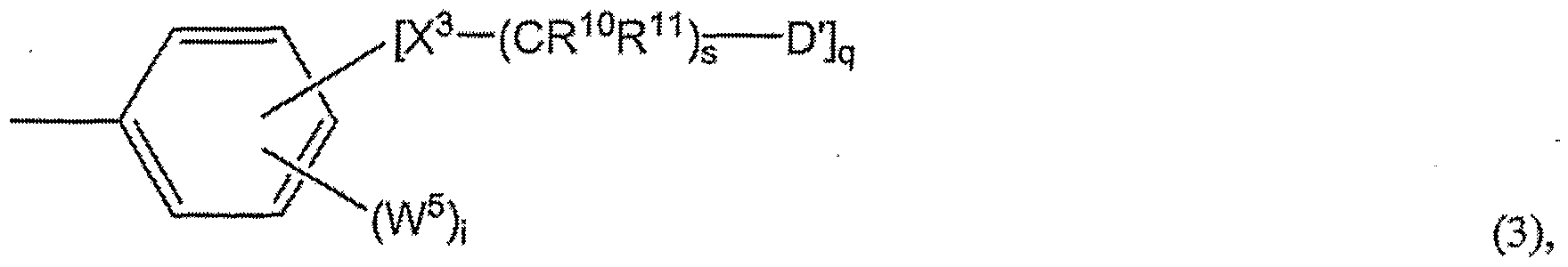WO2006012140A1 - Carboranylporphyrins and uses thereof - Google Patents
Carboranylporphyrins and uses thereof Download PDFInfo
- Publication number
- WO2006012140A1 WO2006012140A1 PCT/US2005/022061 US2005022061W WO2006012140A1 WO 2006012140 A1 WO2006012140 A1 WO 2006012140A1 US 2005022061 W US2005022061 W US 2005022061W WO 2006012140 A1 WO2006012140 A1 WO 2006012140A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound according
- subject
- meta
- carborane
- independently
- Prior art date
Links
- 0 *c1ccccc1 Chemical compound *c1ccccc1 0.000 description 1
- YXFVVABEGXRONW-UHFFFAOYSA-N Cc1ccccc1 Chemical compound Cc1ccccc1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F5/00—Compounds containing elements of Groups 3 or 13 of the Periodic System
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/22—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains four or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K41/00—Medicinal preparations obtained by treating materials with wave energy or particle radiation ; Therapies using these preparations
- A61K41/0057—Photodynamic therapy with a photosensitizer, i.e. agent able to produce reactive oxygen species upon exposure to light or radiation, e.g. UV or visible light; photocleavage of nucleic acids with an agent
- A61K41/0071—PDT with porphyrins having exactly 20 ring atoms, i.e. based on the non-expanded tetrapyrrolic ring system, e.g. bacteriochlorin, chlorin-e6, or phthalocyanines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K41/00—Medicinal preparations obtained by treating materials with wave energy or particle radiation ; Therapies using these preparations
- A61K41/009—Neutron capture therapy, e.g. using uranium or non-boron material
- A61K41/0095—Boron neutron capture therapy, i.e. BNCT, e.g. using boronated porphyrins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/06—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations
- A61K49/08—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations characterised by the carrier
- A61K49/10—Organic compounds
- A61K49/101—Organic compounds the carrier being a complex-forming compound able to form MRI-active complexes with paramagnetic metals
- A61K49/106—Organic compounds the carrier being a complex-forming compound able to form MRI-active complexes with paramagnetic metals the complex-forming compound being cyclic, e.g. DOTA
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/0474—Organic compounds complexes or complex-forming compounds, i.e. wherein a radioactive metal (e.g. 111In3+) is complexed or chelated by, e.g. a N2S2, N3S, NS3, N4 chelating group
- A61K51/0485—Porphyrins, texaphyrins wherein the nitrogen atoms forming the central ring system complex the radioactive metal
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F1/00—Compounds containing elements of Groups 1 or 11 of the Periodic System
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F1/00—Compounds containing elements of Groups 1 or 11 of the Periodic System
- C07F1/08—Copper compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F3/00—Compounds containing elements of Groups 2 or 12 of the Periodic System
Definitions
- Porphyrins in general belong to a class of colored, aromatic tetrapyrrole compounds, some of which are found naturally in plants and animals, e.g., chlorophyll and heme, respectively.
