS)-phenylethyl, n-propyl, tetrahydro-2- furanylmethyl, trifluoromethylsulfonyl, N-carbethoxypiperidyl; or
NR3R4 represent together 4-acetylpiperazinyl, 4-t-butoxycarbonylpiperazinyl, 2-(3,4- dihydro-2(lH)isoquinolinyl), (2R,6S)-2,6-dimethylmorpholinyl, (2R)-2,4-dimethyl-l- piperazinyl, 2-hydroxy-3-oxomorpholinyl, imidazolyl, 2-methyl-3-oxomorpholinyl, 4- methyl-2-oxopiperazinyl, 4-methylpiperazinyl, morpholinyl, (lS,4S)-2-oxa-5-aza- bicyclo[2.2.1]hept-5-yl, 2-oxoimidazolinyl, 3-oxomorpholinyl, 3-oxo-l,4-oxazepinyl, 2- oxooxazolinyl, piperazinyl; piperidinyl; pyrrolidinyl; pyrrolidonyl, thiomorpholinyl; 1,1- dioxido-thiomorpholinyl;
OCONR3R4, wherein R3 and R4 are each independently selected from ethyl, hydrogen or form together with the N-atom to which they are attached morpholinyl;
R5O, wherein R5 is acetyl, benzoyl, benzyl, ethyl, 2-fluoroethyl, 2-furylcarbonyl, hydrogen, isobutyryl, isopropyl, 2-carbomethoxyphenyl, methylsulfonyl, phenyl, n- propionyl, 3-pyridinyl, 2,2,2-trifluoroethyl.
The following list shows particularly preferred( compounds. They are divided into the following categories:
1) Thiophene derivatives of formula (II):
ethyl (4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)acetate (4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)acetic acid 2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)-N-methylacetamide 2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)-N-ethylacetamide 2,5-dichloro-N-(3-chloro-2,3'-bithien-4'-yl)benzenesulfonamide isopropyl (4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)acetate 3 -chloro-N- [4-(2-hydroxyethyl)thien-3 -yl] -2-methylb enzenesulfonamide 3-chloro-N-[4-(2-ethoxyethyl)thien-3-yl]-2-methylbenzenesulfonamide 2-(4- { [(3 -chloro-2-methylphenyl)sulfonyl] amino} thien-3 -yl)-N,N-diethylacetamide methyl (4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)acetate 3 -chloro-N- [4-(2-isopropoxyethyl)thien-3 -yl] -2-methylbenzenesulfonamide 3 -chloro-N- [4-(2-methoxyethyl)thien-3 -yl] -2-methylb enzenesulfonamide 2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl methanesulfonate 2-(4- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-3 -yl) acetamide 3-chloro-N-{4-[2-(2-fluoroethoxy)ethyl]thien-3-yl}-2-methylbenzenesulfonamide 3-chloro-2-methyl-N-{4-[2-(2,2,2-trifluoroethoxy)ethyl]thien-3- yl}benzenesulfonamide
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl acetate 3-chloro-2-methyl-N-[4-(2-morpholin-4-ylethyl)thien-3-yl]benzenesulfonamide N-[4-(2-bromoethyl)thien-3-yl]-3-chloro-2-methylbenzenesulfonamide 2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl morpholine-4- carboxylate • 2-(4- {[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl diethylcarbamate
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl propionate
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl 2- methylpropanoate
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl 2-furoate
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl benzoate
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)-N-methoxy-N- methylacetamide
3-chloro-N-{4-[2-(diethylamino)ethyl]thien-3-yl}-2-methylbenzenesulfonamide
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl ethylcarbamate
N-[2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl]-N- ethylacetamide
3-chloro-2-methyl-N-[4-(2-oxopentyl)thien-3-yl]benzenesulfonamide
N- {4-[2-(l , 1 -dioxidothiomorpholin-4-yl)-2-oxoethyl]thien-3-yl} -4- propylbenzenesulfonamide
2,4,6-trichloro-N-[4-(2-morpholin-4-ylethyl)thien-3-yl]benzenesulfonamide
2,4-dichloro-N-[4-(2-morpholin-4-ylethyl)thien-3-yl]benzenesulfonamide
3-chloro-2-methyl-N-{4-[2-(3-oxomorpholin-4-yl)ethyl]thien-3- yl } benzenesulfonamide
2,4-dichloro-6-methyl-N-[4-(2-morpholin-4-ylethyl)thien-3-yl]benzenesulfonamide
N- [4-(2-morpholin-4-ylethyl)thien-3 -yl] -4-propylbenzenesulfonamide
2,4-dichloro-6-methyl-N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3- yljbenzenesulfonamide
2,4,6-trichloro-N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3-yl]benzenesulfonamide
N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3-yl]-4-propylbenzenesulfonamide
N-[4-(2-oxo-2-thiomorpholin-4-ylethyl)thien-3-yl]-l,r-biphenyl-4-sulfonamide
N-[4-(2-oxo-2-thiomorpholin-4-ylethyl)thien-3-yl]-4-propylbenzenesulfonamide
2,4-dichloro-6-methyl-N-[4-(2-oxo-2-thiomorpholin-4-ylethyl)thien-3- yl]benzenesulfonamide
N-[4-(2-oxo-2-piperidin- 1 -ylethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N-[4-(2-oxo-2-piperidin-l-ylethyl)thien-3-yl]-4-propylbenzenesulfonamide
2,4-dichloro-6-methyl-N-[4-(2-oxo-2-piperidin-l-ylethyl)thien-3- yl]benzenesulfonamide
2,4,6-trichloro-N-[4-(2-oxo-2-piperidin-l-ylethyl)thien-3-yl]benzenesulfonamide
N-(4-phenylthien-3-yl)-4-propylbenzenesulfonamide ethyl (4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)(oxo)acetate
3-chloro-2-methyl-N-(4-phenylthien-3-yl)benzenesulfonamide
3-chloro-N-[4-(4-fluoro-3-methylphenyl)thien-3-yl]-2-methylbenzenesulfonamide
2,4,6-trichloro-N-(4-phenylthien-3-yl)benzenesulfonamide
N-(4-phenylthien-3 -yl)- 1 , 1 '-biphenyl-4-sulfonamide
2,4-dichloro-6-methyl-N-(4-phenylthien-3-yl)benzenesulfonamide
2- {4-[(l , 1 '-biphenyl-4-ylsulfonyl)amino]thien-3-yl} -N-ethyl-N-methylacetamide
N-ethyl-N-methyl-2-(4-{[(4-propylphenyl)sulfonyl]amino}thien-3-yl)acetamide
2-(4-{[(2,4-dichloro-6-methylphenyl)sulfonyl]amino}thien-3-yl)-N-ethyl-N- methylacetamide
N-ethyl-N-methyl-2-(4-{[(2,4,6-trichlorophenyl)sulfonyl]amino}thien-3-yl)acetamide
2,4,6-trichloro-N- [4-(2-oxo-2-thiomorpholin-4-ylethyl)thien-3 - yl]benzenesulfonamide
2- {4-[( 1 , 1 '-biphenyl-4-ylsulfonyl)amino]thien-3 -yl} -N-isopropyl-N-methylacetamide
2- {4-[(l , 1 '-biphenyl-4-ylsulfonyl)amino]thien-3-yl} -N,N-diethylacetamide
N,N-diethyl-2-(4-{[(4-propylphenyl)sulfonyl]amino}thien-3-yl)acetamide
2-(4- { [(2,4-dichloro-6-methylphenyl)sulfonyl] amino } thien-3 -yl)-N,N- diethylacetamide
N,N-diethyl-2-(4-{[(2,4,6-trichlorophenyl)sulfonyl]amino}thien-3-yl)acetamide
2- {4-[(l , 1 '-biphenyl-4-ylsulfonyl)amino]thien-3-yl} -N,N-diisopropylacetamide
N,N-diisopropyl-2-(4-{[(4-propylphenyl)sulfonyl]amino}thien-3-yl)acetamide
2-(4-{[(2,4-dichloro-6-methylphenyl)sulfonyl]amino}thien-3-yl)-N,N- diisopropylacetamide
N,N-diisopropyl-2-(4-{[(2,4,6-trichlorophenyl)sulfonyl]amino}thien-3-yl)acetamide
N-[4-(4-{[(4-propylphenyl)sulfonyl]amino}thien-3-yl)ρhenyl]acetamide
4-propyl-N-(4-pyridin-3-ylthien-3-yl)benzenesulfonamide
N-[4-(2-chloro-5-nitrophenyl)thien-3-yl]-4-propylbenzenesulfonamide
N-[4-(2-chlorophenyl)thien-3 -yl] -4-propylbenzenesulfonamide
3-chloro-N-[4-(2-chloro-5-nitrophenyl)thien-3-yl]-2-methylbenzenesulfonamide
3-chloro-N-(5-chloro-2,3'-bithien-4'-yl)-2-methylbenzenesulfonamide
3-chloro-N-[4-(2-chlorophenyl)thien-3-yl]-2-methylbenzenesulfonamide
N-[4-(4-{[(2,4,6-trichlorophenyl)sulfonyl]amino}thien-3-yl)phenyl]acetamide
2,4,6-trichloro-N-(4-pyridin-3-ylthien-3-yl)benzenesulfonamide
2,4,6-trichloro-N-[4-(2-chloro-5-nitrophenyl)thien-3-yl]benzenesulfonamide
2,4,6-trichloro-N-(5-chloro-2,3'-bithien-4'-yl)benzenesulfonamide
2,4,6-trichloro-N-[4-(2-chlorophenyl)thien-3-yl]benzenesulfonamide
N-(4-{4-[(l,r-biphenyl-4-ylsulfonyl)amino]thien-3-yl}phenyl)acetamide
N-(4-pyridin-3-ylthien-3-yl)- 1 , 1 '-biphenyl-4-sulfonamide
N-[4-(2-chloro-5-nitrophenyl)thien-3-yl]-l, -biphenyl-4-sulfonamide
N- [4-(2-chlorophenyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N-[4-(4- { [(2,4-dichloro-6-methylphenyl)sulfonyl] amino } thien-3-yl)phenyl] acetamide
2,4-dichloro-6-methyl-N-(4-pyridm-3-ylthien-3-yl)benzenesulfonamide
2,4-dichloro-N-[4-(2-chloro-5-nitrophenyl)thien-3-yl]-6-methylbenzenesulfonamide
2,4-dichloro-N-(5-chloro-2,3'-bithien-4'-yl)-6-methylbenzenesulfonamide •
2-(4- { [(3-chloro-2-methylphenyl)sulfonyl] amino } thien-3 -yl)-N,N-dipropylacetamide
3-chloro-2-methyl-N-[4-(2-oxo-2-piperazin-l-ylethyl)thien-3-yl]benzenesulfonamide
2,4-dichloro-N- [4-(2, 5 -dimethyl-3 -furyl)thien-3 -yl] -6-methylbenzenesulfonamide
N-(3-chloro-2,3'-bithien-4'-yl)-4-proρylbenzenesulfonamide
