ETHYLENE COPOLYMERS The present invention relates to a process for making copolymers by polymerising ethylene and at least two other olefins. The use of certain transition metal compounds to polymerise olefins, for example, ethylene or propylene, is well established in the prior art. The use of Ziegler- Natta catalysts, for example, those catalysts produced by activating titanium halides with organometallic compounds such as triethylaluminium, is fundamental to many commercial processes for manufacturing polyolefins. Over the last three decades, advances in the technology have led to the development of Ziegler-Natta catalysts which have such high activities that olefin polymers and copolymers containing very low concentrations of residual catalyst can be produced directly in commercial polymerisation processes. The quantities of residual catalyst remaining in the produced polymer are so small as to render unnecessary their separation and removal for most commercial applications. Such processes can be operated by polymerising the monomers in the gas phase, or in solution or in suspension in a liquid hydrocarbon diluent, or in the case of propylene, in bulk. Commodity polyethylenes are commercially produced in a variety of different types and grades. Homopolymerisation of ethylene with transition metal based catalysts leads to the production of so-called "high density" grades of polyethylene. These polymers have relatively high stiffness and are useful for making articles where inherent rigidity is required. Copolymerisation of ethylene with higher 1 -olefins (e.g. butene, hexene or octene) is employed commercially to provide a wide variety of copolymers differing in density and in other important physical properties. Particularly important
copolymers made by copolymerising ethylene with higher 1 -olefins using transition metal based catalysts are the copolymers having a density in the range of 0.91 to 0.93. These copolymers which are generally referred to in the art as "linear low density polyethylene" are in many respects similar to the so-called "low density" polyethylene produced by the high pressure free radical catalysed polymerisation of ethylene. Such polymers and copolymers are used extensively in the manufacture of flexible blown film. Polypropylenes are also commercially produced in a variety of different types and grades. Homopolymerisation of propylene with transition metal based catalysts leads to the production of grades with a wide variety of applications. Copolymers of propylene with ethylene or terpolymers with ethylene and higher 1 -olefins are also useful materials, often used in film applications. In recent years the use of certain metallocene catalysts (for example biscyclopentadienylzirconiumdichloride activated with alumoxane) has provided catalysts with potentially high activity. Other derivatives of metallocenes have been shown to be potentially useful for producing polypropylene with good activity, molecular weight and tacticity control. However, metallocene catalysts of this type suffer from a number of disadvantages, for example, high sensitivity to impurities when used with commercially available monomers, diluents and process gas streams, the need to use large quantities of expensive alumoxanes to achieve high activity, difficulties in putting the catalyst on to a suitable support and synthetic difficulties in the production of more complex catalyst structures suitable for polymerising propene in a tactic manner. US 5635573 discloses cycloolefin/olefin copolymers (5/95 to 95/5) prepared using a Group 4 metallocene catalyst. The Examples show, inter alia, copolymers of ethylene and propylene with cyclohexene or ethylene norbornene. Significant incorporation of bulky comonomers into a copolymer containing three of more copolymerised olefin monomers has required the use of special catalysts, eg certain metallocene-based catalysts. An object of the present invention is to provide an improved process for making a copolymer comprising copolymerised units of ethylene with two or more other defined olefins. The present invention provides a process for preparing a copolymer comprising
copolymerising monomers comprising (a) ethylene, (b) an acyclic 1-olefin containing 3 to 20 carbon atoms and (c) a cyclic olefin containing at least one endocyclic double bond, to provide a copolymer comprising (a) 99.8 to 80 weight % of units derived from (a), 0.1 to 10 weight % of units derived from (b) and 0.1 to 10 weight % of units derived from (c), in the presence of a catalyst comprising (1) a transition metal compound having the following Formula A, and optionally (2) an activating quantity of a suitable activator,
wherein Z comprises a five-membered heterocyclic group, the five membered heterocyclic group containing at least one carbon atom, at least one nitrogen atom and at least one other hetero atom selected from nitrogen, sulphur and oxygen, the remaining atoms in said ring being selected from nitrogen and carbon; M is a metal from Group 3 to 11 of the Periodic Table or a lanthanide metal; E and E are divalent groups independently selected from (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups and (vi) heterosubstituted derivatives of said groups (i) to (v); D and D are donor atoms or groups; X is an anionic group, L is a neutral donor group; n = m = zero or 1; y and z are independently zero or integers such that the number of X and L groups satisfy the valency and oxidation state of the metal M. Preferably the copolymer comprises copolymerised units wherein (a) 99.6 to 86 weight % of the units are derived from ethylene, (b) 0.2 to 7 weight % of the units are derived from an acyclic 1-olefin containing 3 to 20 carbon atoms and (c) 0.2 to 7 weight % of the units are derived from the defined endocyclic olefin containing 5 to 20 carbon atoms. More preferably the copolymer comprises copolymerised units wherein (a) 99.2 to 88 weight % of the units are derived from ethylene, (b) 0.4 to 6 weight % of the units