- Porphyrins and other tetrapyrroles with relatively long singlet lifetimes have already been used to treat malignant tumors using photodynamic therapy (PDT).
- PDT photodynamic therapy
- the patient is first injected with a photosensitizing drug, typically a porphyrin.
- the tumor cells now photosensitized, are susceptible to destruction when exposed to an intense beam of laser red light.
- the biochemical mechanism of cell damage in PDT is believed to be mediated largely by singlet oxygen, which is produced by transfer of energy from the light-excited porphyrin molecule to an oxygen molecule.
- PDT has been limited predominantly by the photosensitizing compounds, which have lower than adequate selectivity to tumor cells and higher than optimal toxicity to normal tissue.
- BNCT boron neutron-capture therapy
- boron-10 a stable nuclide of boron known as boron-10, or 10 B
- the thermalized neutrons impinge on the boron- 10, causing a nuclear fission (decay reaction).
- the nuclear fission reaction causes the highly localized release of vast amounts of energy in the form of high linear-energy- transfer (LET) radiation, which can kill cells more efficiently (higher relative biological effect) than low LET radiation, such as x-rays.
- LET linear-energy- transfer
- the boron-containing compound In BNCT, the boron-containing compound must be non-toxic or of low toxicity when administered in therapeutically effective amounts, as well as being capable of selectively accumulating in cancerous tissue.
- clinical BNCT for malignant brain tumors was carried out at the Brookhaven National Laboratory Medical Department using p-boronophenylalanine (BPA) as the boron carrier (Chanana et al., Neurosurgery, 44, 1182-1192, 1999).
- BPA p-boronophenylalanine
- BPA has the advantage of low chemical .toxicity, it accumulates in critical normal tissues at levels that are less than desirable.
- the tumor-to-normal brain and tumor-to-blood boron concentrations are in the ratio of approximately 3:1.
- Such low specificity limits the maximum dose of BPA to a tumor since the allowable dose to normal tissue will be the limiting factor.
- TPPs Tetraphenylporphyrins
- TPPs can be controlled by the substituents, generally on the phenyl positions.
- Those TPPs containing sulfonates or carboxylates are water-soluble.
- some of the carborane-containing TPPs have high lipophilic properties, which can require high amounts of non-aqueous excipients before administration into animals.
- High amounts of excipients may reduce the biological effect of the porphyrin by, for example, changing the microlocalization within the tumor cell such that it may be bound to membranes instead of being homogeneously distributed throughout the cell.
- hydrophilic bonds such as amide, ester, or urea bonds, although significantly more hydrophilic than carbon-carbon linkages, are known to hydrolyze under numerous types of conditions. Such hydrolysis is particularly problematic when such hydrophilic bonds are employed to attach the carboranyl group to the porphyrin molecule, since hydrolysis results in loss of the carbonyl group before reaching the target.
- Porphyrins also have the advantage of having the ability to chelate metal ions in its interior. Such chelated porphyrins can additionally function as visualization tools for real-time monitoring of porphyrin concentration and/or diagnostic agents. For example, when chelated to paramagnetic metal ions, porphyrins may function as contrast agents in magnetic resonance imaging (MRI), and when chelated to radioactive metal ions, porphyrins may function as imaging agents for single photon emission computed tomography (SPECT) or positron emission tomography (PET).
- MRI magnetic resonance imaging
- SPECT single photon emission computed tomography
- PET positron emission tomography
- boron concentration and distribution in and around the tumor and all tissues within the irradiated treatment volume can be accurately and rapidly determined noninvasively before and during the irradiation.
- diagnostic information allows BNCT treatment to be performed more quickly, accurately, and safely, by lowering exposures of epithermal neutrons in regions of tissues known to contain high levels of boron. Short irradiations would obviate the inconvenience and discomfort to the patient of long and often awkward positioning of the head at a reactor port.