3-chloro-N-(3-chloro-2,3'-bithien-4'-yl)-2-methylbenzenesulfonamide
2,4,6-trichloro-N-(3-chloro-2,3'-bithien-4'-yl)benzenesulfonamide
2,4-dichloro-N-(3-chloro-2,3'-bithien-4'-yl)-6-methylbenzenesulfonamide
2,4-dichloro-N-[4-(2-chlorophenyl)thien-3-yl]-6-methylbenzenesulfonamide
4-bromo-2-methyl-N- [4-(2-morpholin-4-yl-2-oxoethyl)thien-3 - yljbenzenesulfonamide
N-[4-(2-morρholin-4-yl-2-oxoethyl)thien-3-yl]-2,4- bis(trifluoromethyl)benzenesulfonamide
2-methyl-N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3-yl]-4-
(trifluoromethoxy)benzenesulfonamide
N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3-yl]-4-phenoxybenzenesulfonamide
4-chloro-2,6-dimethyl-N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3- yfjbenzenesulfonamide
2,4-dichloro-N-[4-(2-morpholin-4-yl-2-oxoethyl)thien-3-yl]benzenesulfonamide tert-butyl 4-[(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3- yl)acetyl]piperazine- 1 -carboxylate
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)-N,N-dimethylacetamide
3-chloro-2-methyl-N-{4-[2-(pyridin-3-yloxy)ethyl]thien-3-yl}benzenesulfonamide
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)-N-isopropyl-N- methylacetamide
2-(4- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-3-yl)-N-ethyl-N- methylacetamide
3-chloro-2-methyl-N-[4-(2-oxo-2-thiomorpholin-4-ylethyl)thien-3- yl]benzenesulfonamide
3-chloro-2-methyl-N-[4-(2-mo holin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)-N,N- diisopropylacetamide
3 -chloro-2-methyl-N- [4-(2-oxo-2-pyrrolidin- 1 -ylethyl)thien-3 -yl]benzenesulfonamide
3-chloro-2-methyl-N-[4-(2-oxo-2-piperidin-l-ylethyl)thien-3-yl]benzenesulfonamide
3-chloro-2-methyl-N-[4-(morpholin-4-ylmethyl)thien-3-yl]benzenesulfonamide
3 -chloro-N- {4- [2-( 1 H-imidazol- 1 -yl)ethyl] thien-3 -yl } -2-methylbenzenesulfonamide
2,4,5-trichloro-N-(3-chloro-2,3'-bithien-4'-yl)benzenesulfonamide
2,3,4-trichloro-N-(3-chloro-2,3'-bithien-4'-yl)benzenesulfonamide
2,3,4-trichloro-N-[4-(2-chlorophenyl)thien-3-yl]benzenesulfonamide
N-[4-(4-{[(4-bromo-2,5-difluorophenyl)sulfonyl]amino}thien-3-yl)phenyl]acetamide
4-bromo-N-(3-chloro-2,3'-bithien-4'-yl)-2,5-difluorobenzenesulfonamide
4,5-dichloro-N-[4-(2-chlorophenyl)thien-3-yl]thiophene-2-sulfonamide
N-[4-(4-{[(2,4,5-trichlorophenyl)sulfonyl]amino}thien-3-yl)phenyl]acetamide
4-bromo-5-chloro-N-(3-chloro-2,3'-bithien-4'-yl)thiophene-2-sulfonamide
3-bromo-5-chloro-N-[4-(2-chlorophenyl)thien-3-yl]thiophene-2-sulfonamide
N-[4-(4-{[(2,6-dichlorophenyl)sulfonyl]amino}thien-3-yl)phenyl]acetamide
2,6-dichloro-N-(3-chloro-2,3'-bithien-4'-yl)benzenesulfonamide
N-[2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl]acetamide
3-chloro-2-methyl-N-(4-{2-[(methylsulfonyl)amino]ethyl}thien-3- yl)benzenesulfonamide
3-chloro-2-methyl-N-{4-[2-(3-oxo-l,4-oxazepan-4-yl)ethyl]thien-3- yl } benzenesulfonamide
3-chloro-2-methyl-N- {4-[2-(2-oxopyrrolidin-l -yl)ethyl]thien-3- yl}benzenesulfonamide
2,3,4-trichloro-N-{4-[2,6-dichloro-4-(trifluoromethyl)phenyl]thien-3- yl}benzenesulfonamide
N-[4-(2-chloro-6-fluorophenyl)thien-3-yl]-4-propylbenzenesulfonamide
4-bromo-N-{4-[2,6-dichloro-4-(trifluoromethyl)phenyl]thien-3-yl}-2,5- difluorobenzenesulfonamide
4,5-dichloro-N-[4-(2-chloro-6-fluorophenyl)thien-3-yl]thiophene-2-sulfonamide
4-bromo-5-chloro-N-{4-[2,6-dichloro-4-(trifluoromethyl)phenyl]thien-3- yl} thiophene-2-sulfonamide
2,4-dichloro-N-[4-(2-chloro-6-fluorophenyl)thien-3-yl]-6-methylbenzenesulfonamide
4-bromo-N-[4-(2-chloro-6-fluorophenyl)thien-3-yl]-2-methylbenzenesulfonamide
3-chloro-2-methyl-N-(4-{2-[methyl(methylsulfonyl)amino]ethyl}thien-3- yl)benzenesulfonamide
N-[2-(4-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-3-yl)ethyl]-N- methylcyclopropanecarboxamide
3-chloro-2-methyl-N- {4-[2-(4-methyl-2-oxopiperazin- 1 -yl)ethyl]thien-3- yl}benzenesulfonamide
3 -chl oro-2-methyl-N- [4-(2- { [(trifluoromethyl)sulfonyl] amino } ethyl)thien-3 - yl]benzenesulfonamide
N-[4-(4-{[(4-bromo-5-chlorothien-2-yl)sulfonyl]amino}thien-3-yl)phenyl]acetamide 2,4-dichloro-N-{4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}benzenesulfonamide 2,4-dichloro-6-methyl-N-{4-[2-(3-oxomorpholin-4-yl)ethyl]thien-3- yl}benzenesulfonamide
2,4,6-trichloro-N-{4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}benzenesulfonamide 4-(2-furyl)-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]benzenesulfonamide 5,-fluoro-2'-methoxy-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-l,r-biphenyl-4- sulfonamide
4-(5-methylthien-2-yl)-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
3'-acetyl-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-l,r-biphenyl-4-sulfonamide N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4'-(trifluoromethoxy)-l, -biphenyl-4- sulfonamide
3',4'-dichloro-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl] -1,1 '-biphenyl-4- sulfonamide
4-(l,3-benzodioxol-5-yl)-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
4-(5-chlorothien-2-yl)-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-pyridin-4-ylbenzenesulfonamide N-[4'-( {[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl] amino} sulfonyl)- 1 , 1 '-biphenyl-3- yl] acetamide
N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-thien-3-ylbenzenesulfonamide N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-thien-2-ylbenzenesulfonamide 4'-(methylthio)-N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3 -yl] -1,1 '-biρhenyl-4- sulfonamide
N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-3,,5,-bis(trifluoromethyl)-l,r- biphenyl-4-sulfonamide
• 4'-chloro-N- [4-(2-moφholin-4-yl-2-oxoethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
• N-[4-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-3'-nitro-l,r-biphenyl-4-sulfonamide
• 3-chloro-2-methyl-N-[4-(2- {methyl[(trifluoromethyl)sulfonyl] amino} ethyl)thien-3- yl]benzenesulfonamide • N- [2-(4- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-3 -yl) ethyl] - 1 -methyl- 1 H- imidazole-4-sulfonamide
• 3-chloro-N-{4-[2-(2-hydroxy-3-oxomoφholin-4-yl)ethyl]thien-3-yl}-2- methylbenzenesulfonamide
• 4,5-dichloro-N-{4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}thiophene-2- sulfonamide
• N- {4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl} -4-phenoxybenzenesulfonamide
• 3-fluoro-N-{4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}benzenesulfonamide
• N-{4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}-5-pyridin-2-ylthiophene-2- sulfonamide • N-{2-chloro-4-[({4-[2-(3-oxomoφholin-4-yl)ethyl]thien-3- yl} amino)sulfonyl]ρhenyl} acetamide
2) Thiophene derivatives of formula (III):
• ethyl (3- {[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)acetate
• (3- {[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)acetic acid
• 2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N-methylacetamide
• 2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N-ethylacetamide • 2,5-dichloro-N-(3'-chloro-2,2'-bithien-3-yl)benzenesulfonamide
isopropyl (3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)acetate
3-chloro-N-[2-(2-hydroxyethyl)thien-3-yl]-2-methylbenzenesulfonamide
3-chloro-N-[2-(2-ethoxyethyl)thien-3-yl]-2-methylbenzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N,N-diethylacetamide methyl (3- {[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)acetate (Example
2)
3-chloro-N-[2-(2-isopropoxyethyl)thien-3-yl]-2-methylbenzenesulfonamide
3-chloro-N-[2-(2-methoxyethyl)thien-3-yl]-2-methylbenzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl methanesulfonate
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)acetamide
3-chloro-N-{2-[2-(2-fluoroethoxy)ethyl]thien-3-yl}-2-methylbenzenesulfonamide
3-chloro-2-methyl-N-{2-[2-(2,2,2-trifluoroethoxy)ethyl]thien-3- yl} benzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl acetate
3-chloro-2-methyl-N-[2-(2-moφholin-4-ylethyl)thien-3-yl]benzenesulfonamide
N-[2-(2-bromoethyl)thien-3-yl]-3-chloro-2-methylbenzenesulfonamide
2-(3- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-2-yl)ethyl moφholine-4- carboxylate