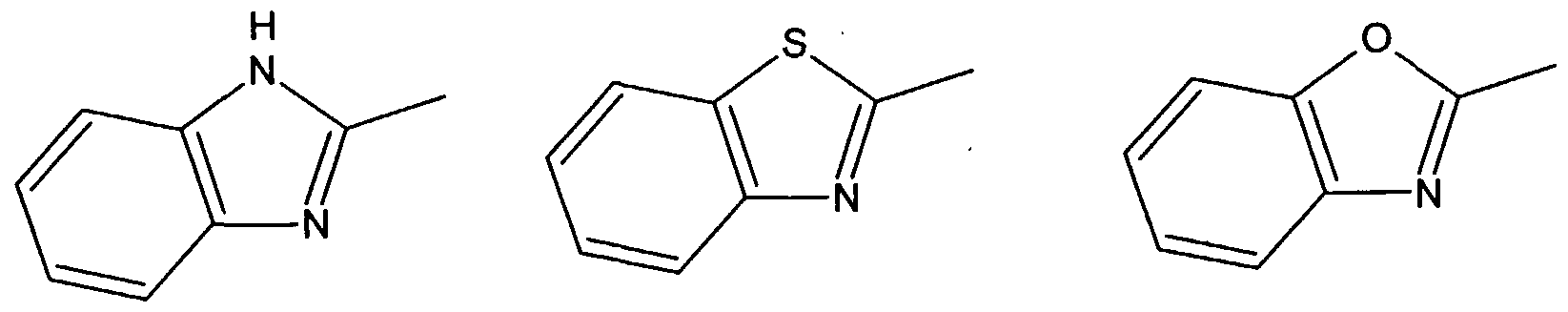
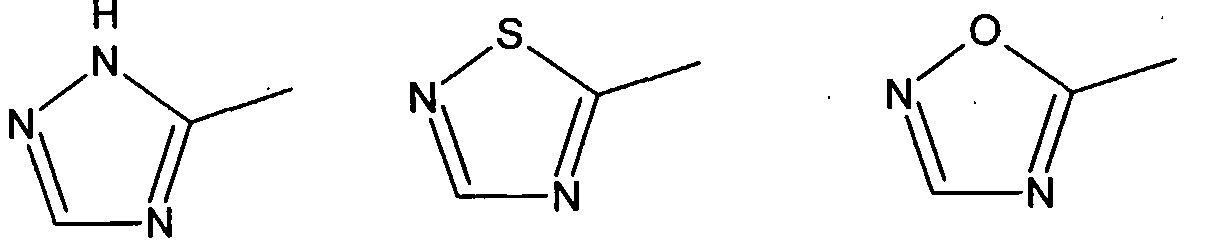
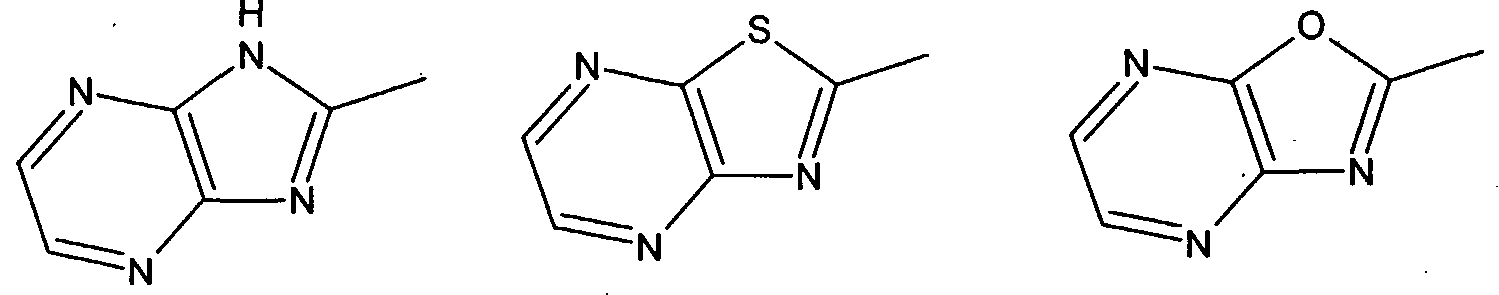
are derived from an acyclic 1-olefin containing 3 to 20 carbon atoms and (c) 0.4 to 6 weight % of the units are derived from the defined endocyclic olefin containing 5 to 20 carbon atoms. The acyclic olefin units are suitably derived from a straight or branched chain 1- olefin, for example, propylene, 1 -butene, 1-pentene, 1-hexene, 4-methyl -1-pentene, 1- octene, 1-decene and 1-dodecene; or diolefin, for example, butadiene, 1,3-pentadiene and isoprene. The defined endocyclic olefin units are suitably derived from an endocyclic mono- , di- or triolefins, examples of which are cyclopentene, cyclohexene, 1- methylcyclopentene, 1 -methylcyclohexene, cyclooctene, 2-bornene (1,7,7- trimethylbicyclo[2.2.1]hept-2-ene), norbornene (bicyclo[2.2.1]-2-heptene), vinylcyclohexene, cyclooctatriene and cyclohexadiene. In addition to the defined units the copolymer of the present invention can contain up to 10 weight % of units derived from one or more other additional comonomers not falling within the definition of a, b or c above. Examples of such additional comonomers are exocyclic olefins such as vinyl cyclohexane, styrene, divinyl benzene, and polar comonomers, for example methyl methacrylate, methyl acrylate, butyl acrylate, acrylonitrile, vinyl acetate and vinyl chloride. In Formula A the divalent groups E1 and E2 are preferably not linked other than through the donor atom or group D1. At least one of the atoms present in the ring of the five-membered heterocyclic group Z is preferably bonded directly to E1 and preferably a second atom in the ring is bonded directly to M. Most preferably the atom in the five-membered ring bonded directly to E1 is adjacent to a second atom in said ring, said second atom being bonded directly to M. The five-membered heterocyclic group Z preferably contains at least 2 carbon atoms in its ring and more preferably at least 3 carbon atoms in its ring. Examples of suitable 5-membered heterocyclic groups are (but are not restricted to):
In a preferred embodiment of the process of the present invention Z, in Formula A of the catalyst, is specifically an imidazole-containing group. The polymerisation catalyst used in the process of the present invention preferably comprises (1) a transition metal compound having the following Formula A, and optionally (2) an activating quantity of a suitable activator,
wherein Z is specifically an imidazole-containing group; M is a metal from Group 3 to 11 of the Periodic Table or a lanthanide metal; E1 and E2 are divalent groups independently selected from (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups and (vi) heterosubstituted derivatives of said groups (i) to (v); D1 and D2 are donor groups; X is an anionic group, L is a neutral donor group; n = m = zero or 1 ; y and z are independently zero or integers such that the number of X and L groups satisfy the valency and oxidation state of the metal M. 1 9 1 D and / or D are donor atoms or groups containing at least one donor atom. D and / or D2 can be, for example, groups having the same formula as recited for group Z.
For example D and / or D can be groups comprising a five-membered heterocyclic group containing at least 2 carbon atoms in its ring and more preferably at least 3 carbon atoms in its ring. D1 and / or D2 can be imidazole-containing groups if desired. When D1 and / or D2 are an imidazole-containing group this or these can be identical with Z. In a preferred embodiment D2 and Z are identical imidazole containing groups. The imidazole-containing group Z is preferably a group of formula I, II or III
Formula I Formula II Formula III
R1 to R1 ' are independently hydrogen or a monovalent (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups, (vi) heterosubstituted derivatives of said groups (i) to (v), and (vii) hydrocarbyl-substituted heteroatom groups. The "free" valence bond on the left of Formulae I, II and III provides at least one of the links of E into the rest of Formula A. The other link or links are preferably provided by at least one of the nitrogen atoms in the imidazole-containing group. These defined groups R1 to R1 ' preferably contain 1 to 30, more preferably 2 to 20, most preferably 2 to 12 carbon atoms. Examples of suitable aliphatic hydrocarbon groups are methyl, ethyl, ethylenyl, butyl, hexyl, isopropyl and tert-butyl. Examples of suitable alicyclic hydrocarbon groups are adamantyl, norbornyl, cyclopentyl and cyclohexyl. Examples of suitable aromatic hydrocarbon groups are phenyl, biphenyl, naphthyl, phenanthryl and anthryl. Examples of suitable alkyl substituted aromatic hydrocarbon groups are benzyl, tolyl, mesityl, 2,6-diisopropylphenyl and 2,4,6-triisopropyl. Examples of suitable heterocyclic groups are 2-pyridinyl, 3-pyridinyl, 2-thiophenyl, 2-furanyl, 2-pyrrolyl, 2-quinolinyl. Suitable substituents for forming heterosubstituted derivatives of said groups R1 to R11 are, for example, chloro, bromo, fluoro, iodo, nitro, amino, cyano, ether, hydroxyl and silyl, methoxy, ethoxy, phenoxy (i.e. -OC6H5), tolyloxy (i.e. -OC6H (CH3)), xylyloxy, mesityloxy, dimethylamino, diethylamino, methylethylamino, thiomethyl, thiophenyl and trimethylsilyl. Examples of suitable heterosubstituted derivatives of said groups (i) to (v) are 2-chloroethyl, 2-bromocyclohexyl, 2-nitrophenyl, 4-ethoxyphenyl, 4-chloro-2-