- the anticipated use of accelerator- generated neutrons would likely produce a significantly lower flux and hence effect longer irradiation times, so that compounds that have longer tumor retention times would become critical.
- the present invention is directed to low toxicity boronated compounds and methods for their use in the treatment, visualization, and diagnosis of tumors. More specifically, the present invention is directed to low toxicity boronated 5, 10, 15, 20- tetraphenylporphyrin compounds and methods for their use particularly in boron neutron capture therapy (BNCT) or photodynamic therapy (PDT) for the treatment of tumors of the brain, head and neck, and surrounding tissue.
- BNCT boron neutron capture therapy
- PDT photodynamic therapy
- the present invention is directed to boron-eontaining 5, 10, 15, 20- tetraphenyl porphyrins of the formula
- Y 1 , Y 2 , Y 3 , and Y 4 are independently on the ortho, meta or para position on the phenyl rings, and are independently hydrogen, alkyl, cycloalkyl, aryl, alkylaryl, arylalkyl, heteroaryl, or an alkyl, cycloalkyl, aryl, alkylaryl, arylalkyl, or heteroaryl group substituted with 1 to 4 hydrophilic groups selected from hydroxy, alkoxy, -C(O)OR 5 , - SOR 6 , -SO 2 R 6 , nitro, amido, ureido, carbamato, -SR 7 , -NR 8 R 9 , or poly-alkyleneoxide; or a substituent represented by the formula
- D represents independently, Z, hydrogen, or a substituent represented by the formula
- At least one D is Z or is represented by formula (3);
- D' represents independently, Z, hydrogen, or a substituent represented by the formula
- Y 5 , Y 6 , Y 7 , and Y 8 are independently on the ortho, meta or para position on the phenyl rings, and are represented by the formula
- W 1 , W 2 , W 3 , W 4 , W 5 , and W 6 are hydrophilic groups independently on the ortho, meta or para position on the phenyl rings, and are independently selected from hydroxy, alkoxy, - C(O)OR 5 , -SOR 6 , -SO 2 R 6 , nitro, amido, ureido, carbamate, -SR 7 , -NR 8 R 9 , or polyalkylene oxide;
- X a , X 1 , X 2 , X 3 , and X 4 are independently oxygen or sulfur;
- R a , R b , R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , and R 13 are independently selected from hydrogen and C 1 to C 4 alkyl;
- Z is a carborane cluster comprising at least two carbon atoms and at least three boron atoms, or at least one carbon atom and at least five boron atoms, within a cage structure;
- n, p, s, t, and v independently represent 0, or an integer from 1 to 20;
- n independently represents 1, 2, or 3;
- q and r independently represent 0, 1, 2, or 3;
- a, b, c, and d independently represent 1 or 2;
- k, l, u, and w independently represent 0, 1, or 2;
- e, f, g, h, i, and j independently represent 0, or an integer from 1 to 5;
- M is either two hydrogen ions; a single monovalent metal ion; two monovalent metal ions; a divalent metal ion; a trivalent metal ion; a tetravalent metal ion; a pentavalent metal ion; a hexavalent metal ion; a radioactive metal ion useful in radioisotope-mediated radiation therapy or imageable by single photon emission computed tomography (SPECT) or positron emission tomography (PET); a paramagnetic metal ion detectable by magnetic resonance imaging (MRI); a metal ion suitable for boron neutron capture therapy (BNCT) or photodynamic therapy (PDT); or a combination thereof; wherein the porphyrin-metal complex derived from a single monovalent metal ion is charge-balanced by a counter cation, and the porphyrin-metal complex derived from a bivalent, tetravalent, pentavalent, hexavalent metal ion is
- Z is preferably selected from the carboranes -C 2 HB 9 H 10 or -C 2 HB 10 H 10 , wherein - C 2 HB 9 H 10 is nido ortho-, meta-, or para-carborane, and -C 2 HB 10 H 10 is closo ortho, meta-, or para-carborane.
- M is preferably vanadium (V), manganese (Mn), iron (Fe), ruthenium (Ru), technetium (Tc), chromium (Cr), platinum (Pt), cobalt (Co), nickel (Ni), copper (Cu), zinc (Zn), germanium (Ge), indium (In), tin (Sn), yttrium (Y), gold (Au), barium (Ba), tungsten (W), or gadolinium (Gd).