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl diethylcarbamate
2-(3- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-2-yl) ethyl propionate
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl 2- methylpropanoate
2-(3- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-2-yl)ethyl 2-furoate
2-(3- { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-2-yl)ethyl benzoate
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N-methoxy-N- methylacetamide
3-chloro-N-{2-[2-(diethylamino)ethyl]thien-3-yl}-2-methylbenzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl ethylcarbamate
N- [2-(3 - { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-2-yl) ethyl] -N- ethylacetamide
3-chloro-2-methyl-N-[2-(2-oxopentyl)thien-3-yl]benzenesulfonamide
N- {2-[2-(l , 1 -dioxidothiomoφholin-4-yl)-2-oxoethyl]thien-3-yl} -4- propylbenzenesulfonamide
2,4,6-trichloro-N-[2-(2-moφholin-4-ylethyl)thien-3-yl]benzenesulfonamide
2,4-dichloro-N-[2-(2-moφholin-4-ylethyl)thien-3-yl]benzenesulfonamide
3-chloro-2-methyl-N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3- yl } benzenesulfonamide
2,4-dichloro-6-methyl-N-[2-(2-moφholin-4-ylethyl)thien-3-yl]benzenesulfonamide
N-[2-(2-moφholin-4-ylethyl)thien-3-yl]-4-propylbenzenesulfonamide
2,4-dichloro-6-methyl-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
2,4,6-trichloro-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]benzenesulfonamide
N- [2-(2-moφholin-4-yl-2-oxoethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-propylbenzenesulfonamide
N- [2-(2-oxo-2-thiomoφholin-4-ylethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N-[2-(2-oxo-2-thiomoφholin-4-ylethyl)thien-3-yl]-4-propylbenzenesulfonamide
2,4-dichloro-6-methyl-N-[2-(2-oxo-2-thiomoφholin-4-ylethyl)thien-3- yl]benzenesulfonamide
N-[2-(2-oxo-2-piperidin- 1 -ylethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N-[2-(2-oxo-2-piperidin-l-ylethyl)thien-3-yl]-4-propylbenzenesulfonamide
2,4-dichloro-6-methyl-N-[2-(2-oxo-2-piperidin-l-ylethyl)thien-3- yl]benzenesulfonamide
2,4,6-trichloro-N-[2-(2-oxo-2-piperidin-l-ylethyl)thien-3-yl]benzenesulfonamide
N-(2-phenylthien-3-yl)-4-propylbenzenesulfonamide ethyl (3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)(oxo)acetate
3-chloro-2-methyl-N-(2-phenylthien-3-yl)benzenesulfonamide
3-chloro-N-[2-(4-fluoro-3-methylphenyl)thien-3-yl]-2-methylbenzenesulfonamide
2,4,6-trichloro-N-(2-phenylthien-3-yl)benzenesulfonamide
N-(2-phenylthien-3-yl)-l, -biphenyl-4-sulfonamide
2,4-dichloro-6-methyl-N-(2-phenylthien-3-yl)benzenesulfonamide
2- {3-[(l , 1 '-biphenyl-4-ylsulfonyl)amino]thien-2-yl} -N-ethyl-N-methylacetamide
N-ethyl-N-methyl-2-(3-{[(4-propylphenyl)sulfonyl]amino}thien-2-yl)acetamide
2-(3-{[(2,4-dichloro-6-methylphenyl)sulfonyl]amino}thien-2-yl)-N-ethyl-N- methylacetamide
N-ethyl-N-methyl-2-(3-{[(2,4,6-trichlorophenyl)sulfonyl]amino}thien-2-yl)acetamide
2,4,6-trichloro-N-[2-(2-oxo-2-thiomoφholin-4-ylethyl)thien-3- yl]benzenesulfonamide
2- {3-[( 1 , 1 '-biphenyl-4-ylsulfonyl)amino]thien-2-yl} -N-isopropyl-N-methylacetamide
2-{3-[(l, -biphenyl-4-ylsulfonyl)amino]thien-2-yl}-N,N-diethylacetamide
N,N-diethyl-2-(3-{[(4-propylphenyl)sulfonyl]amino}thien-2-yl)acetamide
2-(3-{[(2,4-dichloro-6-methylphenyl)sulfonyl]amino}thien-2-yl)-N,N- diethylacetamide
N,N-diethyl-2-(3-{[(2,4,6-trichlorophenyl)sulfonyl]amino}thien-2-yl)acetamide
2- {3-[(l , 1 '-biphenyl-4-ylsulfonyl)amino]thien-2-yl} -N,N-diisopropylacetamide
N,N-diisopropyl-2-(3-{[(4-propylphenyl)sulfonyl]amino}thien-2-yl)acetamide
2-(3-{[(2,4-dichloro-6-methylphenyl)sulfonyl]amino}thien-2-yl)-N,N- diisopropylacetamide
N,N-diisopropyl-2-(3 - { [(2,4,6-trichlorophenyl)sulfonyl] amino } thien-2-yl) acetamide
N- [4-(3 - { [(4-propylphenyl)sulfonyl] amino} thien-2-yl)phenyl] acetamide
4-propyl-N-(2-pyridin-3-ylthien-3-yl)benzenesulfonamide
N-[2-(2-chloro-5-nitrophenyl)thien-3-yl]-4-propylbenzenesulfonamide
N-[2-(2-chlorophenyl)thien-3-yl]-4-propylbenzenesulfonamide
3-chloro-N-[2-(2-chloro-5-nitrophenyl)thien-3-yl]-2-methylbenzenesulfonamide
3-chloro-N-(5'-chloro-2,2'-bithien-3-yl)-2-methylbenzenesulfonamide
3-chloro-N-[2-(2-chlorophenyl)thien-3-yl]-2-methylbenzenesulfonamide
N-[4-(3-{[(2,4,6-trichloroρhenyl)sulfonyl]amino}thien-2-yl)phenyl]acetamide
2,4,6-trichloro-N-(2-pyridin-3-ylthien-3-yl)benzenesulfonamide
2,4,6-trichloro-N-[2-(2-chloro-5-nitrophenyl)thien-3-yl]benzenesulfonamide
2,4,6-trichloro-N-(5'-chloro-2,2'-bithien-3-yl)benzenesulfonamide
2,4,6-trichloro-N-[2-(2-chlorophenyl)thien-3-yl]benzenesulfonamide
N-(4- {3-[(l , 1 '-biphenyl-4-ylsulfonyl)amino]thien-2-yl}phenyl)acetamide
N-(2-pyridin-3 -ylthien-3 -yl)- 1 , 1 '-biphenyl-4-sulfonamide
N-[2-(2-chloro-5-nitrophenyl)thien-3-yl]-l,r-biphenyl-4-sulfonamide
N-[2-(2-chlorophenyl)thien-3-yl]-l, -biphenyl-4-sulfonamide
N-[4-(3-{[(2,4-dichloro-6-methylphenyl)sulfonyl]amino}thien-2-yl)phenyl]acetamide
2,4-dichloro-6-methyl-N-(2-pyridin-3-ylthien-3-yl)benzenesulfonamide
2,4-dichloro-N-[2-(2-chloro-5-nitrophenyl)thien-3-yl]-6-methylbenzenesulfonamide
2,4-dichloro-N-(5'-chloro-2,2'-bithien-3-yl)-6-methylbenzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N,N-dipropylacetamide
3-chloro-2-methyl-N-[2-(2-oxo-2-piperazin-l-ylethyl)thien-3-yl]benzenesulfonamide
2,4-dichloro-N-[2-(2,5-dimethyl-3-furyl)thien-3-yl]-6-methylbenzenesulfonamide
N-(3'-chloro-2,2'-bithien-3-yl)-4-propylbenzenesulfonamide
3-chloro-N-(3'-chloro-2,2'-bithien-3-yl)-2-methylbenzenesulfonamide
2,4,6-trichloro-N-(3'-chloro-2,2'-bithien-3-yl)benzenesulfonamide
2,4-dichloro-N-(3'-chloro-2,2'-bithien-3-yl)-6-methylbenzenesulfonamide
2,4-dichloro-N-[2-(2-chlorophenyl)thien-3-yl]-6-methylbenzenesulfonamide
4-bromo-2-methyl-N- [2-(2-moφholin-4-yl-2-oxoethyl)thien-3 - yl]benzenesulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-2,4- bis(trifluoromethyl)benzenesulfonamide
2-methyl-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-
(trifluoromethoxy)benzenesulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-phenoxybenzenesulfonamide
4-chloro-2,6-dimethyl-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3- yljbenzenesulfonamide
2,4-dichloro-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]benzenesulfonamide tert-butyl 4-[(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2- yl)acetyl]piperazine- 1 -carboxylate
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N,N-dimethylacetamide
3-chloro-2-methyl-N-{2-[2-(pyridin-3-yloxy)ethyl]thien-3-yl}benzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N-isopropyl-N- methylacetamide
2-(3-{[(3-chloro-2-methylρhenyl)sulfonyl]amino}thien-2-yl)-N-ethyl-N- methylacetamide
3-chloro-2-methyl-N-[2-(2-oxo-2-thiomoφholin-4-ylethyl)thien-3- yl]benzenesulfonamide
3-chloro-2-methyl-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)-N,N- diisopropylacetamide
3-chloro-2-methyl-N-[2-(2-oxo-2-pyrrolidin-l-ylethyl)thien-3-yl]benzenesulfonamide
3-chloro-2-methyl-N-[2-(2-oxo-2-piperidin-l-ylethyl)thien-3-yl]benzenesulfonamide
3-chloro-2-methyl-N-[2-(moφholin-4-ylmethyl)thien-3-yl]benzenesulfonamide
3-chloro-N-{2-[2-(lH-imidazol-l-yl)ethyl]thien-3-yl}-2-methylbenzenesulfonamide
2,4,5-trichloro-N-(3'-chloro-2,2'-bithien-3-yl)benzenesulfonamide
2,3,4-trichloro-N-(3'-chloro-2,2'-bithien-3-yl)benzenesulfonamide
2,3,4-trichloro-N-[2-(2-chlorophenyl)thien-3-yl]benzenesulfonamide
N- [4-(3 - { [(4-bromo-2, 5 -difluorophenyl)sulfonyl] amino } thien-2-yl)phenyl] acetamide
4-bromo-N-(3'-chloro-2,2'-bithien-3-yl)-2,5-difluorobenzenesulfonamide
4,5-dichloro-N-[2-(2-chlorophenyl)thien-3-yl]thiophene-2-sulfonamide
N-[4-(3 - { [(2,4,5-trichlorophenyl)sulfonyl] amino } thien-2-yl)phenyl] acetamide
4-bromo-5-chloro-N-(3'-chloro-2,2'-bithien-3-yl)thiophene-2-sulfonamide
3-bromo-5-chloro-N-[2-(2-chlorophenyl)thien-3-yl]thiophene-2-sulfonamide
N-[4-(3- { [(2,6-dichlorophenyl)sulfonyl] amino } thien-2-yl)phenyl] acetamide
2,6-dichloro-N-(3'-chloro-2,2'-bithien-3-yl)benzenesulfonamide
N-[2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl]acetamide
3-chloro-2-methyl-N-(2-{2-[(methylsulfonyl)amino]ethyl}thien-3- yl)benzenesulfonamide