pyridinyl, 4-dimethylaminophenyl and 4-methylaminophenyl. Examples of suitable hydrocarbyl-substituted heteroatom groups are chloro, bromo, fluoro, iodo, nitro, amino, cyano, ether, hydroxyl and silyl, methoxy, ethoxy, phenoxy (i.e. -OC6H5), tolyloxy (i.e. -OC6H4(CH3)), xylyloxy, mesityloxy, dimethylamino, diethylamino, methylethylamino, thiomethyl, thiophenyl and trimethylsilyl. Any of the substituents R1 to R1 ' may be linked to form cyclic structures. Substituents R2 to R11 may also suitably be inorganic groups such as fluoro, chloro, bromo, iodo, nitro, amino, cyano and hydroxyl. Further suitable imidazole-containing groups may be obtained by removal of substituent Rls for example by deprotonation when R1 is hydrogen, to give formally monoanionic imidazole-containing groups. It is preferred that the imidazole-containing group has a structure described in formula III (a "benzimidazole"). R1 is preferably hydrogen, an aliphatic hydrocarbon group, an aromatic hydrocarbon group or is removed to give a formally monoanionic benzimidazole group. R to R are preferably hydrogen, an aliphatic hydrocarbon group or an aromatic hydrocarbon group. E1 and E2 (hereinafter referred to as "E") can be the same or different. E is independently selected from divalent (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups, (vi) heterosubstituted derivatives of said groups (i) to (v), and (vii) hydrocarbyl-substituted heteroatom groups. Examples of suitable divalent groups E are -CH2-, -CH2CH2-, -CH2 CH2CH2-, 1 ,2-phenylene, trans- 1,2-cyclopentane, trans-1,2- cyclohexane, 2,3-butane, l,l '-biphenyl, l,l '-binaphthyl, and -Si(Me)2-. It is preferred that E is an aliphatic or aromatic hydrocarbon group. More preferably the divalent group E is -CH2-. 1 9 D and D can be the same or different donor groups, for example oxygen, sulfur, an amine, an imine or a phosphine. Preferably D1 and D2 are selected from oxygen, sulfur, an amine of formula -N(R12)- or a phosphine of formula -P(R13)- wherein R12 and R13 are hydrogen or (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups, (vi) heterosubstituted derivatives of said groups (i) to (v), (vii) hydrocarbyl-substituted heteroatom groups and (viii) further imidazole-containing groups. Alternatively R12 or R13 may be removed, for example by deprotonation when
they are hydrogen, to give a formally monoanionic fragment; or if both R or R are removed they provide a formally dianionic fragment. More preferably D is an amine of formula -N(R12)- as defined above. R12 is preferably hydrogen, an aliphatic hydrocarbon, an aromatic hydrocarbon or a further imidazole containing group. Preferably D2 is an imidazole-containing group. M is preferably a metal selected from Groups 3 to 11 of the periodic table, preferably from Groups 3 to 7, more preferably selected from Sc, Ti, Zr, Hf, V, Nb, Ta, Cr, Mo, W, Mn and most preferably V, Cr, Ti, Zr and Hf The anionic group X can be, for example, a halide, preferably chloride or bromide; or a hydrocarbyl group, for example, methyl, benzyl or phenyl; a carboxylate, for example, acetate or an acetylacetonate; an oxide; an amide, for example diethyl amide; an alkoxide, for example, methoxide, ethoxide or phenoxide; or a hydroxyl. Alternatively, X can be a non-coordinating or weakly-coordinating anion, for example, tetrafluoroborate, a fluorinated aryl borate or a triflate. The anionic groups X may be the same or different and may independently be monoanionic, dianionic or trianionic. The neutral donor group L can be, for example, a solvate molecule, for example an ether, for example, diethyl ether or THF; an amine, for example, diethyl amine, trimethylamine or pyridine; a phosphine, for example trimethyl phosphine or triphenyl phosphine; or water; or an olefin or a neutral, conjugated or nonconjugated diene, optionally substituted with one or more groups selected from hydrocarbyl or trimethylsilyl groups, said group having up to 40 carbon atoms and forming a pi- complex with M. When L is a diene ligand, it can be, for example s-trans-η4- 1,4- diphenyl- 1 ,3-butadiene; s-trans-η4-3-methyl- 1 ,3-pentadiene; s-trans-η4- 1 ,4-dibenzyl- 1,3- butadiene; s-trans-η4-2,4-hexadiene; s-trans-η4- 1,3-pentadiene; s-trans-η4- 1,4-ditolyl- 1 ,3-butadiene; s-trans-η4- 1 ,4-bis(trimethylsilyl)- 1 ,3-butadiene; s-trans-η4- 1 ,4-diphenyl- 1,3-butadiene; s-cis-η -3-methyl- 1,3-pentadiene; s-cis-η4-l,4-dibenzyl- 1,3-butadiene; s- cis-η4-2,4-hexadiene; s-cis-η4- 1,3-pentadiene; s-cis-η4-l,4-ditolyl-l,3-butadiene; or s- cis-η4-l,4-bis(trimethylsilyl)- 1,3-butadiene, said s-cis isomers forming a .pi.-bound diene complex; The value of y depends on the formal charge on each group Z and D, the charge on the anionic group X and the oxidation state of the metal M. For example, if M is chromium in oxidation state +3, Z is a neutral group and both D groups are neutral, then
y is 3 if X is a monoanionic group (e.g. chloride); if M is chromium in oxidation state +3, the Z group is neutral, one D group is monoanionic and the other D is neutral, then y is 2 if all X groups are monoanionic groups (e.g. chloride). The optional activator (2) for the catalyst employed in the process of the present invention is suitably a Lewis acid. Preferably the Lewis acid is selected from organoaluminium compounds and organoboron compounds or mixtures thereof. Examples of organoaluminium compounds include trialkyaluminium compounds, for example, trimethylaluminium, triethylaluminium, tributylaluminium, tri-n- octylaluminium, ethylaluminium dichloride, diethylaluminium chloride, tris(pentafluorophenyl)aluminium and alumoxanes. Alumoxanes are well known in the art as typically the oligomeric compounds which can be prepared by the controlled addition of water to an alkylaluminium compound, for example trimethylaluminium. Such compounds can be linear, cyclic or mixtures thereof. Commercially available alumoxanes are generally believed to be mixtures of linear, cyclic and cage compounds. The cyclic alumoxanes can be represented by the formula [RI6AlO]s and the linear 1 7 1 R alumoxanes by the formula R (R AlO)s wherein