- M is copper (Cu) or nickel (Ni).
- a, b, c, and d are 1, Y 1 , Y 2 , Y 3 , and Y 4 are represented by formula (2); D is Z; X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are B; n and p are 1; m is 2; Y 1 , Y 2 , Y 3 , and Y 4 are in the meta position on the phenyl ring; the - ⁇ 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5, or 3 and 4, positions on the phenyl ring, and e, f. g, h, k, l, u, and w are 0.
- a, b, c, d, e, f, g, and h are 1 ; k, 1, u, and w are 0; Y 1 , Y 2 ,
- Y 3 , and Y 4 are in the para position on the phenyl ring, W 1 , W 2 , W 3 , and W 4 are in the meta position of the phenyl ring, W 1 , W 2 , W 3 , and W 4 are independently, hydroxy or alkoxy, and the -X 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5, or 3 and 4, positions of the phenyl ring, and D is Z.
- alkoxy is methoxy.
- k, l, u, and w are 1; a, b, c, and d are 1, Y 1 , Y 2 , Y 3 , and Y 4 are represented by formula (2); X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1; and m is 2; Y 1 , Y 2 , Y 3 , and Y 4 are in the meta position on the phenyl rings; Y 5 , Y 6 , Y 7 , and Y 8 are in the para position on the phenyl rings; the -X 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5, or 3 and 4, positions on the phenyl ring; D is Z; X a is O; R a and R b are H; v is 1, and e, f, g, and h are 0.
- a, b, c, and d are 2; Y 1 , Y 2 , Y 3 , and Y 4 are represented by formula (2); X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1 ; m is 2; Y 1 , Y 2 , Y 3 , and Y 4 are in the meta positions on each phenyl ring; the - ⁇ 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5, or 3 and 4, positions on the phenyl ring; D is Z; k, 1, n, and w are 0; and e, f, g, and h are 0. [0021] In yet another embodiment, a, b, c, and d are 1; e, t, g, h, k, l, u, and w are 0; Y 1 ,
- Y 2 , Y 3 , and Y 4 are represented by formula (2); X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1 ; m is 2; Y 1 , Y 2 , Y 3 , and Y 4 are in a meta position on each phenyl ring; the _ X 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5, or 3 and 4, positions on the phenyl ring; D is according to formula (3), wherein X 3 is O, R 10 and R 11 are H, s is 1 , D' is Z, q is 2, and i is 0. Since i is 0, W 5 is not present.
- the counter dianion is a porphyrin compound containing a divalent negative charge.
- the porphyrin compound containing a divalent negative charge may be a carborane-containing porphyrin compound of the present invention, with the proviso that M is absent.
- the present invention also includes methods of tumor imaging by SPECT, PET, or MRI, as well as methods of bimodal cancer treatment such as BNCT and PDT that require the administration to a subject of a composition that comprises one or more of the porphyrin compounds described above.
- the composition is essentially one or more of the porphyrin compounds described above.
- the invention relates to boron-containing 5, 10, 15, 20-tetraphenyl porphyrins having the formula
- Y 1 , Y 2 , Y 3 , and Y 4 are independently on the ortho, meta or para position on the phenyl rings, and a, b, c, and d independently represent 1 or 2.
- Y 1 , Y 2 , Y 3 , and Y 4 are independently hydrogen, alkyl, cycloalkyl, aryl, alkylaryl, arylalkyl, heteroaryl, or an alkyl, cycloalkyl, aryl, alkylaryl, arylalkyl, or heteroaryl group substituted with 1 to 4 hydrophilic groups selected from hydroxy, alkoxy, -C(O)OR 5 , -SOR 6 , -SO 2 R 6 , nitro, amido, ureido, carbamato, -SR 7 , -NR 8 R 9 , or poly-alkyleneoxide; or a substituent represented by the formula
- Y 1 , Y 2 , Y 3 , and Y 4 represents formula (2), [0026]
- X 1 and X 2 axe independently oxygen or sulfur, and R 1 , R 2 , R 3 , and
- R 4 are independently selected from hydrogen and alkyl groups as defined below, except that the alkyl groups for R 1 , R 2 , R 3 , and R 4 contain 1 to 4 carbon atoms.