3-chloro-2-methyl-N-{2-[2-(3-oxo-l,4-oxazepan-4-yl)ethyl]thien-3- yl}benzenesulfonamide
3-chloro-2-methyl-N-{2-[2-(2-oxopyrrolidin-l-yl)ethyl]thien-3- yl } b enzenesulfonamide
2,3,4-trichloro-N-{2-[2,6-dichloro-4-(trifluoromethyl)phenyl]thien-3- yl}benzenesulfonamide
N-[2-(2-chloro-6-fluorophenyl)thien-3-yl]-4-propylbenzenesulfonamide
4-bromo-N-{2-[2,6-dichloro-4-(trifluoromethyl)phenyl]thien-3-yl}-2,5- difluorobenzenesulfonamide
4,5-dichloro-N-[2-(2-chloro-6-fluorophenyl)thien-3-yl]thiophene-2-sulfonamide
4-bromo-5-chloro-N-{2-[2,6-dichloro-4-(trifluoromethyl)phenyl]thien-3- yl}thiophene-2-sulfonamide
2,4-dichloro-N-[2-(2-chloro-6-fluorophenyl)thien-3-yl]-6-methylbenzenesulfonamide
4-bromo-N-[2-(2-chloro-6-fluorophenyl)thien-3-yl]-2-methylbenzenesulfonamide
3-chloro-2-methyl-N-(2-{2-[methyl(methylsulfonyl)amino]ethyl}thien-3- yl)benzenesulfonamide
N- [2-(3 - { [(3 -chloro-2-methylphenyl)sulfonyl] amino } thien-2-yl) ethyl] -N- mefhylcyclopropanecarboxamide
3-chloro-2-methyl-N-{2-[2-(4-methyl-2-oxopiperazin-l-yl)ethyl]thien-3- yl } benzenesulfonamide
3-chloro-2-methyl-N-[2-(2-{[(trifluoromethyl)sulfonyl]amino}ethyl)thien-3- yl]benzenesulfonamide
N-[4-(3-{[(4-bromo-5-chlorothien-2-yl)sulfonyl]amino}thien-2-yl)phenyl]acetamide
2,4-dichloro-N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}benzenesulfonamide
2,4-dichloro-6-methyl-N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3- yl } b enzenesulfonamide
2,4,6-trichloro-N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}benzenesulfonamide
4-(2-furyl)-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]benzenesulfonamide
5'-fluoro-2'-methoxy-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-l,r-biphenyl-4- sulfonamide
4-(5-methylthien-2-yl)-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
3 '-acetyl-N- [2-(2-moφholin-4-yl-2-oxoethyl)fhien-3-yl] -1,1 '-biphenyl-4-sulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4'-(trifluoromethoxy)-l,r-biphenyl-4- sulfonamide
3',4'-dichloro-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl] -1,1 '-biphenyl-4- sulfonamide
4-(l,3-benzodioxol-5-yl)-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
4-(5-chlorothien-2-yl)-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3- yl]benzenesulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-pyridin-4-ylbenzenesulfonamide
N-[4'-( { [2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl] amino } sulfonyl)- 1 , 1 '-biphenyl-3- yl] acetamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-thien-3-ylbenzenesulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-4-thien-2-ylbenzenesulfonamide
4'-(methylthio)-N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl]-l,l'-biphenyl-4- sulfonamide
N-[2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl] -3 ',5 '-bis(trifluoromethyl)- 1,1'- biphenyl-4-sulfonamide
4'-chloro-N- [2-(2-moφholin-4-yl-2-oxoethyl)thien-3 -yl] -1,1 '-biphenyl-4-sulfonamide
N- [2-(2-moφholin-4-yl-2-oxoethyl)thien-3-yl] -3 '-nitro- 1 , 1 '-biphenyl-4-sulfonamide
3-chloro-2-methyl-N-[2-(2-{methyl[(trifluoromethyl)sulfonyl]amino}ethyl)thien-3- yl]benzenesulfonamide
N-[2-(3-{[(3-chloro-2-methylphenyl)sulfonyl]amino}thien-2-yl)ethyl]-l-methyl-lH- imidazole-4-sulfonamide
3-chloro-N-{2-[2-(2-hydroxy-3-oxomoφholin-4-yl)ethyl]thien-3-yl}-2- methylbenzenesulfonamide
4,5-dichloro-N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}thiophene-2- sulfonamide
N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}-4-phenoxybenzenesulfonamide
3-fluoro-N-{2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl}benzenesulfonamide
• N- {2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3-yl} -5-pyridin-2-ylthiophene-2- sulfonamide
• N- {2-chloro-4-[( {2-[2-(3-oxomoφholin-4-yl)ethyl]thien-3- yl} amino)sulfonyl]phenyl} acetamide • methyl (3-{[(5-fluoro-2-methylphenyl)sulfonyl]amino}thien-2-yl)acetate (Example 3)
• methyl (3-{[(3-cyanophenyl)sulfonyl]amino}thien-2-yl)acetate (Example 4)
• methyl (3-{[(4-butoxyphenyl)sulfonyl]amino}thien-2-yl)acetate (Example 5)
• methyl (3- {[(2-methylsulfanylpyrimidin-4-ylthiophene)sulfonyl]amino}thien-2- yl)acetate (Example 6) • methyl (3-{[(l-methylimidazol-4-yl)sulfonyl]amino}thien-2-yl)acetate (Example 7)
• methyl (3-{[(4-methylphenyl)sulfonyl]amino}thien-2-yl)acetate (Example 8) Another object of the present invention is a compound as described above for medical use.
Another object of the present invention is a method for the treatment or prevention of diabetes, syndrome X, obesity, glaucoma, hyperlipidemia, hyperglycemia, hyperinsulinemia, hypertension, osteoporosis, dementia, depression, virus diseases or inflammatory disorders without causing hypoglycemia and to achieve immuno-modulation, preferably tuberculosis, lepra and psoriasis, said method comprising administering to a mammal, including a human, in need of such treatment (e.g., identified as in need thereof) an effective amount of a compound of formula (I) or a composition having a compound of formula (I) in it: wherein
T is an aryl ring or heteroaryl ring, optionally independently substituted by [R]n, wherein n is an integer 0-5, and R is hydrogen, aryl, heteroaryl, a heterocyclic ring, optionally halogenated Cι-6-alkyl, optionally halogenated C1-6-alkoxy, C1-6-alkylsulfonyl, carboxy, cyano, nitro, halogen, amine which is optionally mono- or di-substituted, amide which is optionally mono- or di-substituted, aryloxy, arylsulfonyl, arylamino, wherein aryl, heteroaryl and aryloxy residues and heterocyclic rings can further be optionally substituted in one or more positions independently of each other by C1-6-acyl, C1-6-alkylthio, cyano, nitro, hydrogen, halogen, optionally halogenated C1-6-alkyl, optionally halogenated C1-6-alkoxy, amide which is optionally mono- or di-substituted, (benzoylamino)methyl, carboxy, 2- thienylmethylamino or ({[4-(2-ethoxy-2-oxoethyl)-l,3-thiazol-2-yl]amino}carbonyl);
R1 is hydrogen or C1-6-alkyl;
Bi and B are B3 or Z, provided that Bi and B2 have different meanings, wherein:
• Z is selected from an aryl ring or heteroaryl ring, which can further be optionally substituted in one or more positions independently of each other by hydrogen, C1-6-alkyl, halogenated C1-6-alkyl, halogen, C1-6-alkoxy, nitro, C1-6-alkoxycarbonyl, C1-6- alkylsulfonyl, acetylamino or aryloxy, wherein the aryloxy can further be optionally substituted in one or more positions independently of each other by hydrogen and halogen; or is X-Y-R2, wherein
• X is CH2 or CO; • Y is CH2, CO or a single bond;
• R2 is selected from C1-6-alkyl, azido, arylthio, heteroarylthio, halogen, hydroxymethyl, 2- hydroxyethylaminomethyl, methylsulfonyloxymethyl, 3-oxo-4- moφholinolinylmethylene, C1-6-alkoxycarbonyl, 5-methyl-l,3,4-oxadiazol-2-yl; NR3R4, wherein R3 and R4 are each independently selected from hydrogen, C1-6-alkyl, optionally halogenated C1-6-alkylsulfonyl, C1-6-alkoxy, 2-methoxyethyl, 2-hydroxyethyl,
1-methylimidazolylsulfonyl, C1-6-acyl, cyclohexylmethyl, cyclopropanecarbonyl, aryl, optionally halogenated arylsulfonyl, furylcarbonyl, tetrahydro-2-furanylmethyl, N- carbethoxypiperidyl, or C1-6-alkyl substituted with one or more aryl, heterocyclic or heteroaryl, or NR3R4 represent together heterocyclic systems which can be imidazole, piperidine, pyrrolidine, piperazine, moφholine, oxazepine, oxazole, thiomoφholine, 1,1- dioxidothiomoφholine, 2-(3,4-dihydro-2(lH)isoquinolinyl), (1 S,4S)-2-oxa-5- azabicyclo[2.2.1]hept-5-yl, which heterocyclic systems can be optionally substituted by C1-6-alkyl, C1-6-acyl, hydroxy, oxo, t-butoxycarbonyl; OCONR3R4, wherein R3 and R4 are each independently selected from hydrogen, C1-6- alkyl or form together with the N-atom to which they are attached moφholinyl; R50, wherein R5 is hydrogen, optionally halogenated Q-6-alkyl, aryl, heteroaryl, C1-6- acyl, C1-6-alkylsulfonyl, arylcarbonyl, heteroarylcarbonyl, 2-carbomethoxyphenyl;
• B3 is hydrogen, C1-6-alkyl or dimethylaminomethyl; or a salt, hydrate or solvate thereof.