s is a number from about 2 to 50, and wherein R16, R17, and R18 represent hydrocarbyl groups, preferably d to C6 alkyl groups, for example methyl, ethyl or butyl groups. Examples of suitable organoboron compounds are dimethylphenylammόniumtetra(phenyl)borate, trityltetra(phenyl)borate, triphenylboron, dimethylphenylammonium tetra(pentafluorophenyl)borate, sodium tetrakis[(bis-3,5-trifluoromethyl)phenyl]borate, H+(OEt2)[(bis-3,5- trifluoromethyl)phenyl] borate, trityltetra(pentafluorophenyl)borate and tris(pentafluorophenyl) boron. Mixtures of organoaluminium compounds and organoboron compounds may be used. Another type of Lewis acid that can be employed as the activator in the polymerisation process of the present invention is at least on halogen compound selected from halogenated magnesium compounds, halogenated manganese compounds, halogenated iron compounds, halogenated cobalt compounds and halogenated nickel compounds. Preferred amongst such compounds are the dichlorides. In the preparation of the catalysts employed in the process of the present invention the quantity of activating compound selected from organoaluminium
compounds and organoboron compounds to be employed is easily determined by simple testing, for example, by the preparation of small test samples which can be used to polymerise small quantities of the monomer(s) and thus to determine the activity of the produced catalyst. It is generally found that the quantity employed is sufficient to provide 0.1 to 20,000 atoms, preferably 1 to 2000 atoms of aluminium or boron per atom of M present in the compound of Formula A. Mixtures of different activating compounds may be used. EP 1238989 discloses the use of activators (Lewis acids) selected from (b-1) ionic-bonding compounds having a CdCl2 type or a Cdl2 type of layered crystal structure; (b-2) clays, clay minerals, or ion-exchange layered compounds; (b-3) heteropoly-compounds; and (b-4) halogenated lanthanoid compounds. The activator employed in the process of the present invention may be of the type disclosed in EP 1238989 if desired. Such Lewis acids are those compounds which capable of receiving at least one electron pair and is capable of forming an ion pair by reaction with the transition metal complex. The Lewis acid includes the aforementioned (b-1) ionic-bonding compounds having a layered crystal structure of a CdCl2 type or Cdl2 type (b-2) clay . clay minerals, or ion-exchange layered compounds, (b-3) heteropoly compounds, and (b-4) halogenated lanthanoid compounds. The Lewis acid further includes SiO2, Al2O3, natural and synthetic zeolites which have Lewis acid points formed by heating or a like treatment, and complexes and mixtures thereof. US Patent 6399535 discloses a coordinating catalyst system capable of polymerizing olefins comprising: (I) as a pre-catalyst, at least one non-metallocene, non-constrained geometry, bidentate ligand containing transition metal compound or tridentate ligand containing transition metal compound capable of (A) being activated upon contact with the catalyst support- activator agglomerate of (II) or (B) being converted, upon contact with an organometallic compound, to an intermediate capable of being activated upon contact with the catalyst support-activator agglomerate of (II), wherein the transition metal is at least one member selected from Groups 3 to 10 of the Periodic table; in intimate contact with (II) catalyst support-activator agglomerate comprising a composite of (A) at least
one inorganic oxide component selected from SiO2, Al2O3, MgO, AlPO4, TiO2, ZrO2, and Cr2O and (B) at least one ion containing layered material having interspaces between the layers and sufficient Lewis acidity, when present within the catalyst support-activator agglomerate, to activate the pre-catalyst when the pre-catalyst is in contact with the catalyst support-activator agglomerate, said layered material having a cationic component and an anionic component, wherein said cationic component is present within the interspaces of the layered material, said layered material being intimately associated with said inorganic oxide component within the agglomerate in an amount sufficient to improve the activity of the coordinating catalyst system for polymerizing ethylene monomer, expressed as Kg of polyethylene per gram of catalyst system per hour, relative to the activity of a corresponding catalyst system employing the same pre-catalyst but in the absence of either Component A or B of the catalyst support-activator agglomerate; wherein the amounts of the pre-catalyst and catalyst support-activator agglomerate which are in intimate contact are sufficient to provide a ratio of micromoles of pre-catalyst to grams of catalyst support-activator agglomerate of from about 5: 1 to about 500: 1. The layered material can be, for example, a smectite clay. The catalyst system employed in the process of the present invention can be employed with a catalyst support-activator agglomerate as described in US 6399535 if desired. In addition to the activator compound, it can be advantageous to employ catalytic quantities of certain halogenated compounds that are capable of promoting catalyst activity. Promotors of this type are especially useful in the case that the transition metal in the complex is vanadium. US Patent.5191042 discloses that certain vanadium-based catalysts activated with organoaluminium compounds can be promoted using a variety of halogenated organic compounds, for example, carbon tetrachloride, hexachloroethylene, benzylbromide, benzylchloride and 2,3- or 1,3-dichloropropylene. Other examples of halogenated organic compounds that can be used in this manner are ethyl trichloroacetate, chloroform (CHC13) and n-butylchloride. US Patent.5191042 also refers to the disclosure of Cooper (T. A Cooper, Journ. Am. Chem. Soc, 4158 (1973), which defines in Table 1 an organic halide activity index based on the ability of the halide to oxidize certain vanadium compounds under standard conditions. For example, carbon tetrachloride is assigned a reactivity of 1 in tetrahydrofuran at 20 °C,