- the subscripts n and p independently represent 0, or an integer from 1 to 20, and m independently represents 1, 2, or 3.
- D represents independently, Z, hydrogen, or a substituent represented by the formula
- At least one D is Z or is represented by formula (3).
- X 3 is independently oxygen or sulfur;
- R 10 and R 11 are independently selected from hydrogen and alkyl groups as defined below, except that the alkyl groups for R 10 and R 11 contain 1 to 4 carbon atoms;
- s independently represents 0, or an integer from 1 to 20;
- q independently represents 0, 1, 2, or 3, provided that when q is 0, or when q is not zero and D' is solely hydrogen, then, at least one D is represented by Z, or when, q is not zero and D' is represented by formula (4) and r is zero, then at least one D is represented by Z.
- D' represents independently, Z, hydrogen, or a substituent represented by the formula
- X 4 Is independently oxygen or sulfur;
- R 12 and R 13 are independently selected from hydrogen and alkyl groups as defined below, except that the alkyl groups for R 12 and R 13 contain 1 to 4 carbon atoms;
- t independently represents 0, or an integer from: 1 to 20; and
- r independently represents 0, 1, 2, or 3.
- Z is a carborane cluster comprising at least two carbon atoms and at least three boron atoms, or at least one carbon atom and at least five boron atoms, within a cage structure.
- carborane clusters include the regular polyhedral carborane clusters, also known as closo structures, as well as ionized fragments of the polyhedral clusters, also known as nido structures.
- Some examples of the preferred carboranes of the present invention include -C 2 HB 9 H 10 or -C 2 HB 10 H 10 , wherein -C 2 HB 9 H 10 is nido ortho-, meta-, or para-carborane, and -C 2 HB 10 H 10 is closo ortho-, meta-, or para-carborane.
- W 1 , W 2 , W 3 , W 4 , W 5 , and W 6 are hydrophilic groups independently on the ortho, meta or para position on the phenyl rings, W 1 , W 2 , W 3 , W 4 , W 5 , and W 6 are independently selected from hydroxy, alkoxy, -C(O)OR 5 , -SOR 6 , -SO 2 R 6 , nitro, amido, ureido, carbamato, -SR 7 , -NR 8 R 9 , or polyalkylene oxide.
- R 5 , R 6 , R 7 , R 8 , R 9 are independently selected from hydrogen and alkyl groups as defined below, except that the alkyl groups for R 5 , R 6 , R 7 , R 8 , R 9 contain 1 to 4 carbon atoms,
- the subscripts e, f, g, h, i, and j independently represent 0, or an integer from 1 to 5.
- Y 5 , Y 6 , Y 7 , and Y 8 are independently on the ortho, meta or para position on the phenyl rings, and are represented by the formula In. formula 5, X a is independently oxygen or sulfur; R a and R b are independently selected from hydrogen and alkyl groups as defined below, except that the alkyl groups for R a and R b contain 1 to 4 carbon atoms; v independently represents 0, or an integer from 1 to 20; k, l, u, and w independently represent 0, 1, or 2; provided that each of the sums a + e + k, b + f +l, c + g + u, h + d + w, q + i, r + j, independently represents an integer from 1 to 5; when any of k, 1, u, or w is not zero, then at least one of Y 1 , Y 2 , Y 3 , and Y 4 represents formula (2).
- alkyl is a straight chain or branched alkyl group containing 1 to 20 carbon atoms including, optionally, up to three double or triple bonds.
- alkyl groups include methyl, ethyl, n-propyl, iso-propyl, n- butyl, iso-butyl, sec-butyl, tert-butyl, propenyl, 2-butenyI, 3-butenyl, 3-butynyl, 2-methyI ⁇ 2-butenayl, n-pentyl, dodecyl, hexadecyl, octadecyl, and eicosyl.
- the alkyl group may be unsubstituted or substituted with 1 to 4 hydrophilic groups.