In another aspect, this invention features a method for inhfwting a human 11-β- hydroxysteroid dehydrogenase type 1 enzyme. The method includes administering to a subject (e.g., mammal, human, or animal) in need thereof (e.g., identified as in need thereof) an effective amount of a compound of any of the formulae delineated herein or a composition comprising any of the formulae herein.
The present invention also features a method for treating 11-β-hydroxysteroid dehydrogenase type 1 enzyme-mediated disorders. The method includes administering to a subject (e.g., mammal, human, or animal) in need thereof (e.g., identified as in need thereof) an effective amount of a compound of any of the formulae delineated herein or a composition comprising any of the formulae delineated herein. The 11-β-hydroxy steroid dehydrogenase type 1 enzyme-mediated disorder is any disorder or symptom wherein the 11-β- hydroxysteroid dehydrogenase type 1 enzyme is involved in the process or presentation of the disorder or the symptom. The 11-β-hydroxysteroid dehydrogenase type 1 enzyme- mediated disorders include, but are not limited to, diabetes, syndrome X, obesity, glaucoma, hyperlipidemia, hyperglycemia, hyperinsulinemia, hypertension, osteoporosis, dementia, depression, virus diseases, inflammatory disorders, and immuno-modulation. Preferred examples of immuno-modulation are tuberculosis, lepra, and psoriasis. When the disorder is hyperglycemia, the treatment thereof does not cause hypoglycemia.
The methods delineated herein can also include the step of identifying that the subject is in need of treatment of the diseases or disorders described above. The identification can be in the judgment of a subject or a health professional and can be a subjective (e.g., opinion) or objective (e.g., measurable by a test or diagnostic method).
These compounds may also be used in the manufacture of a medicament for the prevention, management or treatment of diabetes, syndrome X, obesity, glaucoma, hyperlipidemia, hyperglycemia, hyperinsulinemia, hypertension, osteoporosis, dementia, depression, virus diseases or inflammatory disorders without causing hypoglycemia and to achieve immuno-modulation. Preferred examples of immuno-modulation are tuberculosis, lepra, and psoriasis.
It is preferred that:
T is selected from 5-chloro-l,3-dimethyl-lH-pyrazol-4-yl; 4-chloro-2,3,l- benzoxadiazolyl; 5-(dimethylamino)-l-naphthyl; l-methylimidazol-4-yl; 1-naphthyl; 2- naphthyl; 8-quinolinyl; thienyl substituted with one or more of (benzoylamino)methyl, bromo, chloro, 3- isoxazolyl, 2-(methylsulfanyl)-4-pyrimidinyl, l-methyl-5-(trifluoromethyl)pyrazol-3-yl, phenylsulfonyl, pyridyl; phenyl substituted with one or more of acetylamino, 3-acetylaminophenyl, 3- acetylphenyl, benzeneamino, l,3-benzodioxol-5-yl, 2-benzofuryl, benzylamino, 3,5- bis(trifluoromethyl)ρhenyl, bromo, butoxy, carboxy, chloro, 4-carboxyphenyl, 3-chloro-2- cyanophenoxy, 4-chlorophenyl, 5-chloro-2-thienyl, cyano, 3,4-dichlorophenyl, ({[4-(2- ethoxy-2-oxoethyl)-l,3-thiazol-2-yl]amino}carbonyl), fluoro, 5-fluoro-2-methoxyphenyl, 2- furyl, hydrogen, iodo, isopropyl, methanesulfonyl, methoxy, methyl, 4-methyl-l -piperazinyl, 4-methyl-l -piperidinyl, 4-methylsulfanylphenyl, 5-methyl-2 -thienyl, 4-moφholinyl, nitro, 3- nitrophenyl, phenoxy, phenyl, n-propyl, 4-pyridyl, 3-pyridylmethylamino, 1-pyrrolidinyl, 2- thienyl, 3 -thienyl, 2-thienylmethylamino, trifluoromethoxy, 4-trifluoromethoxyphenyl, trifluoromethyl; or
R1 is hydrogen or methyl;
B\ and B2 are B3 or Z, provided that B1 and B2 have different meanings, wherein:
• Z is selected from l-benzothien-3-yl, 3-(2,5-dimethylfuryl), pyridinyl; thienyl optionally substituted with one or more of chloro, methylsulfonyl; phenyl optionally substituted with one or more of ethoxycarbonyl, nitro, fluoro, methyl, methoxy, acetylamino, chloro, 4-chlorophenoxy, trifluoromethyl; or is X-Y-R2, wherein
• X is CH2 or CO;
• Y is CH2, CO or a single bond; • R2 is selected from n-propyl, azido, bromo, chloro, 2-pyridinylsulfanyl, 3-oxo-4- moφholinolinylmethylene, ethoxycarbonyl, 5-methyl- 1 ,3 ,4-oxadiazol-2-yl, hydroxymethyl, 2-hydroxyethylaminomethyl, methylsulfonyloxymethyl;
NR3R4, wherein R3 and R4 are each independently selected from acetyl, benzhydryl, l,3-benzodioxol-5-ylmethyl, benzyl, 3-chloro-2-methylphenylsulfbnyl, cyclohexyl, cyclohexylmethyl, cyclopropanecarbonyl, ethyl, 2-furylcarbonyl, 2-furylmethyl, hydrogen, 2- hydroxyethyl, 2-(lH-indol-3-yl)ethyl, isopropyl, methoxy, 2-methoxyethyl, methyl, 4-(l-
methylimidazolyl)sulfonyl, methylsulfonyl, phenyl, (lS)-phenylethyl, n-propyl, tetrahydro-2- furanylmethyl, trifluoromethylsulfonyl, N-carbethoxypiperidyl; or
NR3R4 represent together 4-acetylpiperazinyl, 4-t-butoxycarbonylpiperazinyl, 2-(3,4- dihydro-2(lH)isoquinolinyl), (2R,6S)-2,6-dimethylmoφholinyl, (2R)-2,4-dimethyl- 1 - piperazinyl, 2-hydroxy-3-oxomoφholinyl, imidazolyl, 2-methyl-3-oxomoφholinyl, 4- methyl-2-oxopiperazinyl, 4-methylpiperazinyl, moφholinyl, (lS,4S)-2-oxa-5-aza- bicyclo[2.2.1]hept-5-yl, 2-oxoimidazolinyl, 3-oxomoφholinyl, 3-oxo-l,4-oxazepinyl, 2- oxooxazolinyl, piperazinyl; piperidinyl; pyrrolidinyl; pyrrolidonyl, thiomoφholinyl; 1,1- dioxido-thiomoφholinyl; OCONR3R4, wherein R3 and R4 are each independently selected from ethyl, hydrogen or form together with the N-atom to which they are attached moφholinyl;
R5O, wherein R5 is acetyl, benzoyl, benzyl, ethyl, 2-fluoroethyl, 2-furylcarbonyl, hydrogen, isobutyryl, isopropyl, methyl, 2-carbomethoxyphenyl, methylsulfonyl, phenyl, n- propionyl, 3-pyridinyl, 2,2,2-trifluoroethyl; • B3 is hydrogen, methyl or dimethylaminomethyl.
Specific examples of compounds according to the present invention are given above.
Another object of the present invention is a pharmaceutical composition comprising at least one compound of formula (I) as defined above, and a pharmaceutically acceptable carrier. Also within the scope of this invention is a method for making a compound of formula (I). The method includes taking any intermediate compound delineated herein, reacting it with one or more reagents to form a compound of formula (I) including any processes specifically delineated herein.
Other features and advantages of the invention will be apparent from the detailed description and claims.
DETAILED DESCRIPTION OF THE INVENTION
The compounds according to the present invention may be used in several indications which involve 11-β-hydroxysteroid dehydrogenase type 1 enzyme. Thus, the compounds according to the present invention may be used against dementia (see WO97/07789),
osteoporosis (see Canalis E 1996, Mechanisms of glucocorticoid action in bone: implications to glucocorticoid-induced osteoporosis, Journal of Clinical Endocrinology and Metabolism, 81, 3441-3447) and may also be used disorders in the immune system (see Franchimont et al, "Inhibition of Thl immune response by glucocorticoids: dexamethasone selectively inhibits IL-12-induced Stat 4 phosphorylation in T lymphocytes", The journal of Immunology 2000, Feb 15, vol 164 (4), pages 1768-74) and also in the above listed indications.
The various terms used, separately and in combinations, in the above definition of the compounds having the formula (I) will be explained.
The term "aryl" in the present description is intended to include aromatic rings (monocyclic or bicyclic) having from 6 to 10 ring carbon atoms, such as phenyl (Ph) and naphthyl, which optionally may be substituted by C1-6-alkyl. Examples of substituted aryl groups are benzyl, and 2-methylphenyl.
The term "heteroaryl" means in the present description a monocyclic, bi- or tricyclic aromatic ring system (only one ring need to be aromatic) having from 5 to 14, preferably 5 to 10 ring atoms such as 5, 6, 7, 8, 9 or 10 ring atoms (mono- or bicyclic), in which one or more of the ring atoms are other than carbon, such as nitrogen, sulfur, oxygen and selenium as part of the ring system. Examples of such heteroaryl rings are pyrrole, imidazole, thiophene, furan, thiazole, isothiazole, thiadiazole, oxazole, isoxazole, oxadiazole, pyridine, pyrazine, pyrimidine, pyridazine, pyrazole, triazole, tetrazole, chroman, isochroman, quinoline, quinoxaline, isoquinoline, phthalazine, cinnolme, quinazoline, indole, isoindole, indoline, isoindoline, benzothiophene, benzofuran, isobenzofuran, benzoxazole, 2,1,3-benzoxadiazole, benzothiazole, 2,1,3-benzothiazole, 2,1,3-benzoselenadiazole, benzimidazole, indazole, benzodioxane, indane, 1,2,3,4-tetrahydroquinoline, 3,4-dihydro-2H-l,4-benzoxazine, 1,5- naphthyridine, 1,8-naphthyridine, acridine, fenazine and xanthene. The term "heterocyclic" in the present description is intended to include unsaturated as well as partially and fully saturated mono-, bi- and tricyclic rings having from 4 to 14, preferably 4 to 10 ring atoms having one or more heteroatoms (e.g., oxygen, sulfur, or nitrogen) as part of the ring system and the reminder being carbon, such as, for example, the heteroaryl groups mentioned above as well as the corresponding partially saturated or fully saturated heterocyclic rings. Exemplary saturated heterocyclic rings are azetidine, pyrrolidine, piperidine, piperazine, moφholine, thiomoφholine and 1,4-oxazepane.