and other listed halogenated organic compounds have reactivities of from about 0.02 to greater than 200 relative to carbon tetrachloride. When it is desired to use a halogenated promotor, it is preferred to use those having a Cooper Index ranging from about 0.01 up to about 30. The use of such promoters, especially in combination with vanadium-based catalysts is generally well known in the art, and for details of use of the such promoters reference may be made to US Patent.5191042 and to other prior art in this field. In the present invention it is possible to employ any halogenated organic compound as a promoter, but the compounds mentioned above are preferred. In a preferred embodiment of the process of the present invention the catalyst comprises
(1) a transition metal compound having the following Formula B or C, and optionally
(2) an activating quantity of a suitable activator,
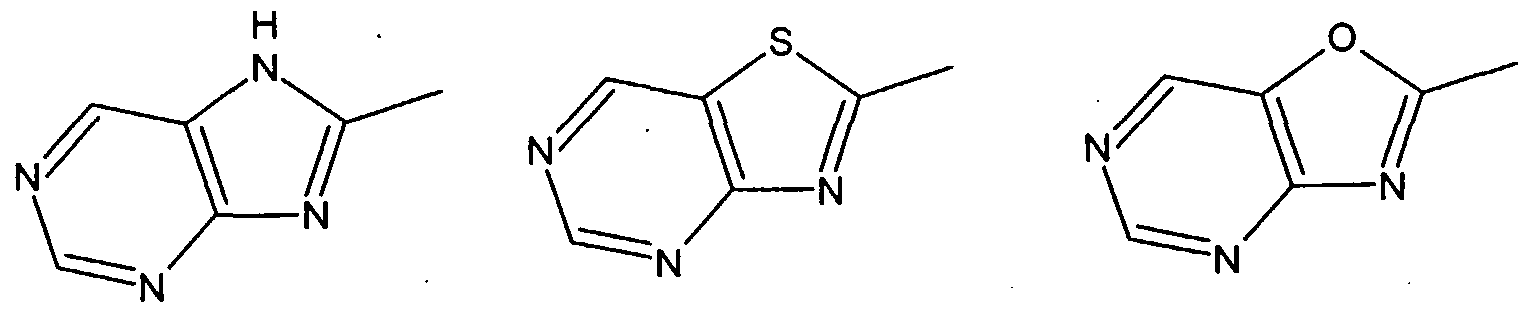
wherein the imidazole nucleus shown within the dotted circle is selected from the divalent groups represented by the Formulae la, Ila, Ilia, IVa, Va and Via,
wherein M is a metal from Group 3 to 11 of the Periodic Table or a lanthanide metal; E and E
2 are divalent groups independently selected from (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups and (vi) heterosubstituted derivatives of said groups (i) to (v); D
1 and D
2 are donor groups; X is an anionic group, L is a neutral donor group; n = m = zero or 1 ; y and z are independently zero or integers such that the number of X and L groups satisfy the valency and oxidation state of the metal M, 9 1 1 wherein the groups R to R are independently hydrogen or a monovalent (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups, (vi) heterosubstituted derivatives of said groups (i) to (v), and (vii) hydrocarbyl-substituted heteroatom groups. M is preferably selected from Groups 3 to 7 of the periodic table. 9 1 1 Groups R to R are preferably selected from the groups defined above in relation to the Formula I, II, III, IV, V and VI groups. In this preferred embodiment of the process of the present invention, D
1 and D
2 can be the same or different donor groups, for example oxygen, sulfur, an amine, an 1 9 imine or a phosphine. Preferably D and D are selected from oxygen, sulfur, an amine of formula -N(R
12)- or a phosphine of formula -P(R
13)- wherein R
12 and R
13 are hydrogen or (i) aliphatic hydrocarbon, (ii) alicyclic hydrocarbon, (iii) aromatic hydrocarbon, (iv) alkyl substituted aromatic hydrocarbon (v) heterocyclic groups, (vi) heterosubstituted derivatives of said groups (i) to (v), (vii) hydrocarbyl-substituted heteroatom groups and (viii) further imidazole-containing groups. Preferably D
1 is nitrogen for example -NR
1 - or =N- or a nitrogen-containing 1 9Ω 1 0 group, for example -N(R )-R - wherein R represents a monovalent group and R represents a divalent group derived from, for example, aliphatic hydrocarbon groups
such as methyl, ethyl, ethylenyl, butyl, hexyl, isopropyl and tert-butyl. Examples of suitable alicyclic hydrocarbon groups are adamantyl, norbornyl, cyclopentyl and cyclohexyl. Examples of suitable aromatic hydrocarbon groups are phenyl, biphenyl, naphthyl, phenanthryl and anthryl. Examples of suitable alkyl substituted aromatic hydrocarbon groups are benzyl, tolyl, mesityl, 2,6-diisopropylphenyl and 2,4,6- triisopropyl. Examples of suitable heterocyclic groups are 2-pyridinyl, 3-pyridinyl, 2- thiophenyl, 2-furanyl, 2-pyrrolyl, 2-quinolinyl. Suitable substituents for forming heterosubstituted derivatives of said groups R
1 to R
1 ' are, for example, chloro, bromo, fluoro, iodo, nitro, amino, cyano, ether, hydroxyl and silyl, methoxy, ethoxy, phenoxy (i.e. -OC
6H
5), tolyloxy (i.e. -OC
6H (CH
3)), xylyloxy, mesityloxy, dimethylamino, diethylamino, methylethylamino, thiomethyl,,thiophenyl and trimethylsilyl. Examples of suitable heterosubstituted derivatives of said groups (i) to (v) are 2-chloroethyl, 2- bromocyclohexyl, 2-nitrophenyl, 4-ethoxyphenyl, 4-chloro-2-pyridinyl, 4- dimethylaminophenyl and 4-methylaminophenyl. Examples of suitable hydrocarbyl- substituted heteroatom groups are chloro, bromo, fluoro, iodo, nitro, amino, cyano, ether, hydroxyl and silyl, methoxy, ethoxy, phenoxy (i.e. -OC
6H
5), tolyloxy (i.e. - OC
6H
4(CH
3)), xylyloxy, mesityloxy, dimethylamino, diethylamino, methylethylamino, thiomethyl, thiophenyl and trimethylsilyl. Any of the substituents R
1 to R
11 may be 9 1 1 linked to form cyclic structures. Substituents R to R may also suitably be inorganic groups such as fluoro, chloro, bromo, iodo, nitro, amino, cyano and hydroxyl Preferably D is a imidazole group selected from the groups of Formula la, Ila and Ilia above. As indicated above, the values of m and n in the preferred catalyst employed in the process of the present invention are such that m = n = zero or one. For the avoidance of doubt, this means that for a given complex, when m is zero, n is also zero. And when m is 1 , n is also 1. When m and n are zero in Formula A, the Formula reduces to Formula D, preferably to Formula E or Formula F
wherein Dl, El, Z, M, X, L, y and z are as defined above, and wherein the imidazole nucleus within the dotted circle is selected from the divalent groups represented by the Formulae la, Ila, Ilia, IVa, Va and Via
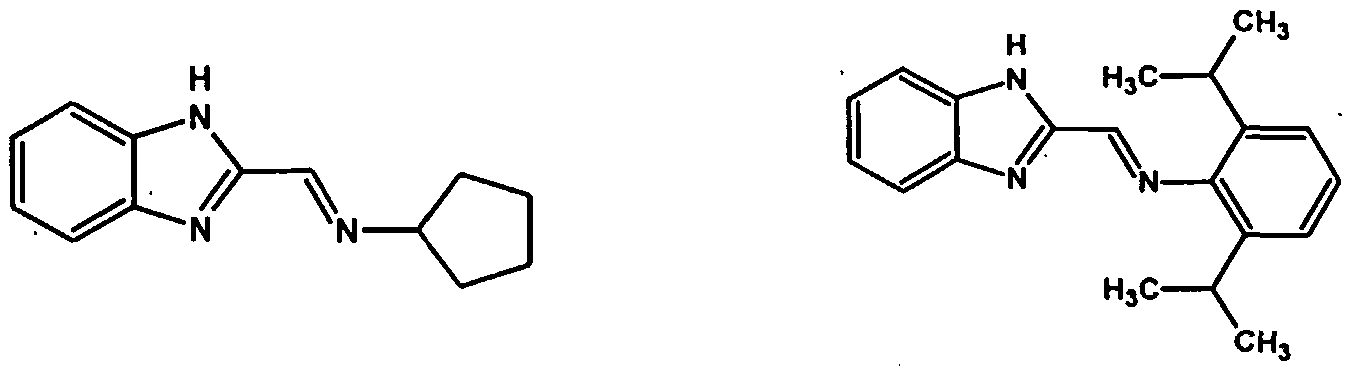
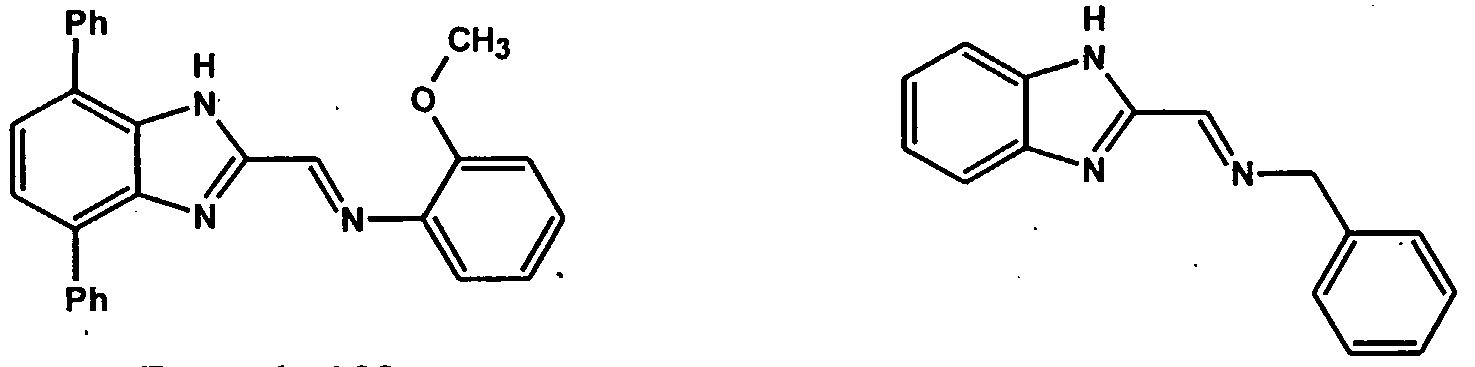
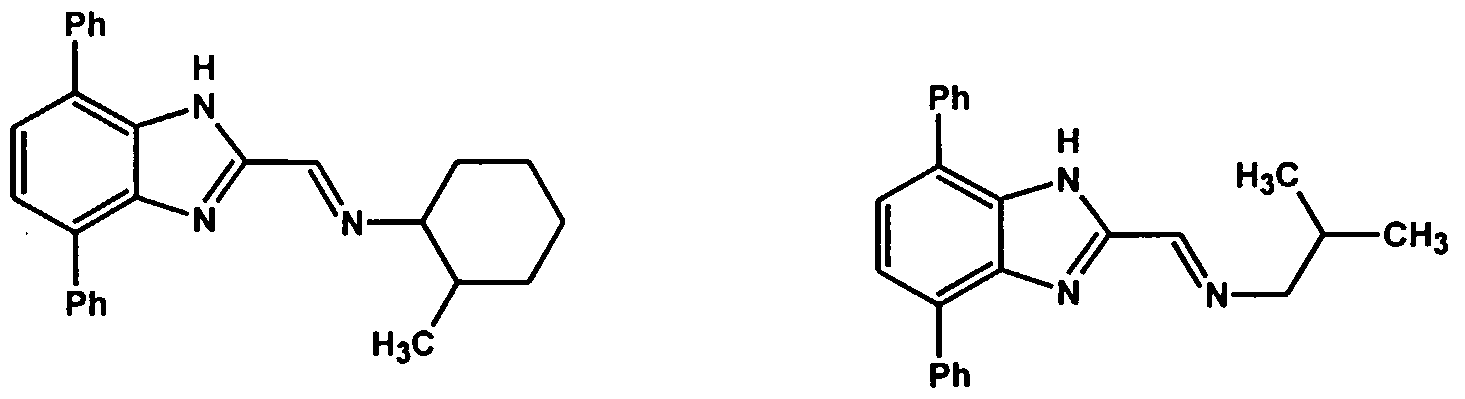
The following ligands represent some examples of those suitable for making the complexes of Formula C and D which can be employed in the process of the present invention.