- suitable hydrophilic groups include hydroxy, alkoxy, - C(O)OR 5 , -SOR 6 , -SO 2 R 6 , nitro, amido, ureido, carbamato, -SR 7 , -NR 8 R 9 , and poly- alkyleneoxide.
- R 5 , R 6 , R 7 , R 8 , and R 9 are independently selected from hydrogen and alkyl groups as defined above, except that the alkyl groups for R 5 , R 6 , R 7 , R 8 contain 1 to 4 carbon atoms,
- the carbon atoms of the alkyl group may also be substituted with 1 to 4 heteroatoms.
- heteroatoms axe O, N, or S.
- the heteroatoms are not adjacent, and are separated by at least one carbon atom.
- the cycloalkyl ring is a 4, 5, 6, or 7 member cycloalkyl ring.
- the ring may be saturated, or may contain 1 to 4 unsaturated (i.e., double or triple) bonds.
- saturated cycloalkyl rings include cyclobutane, cyclopentane, cyclohexane, and cyclopentane rings.
- unsaturated cycloalkyl rings include cyclobutene, cyelopentene, cyclohexene, and 1,3- cycloheptadiene rings.
- the cycloalkyl ring may optionally be substituted with 1 to 4 heteroatoms of O, N, or S.
- Some examples of cycloalkyl rings substituted with heteroatoms include pyrrolidine, piperidine, piperazine, tetrahydrofuran, furan, thiophene, 1,3-oxazolidine, imidazole, and pyrrole rings,
- the cycloalkyl rings may be optionally substituted with alkyl as defined above, or with 1 to 4 hydrophilic groups, also as defined above.
- the cycloalkyl ring maybe fused to 1 to 3 additional 4, 5, 6, or 7 member cycloalkyl or phenyl rings.
- fused cycloalkyl rings are bicyclo[3.3.0]octane, bicyclo[4.3.0]non-3-ene, triphenylene, and 1,2,3,4- tetrahydronaphthatene rings.
- aryl is a 5, 6, or 7 member aromatic ring, preferably a phenyl ring.
- the aryl rings maybe optionally substituted with alkyl as defined above to produce alkylaryl or arylalkyl groups, .
- the aryl, alkylaryl, and arylalkyl groups may be substituted with 1 to 4 hydrophilic groups, as defined above.
- the aryl ring may optionally be substituted with 1 to 4 heteroatoms of O, N, or S, resulting in a heteroaryl ring.
- heteroaryl rings include thiophene, pyridine, oxazole, thiazole, oxazine, and pyrazine rings.
- the heteroaryl ring may be substituted with 1 to 4 hydrophilic groups, as defined above.
- the aryl or heteroaryl ring may also be fused to 1 to 3 additional 5, 6, or 7 member aryl or heteroaryl rings.
- fused aryl and heteroaryl rings include napthalene, anthracene, phenanthrene, triphenylene, chrysene, indol ⁇ ne, quinoline, and tetraazanaphthalene (pteridine) rings.
- an alkoxy group contains an alkyl portion as defined above.
- Some examples of alkoxy groups include methoxy, ethoxy, propoxy, n-butoxy, t-butoxy, and dodecyloxy.
- a polyalkylene oxide is defined according to the formula -(CH 2 ) d -O-[(CH 2 ) c -O-] x - [(CH 2 ) f -O-] y -(CH 2 ) g -OR', wherein, independently, d is 0, or an integer from 1 to 10, e is 0, or an integer from 1 to 10, f is 1 to 10, g is 1 to 10, x and y are each independently 1 or 0, and R' is either H or an alkyl group as defined previously, provided that when e is 0, then x is 0; when f is 0, then y is 0; when e is not 0, then x is 1 ; and when f is not 0, then y is 1.
- a preferable polyalkylene oxide of the invention is polyethylene oxide.
- Polyethylene oxide is defined according to the formula -(CH 2 ) d -O-[(CH 2 ) e -O-] x -[(CH 2 ) f -
- M may be two hydrogen ions, a single monovalent metal ion, or two monovalent metal ions.