C1-6-alkyl in the compound of formula (I) according to the present application, which may be straight, branched or cyclic, is preferably C1-4-alkyl. Exemplary alkyl groups include methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, isopentyl, hexyl, isohexyl, and cyclohexyl. For parts of the range "C1-6-alkyl" all subgroups thereof are contemplated such as C1-5-alkyl, Cι-4-alkyl, Cι-3-alkyl, C1-2-alkyl, C -6-alkyl, C2-5-alkyl, C2-4- alkyl, C2-3-alkyl, C3-6-alkyl, C4-5-alkyl, etc.
C1-6-alkoxy, in the compound of formula (I) according to the present application may be straight or branched, is preferably C1-4-alkoxy. Exemplary alkoxy groups include methoxy, ethoxy, propoxy, isopropoxy, butoxy, sec-butoxy, tert-butoxy, pentyloxy, isopentyloxy, hexyloxy, and isohexyloxy. For parts of the range "Cι-6-alkoxy" all subgroups thereof are contemplated such as C1-5-alkoxy, C1-4-alkoxy, Cι-3-alkoxy, C1-2-alkoxy, C2-6- alkoxy, C2-5-alkoxy, C2-4-alkoxy, C2-3-alkoxy, C3-6-alkoxy, C4-5-alkoxy, etc.
Cι-6-acyl, in the compound of formula (I) according to the present application may be saturated or unsaturated and is preferably C1-4-acyl. Exemplary acyl groups include formyl, acetyl, propionyl, butyryl, isobutyryl, valeryl, isovaleryl, butenoyl (e.g. 3-butenoyl), hexenoyl (e.g. 5-hexenoyl). For parts of the range "Cι-6-acyl" all subgroups thereof are contemplated such as C1- -acyl, C1-4-acyl, C1- -acyl, C1-2-acyl, C2-6-acyl, C2-5-acyl, C2- -acyl, C2-3-acyl, C3-6-acyl, C -5-acyl, etc.
C2-6-alkenyl in the compound of formula (I) according to the present application, which may be straight, branched or cyclic, is preferably C2-4-alkenyl. Exemplary alkenyl groups include vinyl, 1-propenyl, 2-propenyl, isopropenyl, 1-butenyl, 2-butenyl, 1-pentenyl, 2-pentenyl, 1-hexenyl, 2-hexenyl, and 1-cyclohexenyl. For parts of the range "C2-6-alkenyl" all subgroups thereof are contemplated such as C2-5-alkenyl, C2- -alkenyl, C2-3-alkenyl, C -6- alkenyl, C -5-alkenyl, etc. The term "halogen" in the present description is intended to include fluorine, chlorine, bromine and iodine.
The term "sulfanyl" in the present description means a thio group. With the expression "mono- or di-substituted" is meant in the present description that the functionalities in question may be substituted with independently Cι-6-acyl, C2-6-alkenyl, C1-6-(cyclo)alkyl, aryl, pyridylmethyl, or heterocyclic rings e.g. azetidine, pyrrolidine, piperidine, piperazine, moφholine and thiomoφholine, which heterocyclic rings optionally
maybe substituted with C1-6-alkyl. With the expression "optionally mono- or disubstituted" is meant in the present description that the functionalities in question may also be substituted with independently hydrogen.
Combinations of substituents and variables envisioned by this invention are only those that result in the formation of stable compounds. The term "stable", as used herein, refers to compounds which possess stability sufficient to allow manufacture and which maintains the integrity of the compound for a sufficient period of time to be useful for the puφoses detailed herein (e.g., therapeutic administration to a subject for the treatment of disease, 11-β-HSDl inhibition, 11-β-HSDl -mediated disease). The term "prodrug forms" in the present description means a pharmacologically acceptable derivative, such as an ester or an amide, which derivative is biotransformed in the body to form the active drug (see Goodman and Gilman's, The Pharmacological basis of Therapeutics, 8th ed., McGraw-Hill, Int. Ed. 1992, "Biotransformation of Drags, p. 13-15). "Pharmaceutically acceptable" means in the present description being useful in preparing a pharmaceutical composition that is generally safe, non-toxic and neither biologically nor otherwise undesirable and includes being useful for veterinary use as well as human pharmaceutical use.
"Pharmaceutically acceptable salts" mean in the present description salts which are pharmaceutically acceptable, as defined above, and which possess the desired pharmacological activity. Such salts include acid addition salts formed with organic and inorganic acids, such as hydrogen chloride, hydrogen bromide, hydrogen iodide, sulfuric acid, phosphoric acid, acetic acid, glycolic acid, maleic acid, malonic acid, oxalic acid, methanesulfonic acid, trifluoroacetic acid, fumaric acid, succinic acid, tartaric acid, citric acid, benzoic acid, ascorbic acid and the like. Base addition salts may be formed with organic and inorganic bases, such as sodium, ammonia, potassium, calcium, ethanolamine, diethanolamine, N-methylglucamine, choline and the like. Included in the invention are pharmaceutically acceptable salts or compounds of any of the formulae herein.
Pharmaceutical compositions according to the present invention contain a pharmaceutically acceptable carrier together with at least one of the compounds comprising the formula (I) as described herein above, dissolved or dispersed therein as an active, antimicrobial, ingredient. In a preferred embodiment, the therapeutic composition is not
immunogenic when administered to a human patient for therapeutic puφoses, unless that puφose is to induce an immune response.
The preparation of a pharmacological composition that contains active ingredients dissolved or dispersed therein is well understood in the art. Typically such compositions are prepared as sterile injectables either as liquid solutions or suspensions, aqueous or non- aqueous, however, solid forms suitable for solution, or suspensions, in liquid prior to use can also be prepared. The preparation can also be emulsified.
The active ingredient may be mixed with excipients, which are pharmaceutically acceptable and compatible with the active ingredient and in amounts suitable for use in the therapeutic methods described herein. Suitable excipients are, for example, water, saline, dextrose, glycerol, ethanol or the like and combinations thereof. In addition, if desired, the composition may contain minor amounts of auxiliary substances such as wetting or emulsifying agents, pH buffering agents and the like which enhance the effectiveness of the active ingredient. Adjuvants may also be present in the composition. Pharmaceutically acceptable carriers are well known in the art. Exemplary of liquid carriers are sterile aqueous solutions that contain no materials in addition to the active ingredients and water, or contain a buffer such as sodium phosphate at physiological pH value, physiological saline or both, such as phosphate-buffered saline. Still further, aqueous carriers can contain more than one buffer salt, as well as salts such as sodium and potassium chlorides, dextrose, propylene glycol, polyethylene glycol and other solutes.
Liquid compositions can also contain liquid phases in addition to and to the exclusion of water. Exemplary of such additional liquid phases are glycerine, vegetable oils such as cottonseed oil, organic esters such as ethyl oleate, and water-oil emulsions.
The pharmaceutical composition according to one of the preferred embodiments of the present invention comprising compounds comprising the formula (I), may include pharmaceutically acceptable salts of that component therein as set out above. Pharmaceutically acceptable salts include the acid addition salts (formed with the free amino groups of the polypeptide) that are formed with inorganic acids such as, for example, hydrochloric or phosphoric acids, or such organic acids as acetic acid, tartaric acid, mandelic acid and the like. Salts formed with the free carboxyl groups can also be derived from inorganic bases such as, for example, sodium, potassium, ammonium, calcium or ferric
hydroxides, and such organic bases as isopropylamine, trimethylamine, 2-ethylamino ethanol, histidine, procaine and the like.
The preparations according to the preferred embodiments may be administered orally, topically, intraperitoneally, intraarticularly, intracranially, intradermally, intramuscularly, intraocularly, intrathecally, intravenously, subcutaneously. Other routes are known to those of ordinary skill in the art.
The orally administrable compositions according to the present invention may be in the form of tablets, capsules, powders, granules, lozenges, liquid or gel preparations, such as oral, topical or sterile parenteral solutions or suspensions. Tablets and capsules for oral administration may be in unit dose presentation form and may contain conventional excipients such as binding agents, for example syrup, acacia, gelatin, sorbitol, traganath or polyvinyl-pyrrolidone; fillers e.g. lactose, sugar, maize-starch, calcium phosphate, sorbitol or glycine; tabletting lubricant e.g. magnesium stearate, talc, polyethylene glycol or silica; disintegrants e.g. potato starch, or acceptable wetting agents such as sodium lauryl sulfate. The tablets may be coated according to methods well known in normal pharmaceutical practice. Oral liquid preparations may be in the fonn of e.g. aqueous or oily suspensions, solutions, emulsions, syrups or elixirs or may be presented as a dry product for reconstitution with water or other suitable vehicle before use. Such liquid preparations may contain conventional additives such as suspending agents, e.g. sorbitol, syrup, methyl cellulose, glucose syrup, gelatin hydrogenated edible fats; emulsifying agents e.g. lecithin, sorbitan monooleate or acacia, non-aqueous vehicles (which may include edible oils), e.g. almond oil, fractionated coconut oil, oily esters such as glycerine, propylene glycol, or ethyl alcohol; preservatives e.g. methyl or propyl p-hydroxybenzoate or sorbic acid, and if desired conventional flavouring or colouring agents. "An effective amount" refers to an amount of a compound which confers a therapeutic effect on the treated subject. The therapeutic effect may be objective (i.e., measurable by some test or marker) or subjective (i.e., subject gives an indication of or feels an effect). A pharmaceutical composition according to the present invention, may comprise typically an amount of at least 0.1 weight percent of compound comprising the formula (I) per weight of total therapeutic composition. A weight percent is a ratio by weight of total composition. Thus, for example, 0.1 weight percent is 0.1 grams of compound comprising
the formula (I) per 100 grams of total composition. A suitable daily oral dose for a mammal, preferably a human being, may vary widely depending on the condition of the patient. However a dose of compound comprising the formula (I) of about 0.1 to 300 mg/kg body weight may be appropriate. The compositions according to the present invention may also be used veterinarily and thus they may comprise a veterinarily acceptable excipient or carrier. The compounds and compositions may be thus administered to animals, e.g., cats, dogs, or horses, in treatment methods.