Formula 34 Formula 35
Formula 36
Formula 50
Formula 52 Formula 53
Formula 58 Formula 59
Formula 61 Formula 60
Formula 66 Formula 67
Formula 68 Formula 69
Formula ό 74 ormula 75
Formula 76 Formula 77
Formula 82 Formula 83
Formula 84 Formula 85
Formula 90 Formula 91
Formula 92 Formula 93
F Formula 106 Formula 107
Formula 109 Formula 108
Formula 114 Formula 115
Formula 117 Formula 1 16
Ph Formula 121 Formula 120
Formula 122 Formula 123
Formula 124 Formula 125
Formula 130 Formula 131
Formula 132 Formula 133
Formula 138 Formula 139
Formula 140 Formula 141
Formula 147 Formula 146
Formula 148 Formula 149
Formula 155
Formula 157 Formula 156
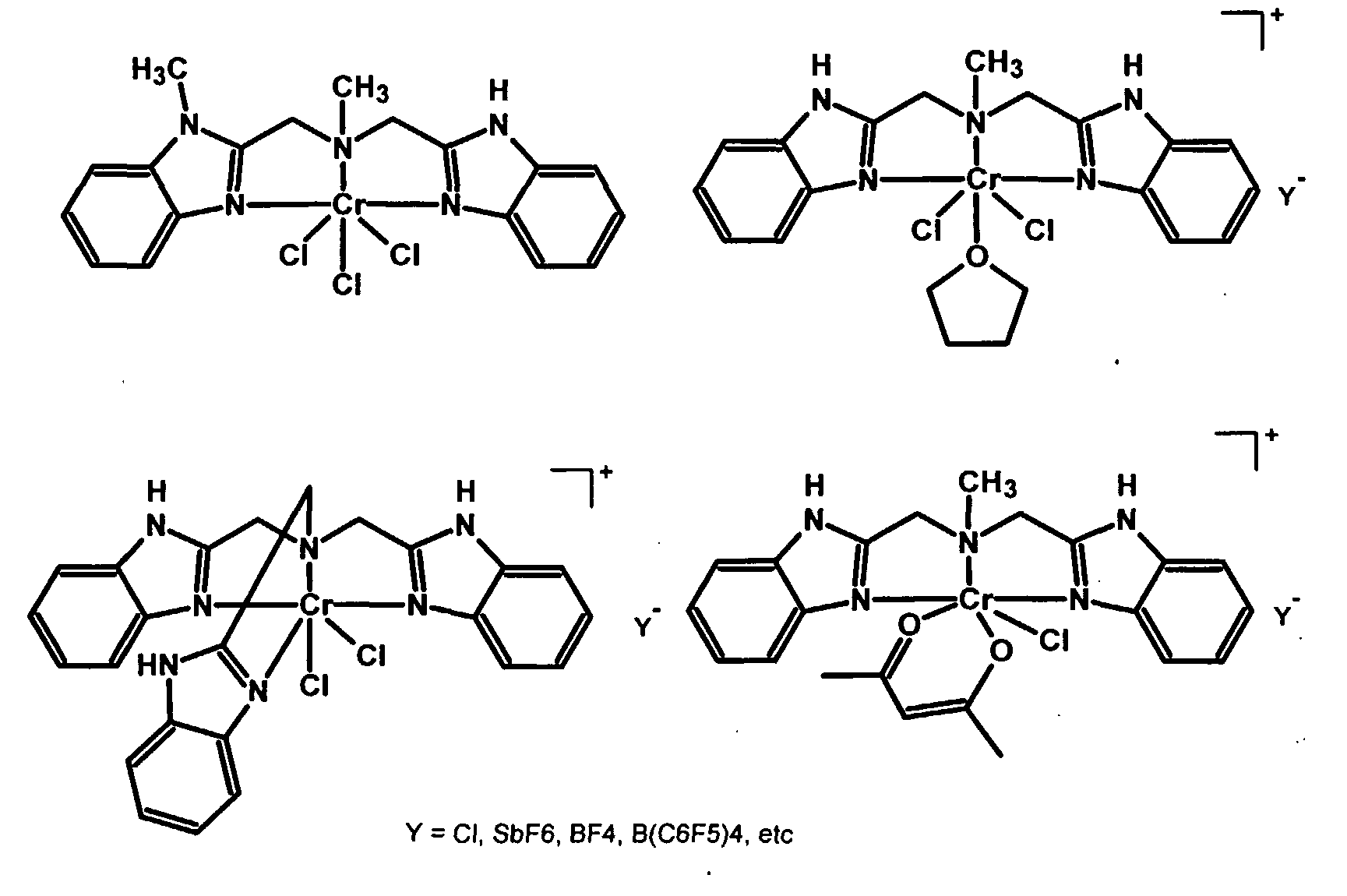
These ligands can be used to make complexes and catalysts suitable for use in the process of the present invention wherein the transition metal is preferably titanium, zirconium, hafnium, vanadium or chromium. The following are examples of transition metal complexes that can be employed as catalyst in the process of the present invention:
The following Formulae illustrate the transition metal compounds according to the present invention wherein L is a diene:
R1 - R4 = alkyl, aryl, etc. Z = divalent organic or inorganic radical as -CH2-, -0-,
Z = divalent organic or inorganic radical as -CH2-, -0-, etc. X = CI, Br, I, NMe2, OR, SR, etc.
The catalysts employed in the process of the present invention can, if desired, be utilised on a support material. Suitable support materials are, for example, silica, alumina, or zirconia, magnesia, magnesium chloride or a polymer or prepolymer, for example polyethylene, polystyrene, or poly(aminostyrene). The catalysts can if desired comprise more than one of the defined transition metal compounds. In addition to said one or more defined transition metal compounds, the catalysts can also include one or more other catalysts for polymerising 1 -olefins. Preferably such catalysts are other types of transition metal compounds or catalysts, for example, transition metal compounds of the type used in conventional Ziegler-Natta catalyst systems, metallocene-based catalysts, or heat activated supported chromium oxide catalysts (e.g. Phillips-type catalyst). The catalysts used in the process of the present invention may also used in conjunction with other catalysts producing only 1 -olefins, either inside or outside the polymerisation reactor, and in this way make copolymers of ethylene or propylene and these 1 -olefins. Suitable catalysts for producing 1 -olefins may produce only 1 -butene, only 1-hexene or a distribution (for example, a Schulz- Flory distribution) of 1 -olefins. If desired the catalysts can be formed in situ in the presence of the support material, or the support material can be pre-impregnated or premixed, simultaneously or sequentially, with one or more of the catalyst components. The catalysts can if desired be supported on a heterogeneous catalyst, for example, a magnesium halide supported Ziegler Natta catalyst, a Phillips type (chromium oxide) supported catalyst or a supported metallocene catalyst. Formation of the supported catalyst can be achieved for example by treating the transition metal compounds described above with alumoxane in a suitable inert diluent, for example a volatile hydrocarbon, slurrying a particulate support material with the product and evaporating the volatile diluent. The produced supported catalyst is preferably in the form of a free-flowing powder. The quantity of support material employed can vary widely, for example from 100,000 to 1 grams per gram of metal present in the transition metal compound. The quantity of the monomeric (a) ethylene, (b) acyclic 1-olefin containing 3 to
20 carbon atoms and (c) cyclic olefin containing at least one endocyclic double bond, employed in the process of the present invention to provide the desired copolymer can
be determined by simple experiment. Generally the ethylene will exhibit the highest relative reactivity and so the molar concentration of ethylene in the polymerisation reaction mixture will, in general, be lower than the molar proportion required in the final copolymer. On the other hand, the defined cyclic olefin will, in general exhibit a relatively low reactivity and hence will normally require a higher molar concentration in the polymerisation reaction than that desired in the final copolymer. The polymerisation conditions can be, for example, bulk phase, solution phase, slurry phase or gas phase. If desired, the monomers can be copolymerised under high pressure/high temperature process conditions wherein the polymeric material forms as a melt in supercritical ethylene. However, it is preferred to carry out the copolymerisation under relatively mild conditions, e.g. at a temperature in thee range 50 to 120°C under a pressure of 0.5 to 50 bars. Preferably the polymerisation is conducted under gas phase fluidised or stirred bed conditions. Slurry phase polymerisation conditions or gas phase polymerisation conditions are particularly useful for the production of high-density grades of polyethylene. In these processes the polymerisation conditions can be batch, continuous or semi- continuous. In the slurry phase process and the gas phase process, the catalyst is generally fed to the polymerisation zone in the form of a particulate solid. This solid can be, for example, an undiluted solid catalyst system formed from the complex A and an activator, or can be the solid complex A alone. In the latter situation, the activator can be fed to the polymerisation zone, for example as a solution, separately from or together with the solid complex. Preferably the catalyst system or the transition metal complex component of the catalyst system employed in the slurry polymerisation and gas phase polymerisation is supported on a support material. Most preferably the catalyst system is supported on a support material prior to its introduction into the polymerisation zone. Suitable support materials are, for example, silica, alumina, zirconia, talc, kieselguhr, magnesia, magnesium chloride and polymers. Impregnation of the support material can be carried out by conventional techniques, for example, by forming a solution or suspension of the catalyst components in a suitable diluent or solvent, and slurrying the support material therewith. The support material thus impregnated with catalyst can then be separated from the diluent for example, by filtration or evaporation techniques.
In the slurry phase polymerisation process the solid particles of catalyst, or supported catalyst, are fed to a polymerisation zone either as dry powder or as a slurry in the polymerisation diluent. Preferably the particles are fed to a polymerisation zone as a suspension in the polymerisation diluent. The polymerisation zone can be, for example, an autoclave or similar reaction vessel, or a continuous loop reactor, e.g. of the type well know in the manufacture of polyethylene. The loop processes can be, for example, processes wherein a slurry of polymer suspended in a suitable diluent, for example, isobutane or hexane, is pumped continuously around a single or multiple loop reactor in the presence of monomer and catalyst under conditions such that polymer forms as particles suspended in the diluent. When the polymerisation process of the present invention is carried out under slurry conditions the polymerisation is preferably carried out at a temperature above 0°C, most preferably above 15°C. The polymerisation temperature is preferably maintained below the temperature at which the polymer commences to soften or sinter in the presence of the polymerisation diluent. If the temperature is allowed to go above the latter temperature, fouling of the reactor can occur. Adjustment of the polymerisation within these defined temperature ranges can provide a useful means of controlling the average molecular weight of the produced polymer. A further useful means of controlling the molecular weight is to conduct the polymerisation in the presence of hydrogen gas which acts as chain transfer agent. Generally, the higher the concentration of hydrogen employed, the lower the average molecular weight of the produced polymer. The use of hydrogen gas as a means of controlling the average molecular weight of the polymer or copolymer applies generally to the polymerisation process of the present invention. For example, hydrogen can be used to reduce the average molecular weight of polymers or copolymers prepared using gas phase, slurry phase or solution phase polymerisation conditions. The quantity of hydrogen gas to be employed to give the desired average molecular weight can be determined by simple "trial and error" polymerisation tests. Methods for operating gas phase polymerisation processes are well known in the art. Such methods generally involve agitating (e.g. by stirring, vibrating or fluidising) a bed of catalyst, or a bed of the target polymer (i.e. polymer having the same or similar physical properties to that which it is desired to make in the polymerisation process)
containing a catalyst, and feeding thereto a stream of monomer at least partially in the gaseous phase, under conditions such that at least part of the monomer polymerises in contact with the catalyst in the bed. The bed is generally cooled by the addition of cool gas (e.g. recycled gaseous monomer) and/or volatile liquid (e.g. a volatile inert hydrocarbon, or gaseous monomer which has been condensed to form a liquid). The polymer produced in, and isolated from, gas phase processes forms directly a solid in the polymerisation zone and is free from, or substantially free from liquid. As is well known to those skilled in the art, if any liquid is allowed to enter the polymerisation zone of a gas phase polymerisation process the quantity of liquid is small in relation to the quantity of polymer present in the polymerisation zone. This is in contrast to
"solution phase" processes wherein the polymer is formed dissolved in a solvent, and "slurry phase" processes wherein the polymer forms as a suspension in a liquid diluent. The gas phase process can be operated under batch, semi-batch, or so-called "continuous" conditions. It is preferred to operate under conditions such that monomer is continuously recycled to an agitated polymerisation zone containing polymerisation catalyst, make-up monomer being provided to replace polymerised monomer, and continuously or intermittently withdrawing produced polymer from the polymerisation zone at a rate comparable to the rate of formation of the polymer, fresh catalyst being added to the polymerisation zone to replace the catalyst withdrawn form the polymerisation zone with the produced polymer. When using the process of the present invention is carried out under gas phase polymerisation conditions, the catalyst, or one or more of the components employed to form the catalyst can, for example, be introduced into the polymerisation reaction zone in liquid form, for example, as a solution in an inert liquid diluent. Thus, for example, the transition metal component, or the activator component, or both of these components can be dissolved or slurried in a liquid diluent and fed to the polymerisation zone. Under these circumstances it is preferred the liquid containing the component(s) is sprayed as fine droplets into the polymerisation zone. The droplet diameter is preferably within the range 1 to 1000 microns. EP-A-0593083, the teaching of which is hereby incorporated into this specification, discloses a process for introducing a polymerisation catalyst into a gas phase polymerisation. The methods disclosed in EP- A-0593083 can be suitably employed in the polymerisation process of the present
invention if desired. The invention is further illustrated with reference to the following Examples. In the Examples all manipulations of air/moisture-sensitive materials were performed on a conventional vacuum/inert atmosphere (nitrogen) line using standard Schlenk line techniques, or in an inert atmosphere glove box. Examples 1 - 4
Preparation of [N,N-bis(lH-benzimidazol-2-ylmethyl)-N- methylamine]dipropoxyoxovanadium (V-2)
A cooled to -78 C solution of 0.42 g (1.72 mmol) tripropoxyoxovanadium in THF
(10 ml) was added to a cold (-78°C) slurry of [N,N-bis(lH-benzimidazol-2-ylmethyl)-N- methylamine] in THF (20 ml). The mixture was allowed to warm up to room temperature and stirred for 30 min. The orange-red solution was filtered to remove any traces of insoluble materials (ligand) and the filtrate evaporated to about 5 ml. Addition of pentane (50 ml) resulted in the formation of an orange solid which was filtered, washed with pentane (2 x 5 ml) and dried under reduced pressure. Yield of V-2 = 0.72g (88.0%). Microanalysis, %: Calculated for C23H30Ν5O3V: C 58.10, H 6.36, N 14.73. Found: C 57.93, H 6.26, N 14.77. 'H NMR (250 MHz, D2-DCM), δ: 0.85 (t, JHH = 14.7 Hz 6H), 1.62 (m, 4H), 2.85 (s, 3H), 3.08 (d, JHH = 15.9 Hz 2H), 4.02 (d, JHH= 16.2 Hz, 2H), 4.88 (dt, JHH= 13.4 Hz, JHH = 11.6 Hz, 2H), 5.34 (dt, JHH = 12.5 Hz, JHH= 11-3 Hz,
2H), 7.14 (m, 4H), 7.42 (m , 2H), 8.05 (m, 2H). 51V NMR [131 MHz, V(O)Cl3, d2- DCM], δ, ppm: -560.2. Ethylene terpolvmerisation with V-2 The ethylene copolymerisation tests were carried out using the following procedure. The required amounts of comonomers were preloaded in the polymerisation reactor. The ethylene polymerisation reactions in Examples 1 - 3 were carried out either in a 400 ml "Fischer-Porter" glass reactor (FPR) equipped with a gas inlet, a catalyst inlet, a mechanical stirrer and a digital thermometer. In Example 4 the copolymerisation test was carried out in a 1 litre stainless-steel reactor (SSR) equipped with an integral system for control of reaction temperature, ethylene pressure and ethylene flow. An aliquot of the catalyst solution (1-5 ml) was injected in the reactor containing 200 - 300 (FPR) or 400- 800 (SSR) ml solvent (usually toluene, n-hexane, n-heptane or isobutane), 0.1 - 2 mmol scavenger (usually DMAC) and 10 -60 μmol reactivator (ethyl trichloroacetate - "ETA" ). The reactor was then connected to the ethylene gas supply at the desired pressure and the temperature brought quickly to the required value. The reaction was carried out for 10 - 120 min (usually 60 min). The reaction mixture was then cooled down to room temperature (if necessary) and the reaction terminated by venting the ethylene off. If toluene was used as a polymerisation medium, the reactor content was then poured into a beaker containing 400 ml methanol and a few drops of 2M HC1. The polymer was filtered, washed with methanol (if necessary) and dried at 60°C under vacuum. Table 1 - Polymerisation in toluene. Fisher-Porter glass reactor.