The compounds of the present invention in labelled form, e.g. isotopically labelled, may be used as a diagnostic agent.
This invention relates to methods of making compounds of any of the formulae herein comprising reacting any one or more of the compounds of the formulae delineated herein, including any processes delineated herein. The compounds of formula (I) above may be prepared by, or in analogy with, conventional methods, and especially according to or in analogy with the following methods. Further, the pharmacology in-vitro was studied using the following reagents and methods.
The chemicals used in the synthetic routes delineated herein may include, for example, solvents, reagents, catalysts, and protecting group and deprotecting group reagents. The methods described above may also additionally include steps, either before or after the steps described specifically herein, to add or remove suitable protecting groups in order to ultimately allow synthesis of the compounds. In addition, various synthetic steps may be performed in an alternate sequence or order to give the desired compounds. Synthetic chemistry transformations and protecting group methodologies (protection and deprotection) useful in synthesizing applicable compounds are known in the art and include, for example, those described in R. Larock, Comprehensive Organic Transformations, VCH Publishers
(1989); T.W. Greene and P.G.M. Wuts, Protective Groups in Organic Synthesis, 3rd Ed., John Wiley and Sons (1999); L. Fieser and M. Fieser, Fieser andFiesers ' Reagents for Organic Synthesis, John Wiley and Sons (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995) and subsequent editions thereof.
All publications mentioned herein are hereby incoφorated by reference. By the expression "comprising" means "including but not limited to." Thus, other non-mentioned substances, additives or carriers may be present.
The invention will now be described in reference to the following Examples. These Examples are not to be regarded as limiting the scope of the present invention, but shall only serve in an illustrative manner.
EXAMPLES
EXPERIMENTAL METHODS
Scintillation Proximity Assay
[1, 2(n) - 3H] -cortisone was purchased from Amersham Pharmacia Biotech. Anti- cortisol monoclonal mouse antibody, clone 6D6.7 was obtained from Immunotech and Scintillation proximity assay (SPA) beads coated with monoclonal antimouse antibodies were from Amersham Pharmacia Biotech. NADPH, tetrasodium salt was from Calbiochem and glucose-6-phosphate (G-6-P) was supplied by Sigma. The human 11-β-hydroxysteroid dehydrogenase type-1 enzyme (11-β-HSD^ was expressed in Pichiapastoris. 18-β- glycyrrhetinic acid (GA) was obtained from Sigma. The serial dilutions of the compounds were performed on a Tecan Genesis RSP 150. Compounds to be tested were dissolved in DMSO (1 mM) and diluted in 50 mM Tris-HCl, pH 7.2 containing 1 mM EDTA.
The multiplication of plates was done on a WallacQuadra. The amount of the product [3H]-cortisol, bound to the beads was determined in a Packard, Top Count microplate liquid scintillation counter.
The 11-β-HSDi enzyme assay was carried out in 96 well microtiter plates (Packard, Optiplate) in a total well volume of 220 μL and contained 30 mM Tris-HCl, pH 7.2 with 1 mM EDTA, a substrate mixture tritiated Cortisone/NADPH (175 nM / 181 μM), G-6-P (1 mM) and inhibitors in serial dilutions (9 to 0.15 μM). Reactions were initiated by the addition of human 11-β-HSDu either as Pichiapastoris cell homogenate or microsomes prepared from Pichiapastoris (the final amount of enzyme used was varied between 0.057 to 0.11 mg/mL). Following mixing, the plates were shaken for 30 to 45 minutes at room temperature. The reactions were terminated with 10 μL 1 mM GA stop solution. Monoclonal
mouse antibody was then added (10 μL of 4 μM) followed by 100 μL of SPA beads (suspended according to the manufacturers instructions). Appropriate controls were set up by omitting the 11-β-HSDt to obtain the non-specific binding (NSB) value.
The plates were covered with plastic film and incubated on a shaker for 30 minutes, at room temperature, before counting. The amount of [ H]-cortιsol, bound to the beads was determined in a microplate liquid scintillation counter.
The calculation of the Kj values for the inhibitors was performed by use of Activity Base. The K; value is calculated from IC50 and the Km value is calculated using the Cheng Prushoff equation (with reversible inhibition that follows the Michaelis-Menten equation): Kj = IC50(l+[S]/Km) [Cheng, Y.C; Prushoff, W.H. Biochem. Pharmacol. 1973, 22, 3099-3108]. The IC50 is measured experimentally in an assay wherein the decrease of the turnover of cortisone to cortisol is dependent on the inhibition potential of each substance. The Ki values of the compounds of the present invention for the 11-β-HSDl enzyme lie typically between about 10 nM and about 10 μM.
COMPOUND PREPARATION General:
For preparative straight phase HPLC purification a Phenomenex column (250 x 21.1 mm, 10 μm) was used on a Gilson system eluting with ethanol in chloroform (gradient from 0 - 10%) in 10 min) with a flow of 20 mL/min. Column chromatography was performed on silica using Silica gel 60 (230-400 mesh), Merck. Melting points were determined on a Gallenkamp apparatus. Elemental analyses were recorded using a Vario EL instrument. HPLC analyses were performed using a Hypersil Elite column (150 x 4.6 mm, 3μ) with a flow of 3 mL / min on a Waters 600E system with monitoring at 254 nm. Reverse phase preparative HPLC was carried out on a 100 x 21.2 mm, 5μ Hypersil Elite column eluting with a gradient of 5% ACN in 95% water to 95% ACN in 5% water (0.2%> TFA buffer) over 10 mins at a flow rate of 20 mL / min with the UV detector set at 254 nm. Thin layer chromatography was carried out using pre-coated silica gel F-254 plates (thickness 0.25 mm). Electrospray MS spectra were obtained on a Micromass platform LCMS spectrometer. Crude, worked up compounds were purified by flash column chromatography using pre
packed silica SPE columns (10 g silica) on an Isco Foxy 200 Combiflash system, and a gradient of 16.67% ethyl acetate in hexane increasing incrementally to 100% ethyl acetate.
List of Abbreviations
ACN = acetonitrile
DCM = dichloromethane
DIEA = N,N-diisopropylethylamine
DMAP = 4-dimethylaminopyridine DME = ethyleneglycol dimethyl ether
DMF = dimethylformamide
DMSO = dimethyl sulfoxide
EDCI = l-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride
EDTA = ethylenediaminetetraacetic acid HCOOH = formic acid
HO AT = l-hydroxy-7-azabenzotriazole
HOBT = 1-hydroxybenzotriazole hydrate
MTBE = tert-butyl methyl ether
TEA = triethylamine TFA = trifluoroacetic acid
THF = tetrahydrofuran
SULFONAMIDE COUPLINGS: METHOD A: 1 Eq of the thiophene derivative with an exocyclic amino group was dissolved in pyridine (0.5 M solution). The sulfonyl chloride (1.2 eq) was added and the reaction mixture was stirred at ambient temperature under nitrogen atmosphere for 15 h. The reaction mixture was poured into aqueous HCl (1 M). If the product precipitated it was collected on a filter and washed with aqueous HCl (1 M) and recrystallised from ethanol. In case an oil was obtained, the crude was extracted with DCM and worked up and purified using standard procedures.
METHOD B:
A solution of the thiophene derivative with an exocyclic amino group (1 eq), triethylamine (2 eq) and DMAP (1 eq) in DMF (1 M) and DCM (0.225 M) was dispensed into a reaction vial. The sulfonyl chloride (1.2 eq) was dissolved in DCM (0.33 M) and added. The reaction mixtures were kept at room temperature over night. The mixture was then added to petroleum ether (10 times reaction volume). After some hours in refrigerator the supernatants were decanted and (a portion of) the residual materials were dissolved in DMSO-methanol-acetic acid (300 μL + 500 μL + 50 μL) and purified by preparative LCMS (acetonitrile-water gradients). The purest fractions were collected and lyophilized.
Alternatively, the crude was isolated using extractive work-up and purified using standard procedures.
SAPONIFICATIONS: METHOD C:
1 Eq of the ester was suspended in 95%> ethanol (0.1 M) and treated with KOH (aqueous, 6 eq). Water was added until a clear solution was achieved. The reaction mixture was stirred for 2-3 h at ambient temperature. The solvent was removed under reduced pressure and the crude was redissolved in water. Addition of cone. HCl until pH 2 gave a precipitate which was collected on a filter and washed with cold water and dried.
AMIDE COUPLINGS: METHOD D:
The carboxylic acid ester was dissolved (0.05 M) in a large excess of the amine in 40 or 70% water-solution. The reaction mixture was stirred at ambient temperature over night. The solvent was removed under reduced pressure and the crude product was purified by flash column cliromatography on silica gel eluting with methanol (0-»6%>) in DCM.
METHOD E: The carboxylic acid was suspended in DCM (0.05M) followed by the addition of
EDCI (1.1 eq), triethylamine (3 eq), DMAP (0.5 eq) and the amine of choice (1.2 eq). DMF
was added when the starting materials did not dissolve properly. The reaction mixture was stirred at ambient temperature over night. The organic phase was washed with aqueous HCl (1 M), dried over sodium sulfate, filtered and evaporated in vacuo. The crude product amide was purified by flash column chromatography on silica gel, eluting with methanol ( 1 →3 →6%) in DCM or ethyl acetate.
METHOD F:
The carboxylic acid was suspended in DCM (0.1 M) and cooled to 0°C under nitrogen (g) atmosphere. EDCI (1 eq), HOAT (1 eq) or HOBT (1 eq) was added, followed by TEA (2.2 eq). After 10 min, the amine of choice (1.2 eq) was added and the reaction mixture was allowed to warm to ambient temperature. After 5 h, the DCM phase was washed with aqueous HCl (1 M) and worked up and purified as described in METHOD E.