Table 2 - Polymer properties
Notes on Tables 1 - 4 # 1. ECA = ethyl trichloroacetate - C2H5OC(O)CCl3 #2. nb = norbornene (2,2,l-bicyclohept-2-ene); hx = 1-hexene #3. Activity measured in units of; g mmol'1 h"1 bar-1#4 DMAC = Me2AlCl
Examples 5 Preparation of {N-[(lE)-lH-benzimidazol-2-ylmethylene]-N-[2- (trifluoromethyl)benzy 1] amine } tetra-hydrofuranotrichloro vanadium (III) (V-4)
A mixture of 0.40 (1.3 mmol) g N-[(lE)-lH-benzimidazol-2-ylmethylene]-N-[2- (trifluoromethyl)-benzyl] amine and 0.52 g (1.3 mmol) tris(tetrahydro-furan)vanadium trichloride in 40 ml THF was stirred at room temperature for 3 hours. The volume of the reaction mixture was then reduced to approximately 10 ml and 70 ml pentane were added. The formed precipitate was filtered, washed with 3 x 5 ml pentane and dried under reduced pressure. Yield of V-4 = 0.62 g (89.6%). Ethylene copolymerisation using V-4 The ethylene copolymerisation tests were carried out using the "Fischer-Porter procedure similar to that described in the Examples above. The required amounts of comonomers were preloaded in the polymerisation reactor.
Table 5 - Polymerisation in toluene. Fisher-Porter glass reactor.
Notes on Table 5 # 1. ECA = ethyl trichloroacetate - C
2H
5OC(O)CCl
3 #2. nb = norbornene (2,2,l-bicyclohept-2-ene); hx = 1-hexene #3. Activity measured in units of; g mmol
"1 h
"1 bar
_1#4 DMAC = Me
2AlCl
Comparative Example 6 and Example 7 Preparation of supported V-2 catalyst
Decane (45ml) and ethyl-2-hexanol (11.5 ml) were added to MgCl2 (2.333g) under an inert nitrogen atmosphere. The reaction mixture was heated to 140°C for 4 h. To the resulting solution were added dry toluene (188.5ml) to obtain a MgCl2 concentration of 0.1M. This solution (62.2 ml) was added to a flask containing dry toluene (3.8ml). The solution was heated to 75°C. At this temperature, triethylaluminium solution (18.9 ml, 1M in toluene) was added. After 5 minutes the resulting slurry was allowed to cool to ambient temperature. In a separate flask, V-2 in toluene (12.6 ml of 1 mM solution) was pre-contacted with of DMAC (2.52 ml of 1M solution in toluene) and then added to the MgCl2 slurry, which resulted in the formation of the supported V-2 catalyst.
Ethylene co- and terpolymerisation using MgCl? supported V-2
A 5 litre autoclave was charged with the 50 ml of the above catalyst slurry, followed by DMAC, norbornene and 2 litres of isobutane. The reactor contents were heated to 80°C and ethylene, hydrogen and optionally 1-hexene were added. This was followed by the introduction of ECA. The polymerisation reaction was terminated after 60 minutes by degassing the autoclave.
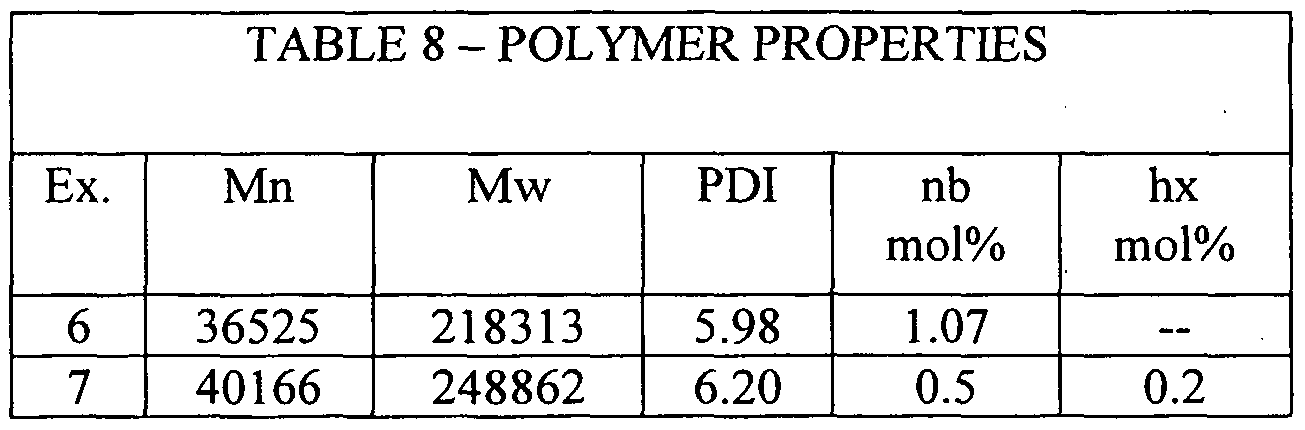
Table 7 - Polymerisation in isobutane. 5 litre autoclave The polymerisation tests (Examples 6 and 7) were conducted using 6.3 μmol catalyst, 11.2 mmol DMAC and 6.3 mmol ethyl trichloroacetate under a pressure of 10 bars ethylene. The polymerisation was terminated after 1 hour. The results are shown in Table 7.
Notes on Table 7 # 1. nb = norbomene (2,2, 1 -bicyclohept-2-ene); hx = 1 -hexene #2. Activity measured in units of; g mmol-1 h-1 bar-1
Example 6 illustrates a copolymer containing only ethylene and norbomene and is thus provided by way of comparison