METHOD G: Under N2-atmosphere, aluminium chloride (1 eq) was suspended in DCM (0.1 M) and treated with the amine of choice (4 eq) at ambient temperature. After 10 min, the alkyl ester (1 eq) was added and the reaction mixture was stirred until starting material had been consumed (TLC). Quenching with saturated aqueous sodium hydrogen carbonate or aqueous HCl (1 M) and extractive workup with ethyl acetate gave the crude products which were then purified by flash chromatography on silica gel eluting with DCM / methanol mixtures.
ACYLATIONS: METHOD J:
To a solution of the alcohol in dry pyridine (0.3 M), 1.1 eq of acid chloride was added at 0 °C The reaction mixture was stirred at room temperature for 6 h, concentrated, co- evaporated with acetonitrile, re-dissolved in DCM, washed with aqueous HCl (0.5 M), dried with sodium sulfate and chromatographed on silica gel using petroleum-ether and ethyl acetate as eluents.
CARBAMATES:
METHOD K:
To a solution of the alcohol in dry pyridine (0.3 M), 1.5 eq of 4-nitrophenyl chloroformate (0.5 M in dry pyridine) was added at 0°C After the reaction mixture was stirred at room temperature for 12 h, 5 eq of primary or secondary amine were added at 0°C The solution was stirred at room temperature for 3 h, concentrated, co-evaporated with acetonitrile, re-dissolved in DCM, washed with aqueous HCl (0.5 M) and saturated aqueous sodium bicarbonate, dried with sodium sulfate and chromatographed on silica gel using DCM and methanol as eluents.
SULFONYL CHLORIDES Arylsulfonyl chlorides that were not commercially available were prepared from the aniline derivatives according to literature procedures (see for instance: Hoffman, R. V. (1981) Org. Synth. 60: 121).
PREPARATION OF COMPOUNDS IN EXAMPLES 1-8
EXAMPLE 1 - preparation of starting materials for Examples 2-8
Step a - preparation of dimethyl 2-(3-nitrofhien-2-yl)malonate
A suspension of NaH (320 mg, 13.4 mmol) in 7 mL anhydrous DMF was stirred and treated drop wise with a solution of dimethylmalonate (1.53 mL, 13.4 mmol) in 4 mL DMF. After gas evolution (-10 min) the mixture was added 1 g (6.1 mmol) 2-chloro 3- nitrothiophene in 7 mL DMF. The mixture turned red and was left at 100°C over night. The solvent was then evaporated and the residue was treated with 7 mL H2O and extracted with 3 x 5 mL CH2C12, dried (Na2SO4) and evaporated. After flash cliromatography (10% EtOAc in cyclohexane) and recrystaUisation from petroleumether 515 mg, 33%, of the product was recovered.
1H NMR (400 MHz, CDC13) δ ppm 3.73 (s, 3 H), 4.20 (s, 2 H), 7.17 (d, J=5.86 Hz, 1 H), 7.61 (d, J=5.62 Hz, 1 H); MS (El) M 259.
Step b - preparation of methyl (3-nitrothien-2-yl acetate
2.5 g (9.63 mmol) of dimethyl 2-(3-nitrothien-2-yl)malonate was added 50 mL 5M HCl (aq) and the reaction was refluxed for 15 hours. 100 mL H2O was added and the mixture extracted with 3 x 30 mL EtOAc, dried (MgSO ) and evaporated. This gave 1.54 g of a brown solid. The solvent was dissolved in 50 mL MeOH and 0.250 mL cone. H2SO4 was added. The reaction was then refluxed for another 15 hours. 100 mL H2O was then added and the product extracted with 3 x 30 mL CH C12, dried with MgSO4 and evaporated. After flash chromatography (20%> EtOAc in cyclohexane) 1.37 g (6.82 mmol, 71%>) of the product was recovered as a yellow crystal.
1H NMR (400 MHz, CDC13) δ ppm 3.73 (s, 3 H) 4.20 (s, 2 H) 7.17 (d, J=5.86 Hz, 1 H) 7.61 (d, J-5.62 Hz, 1 H); MS (El) M 201.
Step c - preparation of methyl (3-aminothien-2-yl acetate
300 mg (1.49 mmol) of methyl (3-nitrothien-2-yl)acetate and 100 mg 10% Pd/C were dissolved in 10 mL EtOAc and 2 mL EtOH. The reaction was placed in a bomb under 40 psi of H2 for 12 hours. The Pd/C was then filtered off and the solvent removed under vacuum to give the product as a brown-yellow oil. 201 mg (1.17 mmol, 79%>).
1H NMR (400 MHz, CDC13) δ ppm 3.69 (s, 3 H) 3.91 (s, 2 H) 7.01 (d, J=5.37 Hz, 1 H) 7.16 (d, J-5.62 Hz, 1 H) 8.83 (s, 2 H); MS (ES) M+H 172.
In Examples 2-8 below, either of the following methods were used:
Method A
200 mg (1.17 mmol) of methyl (3-aminothien-2-yl)acetate and 1.20 mmol of the sulfonyl chloride were mixed in 3 mL toluene and 0.1 mL pyridine was added. The reaction was put in the micro wave oven at 120°C for 10 minutes. The solvent was removed under vacuum and the product purified by prep HPLC.
Method B
185 mg (1.08 mmol) of methyl (3-aminothien-2-yl)acetate and 1.3 mmol of the sulfonyl chloride were dissolved in 3mL pyridine and stirred at room temperature for 24
hours. The mixture was then poured in to 15 mL IM HCl and extracted with 3x7 mL CH2C12, dried (MgSO4) and evaporated. The product was purified by prep HPLC.
EXAMPLE 2 - methyl (3-{ ("3-chloro-2-methylphenyl)sulfonyl1aminolthien-2-yl acetate Method B was used.
!H NMR (400 MHz, CDC13) δ ppm 2.70 (s, 3 H) 3.56 (s, 2 H) 3.72 (s, 3 H) 6.72 (d, J=5.37 Hz, 1 H) 7.02 (d, J=5.62 Hz, 1 H) 7.19 (d, J=7.81 Hz, 1 H) 7.47 (s, 1 H) 7.57 (d, J=8.06 Hz, 1 H) 7.83 (d, J=7.81 Hz, 1 H); MS (ES) M+H 260.
EXAMPLE 3 - methyl ("3-{ (5-fluoro-2-methylphenyl)sulfonyl1amino}thien-2-yl acetate
Method B was used.
1H NMR (400 MHz, CDC13) δ ppm 2.59 (s, 3 H) 3.54 (s, 2 H) 3.74 (s, 3 H) 6.75 (d,
J=5.62 Hz, 1 H) 7.05 (d, J=5.37 Hz, 1 H) 7.17 (m, 1 H) 7.29 (m, 1 H) 7.48 (s, 1 H) 7.61 (dd,
J=8.55, 2.93 Hz, 1 H); MS (ES) M+H 344.
EXAMPLE 4 - methyl (3-{[(3-cvanophenyl sulfonyl]amino}thien-2-yQacetate
Method B was used.
1H NMR (400 MHz, CDC13) δ ppm 3.46 (m, 2 H) 3.73 (s, 3 H) 6.86 (d, J=3.91 Hz, 1
H) 7.13 (d, J=5.37 Hz, 1 H) 7.51 (s, 1 H) 7.62 (t, J=7.81 Hz, 1 H) 7.86 (d, 1 H) 7.97 (d, 1 H) 8.06 (s, 1 H); MS (ES) M+H 337.
EXAMPLE 5 - methyl (3-{ (4-butoxyphenyl)sulfonyl]amino}thien-2-yl acetate Method B was used.
1H NMR (400 MHz, CDC13) δ ppm 0.99 (t, J=7.32 Hz, 3 H) 1.50 (m, 2 H) 1.79 (m, 2 H) 3.40 (s, 2 H) 3.70 (s, 3 H) 4.00 (t, J=6.59 Hz, 2 H) 6.89 (m, 3 H) 7.08 (m, 2 H) 7.64 (m, 2 H); MS (ES) M+H 384.
EXAMPLE 6 - methyl (3-{["(,2-methylsulfanylpyrimidin-4-ylthiophene sulfonyl1- amino}thien-2-yl)acetate Method A was used. 1H NMR (400 MHz, CDC13) δ ppm 2.58 (s, 3 H) 3.50 (s, 2 H) 3.70 (s, 3 H) 6.98 (d,
J=5.37 Hz, 1 H) 7.12 (d, J=5.37 Hz, 1 H) 7.21 (d, J=5.13 Hz, 1 H) 7.42 (d, J=4.15 Hz, 1 H) 7.53 (s, 1 H) 7.58 (d, J=3.91 Hz, 1 H) 8.53 (d, J=5.13 Hz, 1 H); MS (ES) M+H 442.
EXAMPLE 7 - methyl 3-(r(l-πιethylimidazol-4-yl sulfonyl1amino}thien-2-vDacetate Method A was used.
1H NMR (400 MHz, DMSO-d6) δ ppm 3.59 (s, 3 H) 3.63 (s, 3 H) 3.75 (s, 2 H) 6.75 (d, j=5.37 Hz, 1 H) 7.27 (d, J=5.37 Hz, 1 H) 7.57 (d, j=1.22 Hz, 1 H) 7.76 (d, j=0.98 Hz, 1 H) 9.60 (s, 1 H); MS (ES) M+H 316.
EXAMPLE 8 - methyl (3-{r(4-methylphenyl sulfonyl]amino}thien-2-yl')acetate Method A was used.
1H NMR (400 MHz, CDC13) δ ppm 2.40 (s, 3 H) 3.34 (s, 2 H) 3.68 (s, 3 H) 6.86 (d, J=5.37 Hz, 1 H) 7.06 (d, J=5.62 Hz, 1 H) 7.15 (s, 1 H) 7.23 (d, J=8.55 Hz, 2 H) 7.60 (d, J=8.06 Hz, 2 H); MS (ES) M+H 326.
Various embodiments of the present invention have been described above but a person skilled in the art realizes further minor alterations which would fall into the scope of the present invention. The breadth and scope of the present invention should not be limited by any of the above-described exemplary embodiments, but should be defined only in accordance with the following claims and their equivalents.