- suitable monovalent metal ions include Li +1 , Na +1 , K +1 , Cu +1 , Ag +1 , Au +1 , and Tl +1 .
- counter cations include any of the foregoing monovalent metal ions, and ammonium and phosphonium cations, such as tetramethylammonium, tetrabutylammonium, and tetraphenylammonium.
- the counter cation may be either bound or associated in some form with the porphyrin-metal complex.
- M may also be a divalent metal ion.
- suitable divalent metal ions include V +2 , Mn +2 , Fe +2 , Ru +2 , Co +2 , Ni +2 , Cu +2 , Pd +2 , Pt +2 , Zn +2 , Ca +2 , Mg +2 , Sr +2 , and Ba +2 .
- M maybe a trivalent, tetravalent, pentavalent, or hexavalent metal ion.
- suitable trivalent metal ions include Gd +3 , Y +3 , In +3 , Cr +3 , Ga +3 , Al +3 , Eu +3 , and Dy +3 .
- suitable tetravalent metal ions include Tc +4 , Ge +4 , Sn +4 , and Pt +4 .
- An example of a suitable pentavalent metal ion is Tc +5 .
- suitable hexavalent metal ions include W +6 , Tc +6 , and Mo +6 .
- the resulting porphyrin-metal complex cation is charge-balanced by an appropriate number of counter anions, dianions, or trianions.
- a porphyrin-metal complex cation derived from a trivalent metal ion may be charge-balanced by a single counter anion, and such a complex derived from a tetravalent metal ion may, for example, be charge-balanced by a single counter dianion or two counter anions, and so on.
- suitable counter anions include chloride, perchlorate, sulfate, nitrate, and tetrafluoroborate.
- Suitable counter dianions include oxide, sulfide, or a porphyrin compound containing a divalent negative charge.
- the porphyrin compound containing a divalent negative charge may be a porphyrin compound of the present invention with the proviso that M is absent.
- An example of a suitable counter trianion includes phosphate.
- the counter anion, dianion, or trianion maybe either bound or associated in some form with a carborane-containing porphyrin compound of the present invention.
- the carborane-containing porphyrin compound may also be bound to or associated with neutrally charged molecules, such as molecules of solvation, for example, water, acetonitrile, methanol, and so on.
- M may be a radioactive metal ion imageable by single photon emission computed tomography (SPECT) or positron emission tomography (PET).
- SPECT single photon emission computed tomography
- PET positron emission tomography
- radioactive metals suitable for SPECT are 67 Cu, 99m Tc, 111 In, and those for PET include 64 Cu, 55 Co
- M may also be a radioactive metal useful as a radiopharmaceutical for therapy.
- radioactive metals suitable for such therapy include 90 Y, 188 Re, 67 Cu.
- M may also be a paramagnetic metal ion detectable by magnetic resonance imaging (MRI).
- MRI magnetic resonance imaging
- metals include Mn, Fe, Co, and Gd.
- M may be a metal ion suitable for boron neutron capture therapy (BNCT) or photodynamic therapy (PDT); or a combination thereof.
- the metal ions suitable for BNCT include those described thus far, with the exclusion of those that are photoactive, such as Zn and Sn. Such photoactive metals, and particularly those with long-lived triplet states, are preferable for PDT. Since the dosage for BNCT is 100 to 1000 times greater than the dosage for PDT, a significant accumulation of photoactive metal in the skin could result if such photoactive metals were used in BNCT. Such an accumulation of photoactive metal may cause biological damage.
- the invention also relates to methods of treating tumors. In.
- the method of treating malignant tumors, especially brain tumors is via BNCT.
- BNCT is abimodal cancer treatment based on the selective accumulation of a stable nuclide of boron known as boron- 10, or 10 B, in the tumor, followed by irradiation of the tumor with thermalized neutrons.
- the thermalized neutrons impinge on the boron- 10, causing a nuclear fission reaction.
- the nuclear fission causes the highly localized release of vast amounts of energy in the form of high linear-energy-transfer (LET) radiation, which can more effectively kill cells than low LET radiation, such as x-rays.
- LET linear-energy-transfer
- a boron-10 nucleus captures a neutron forming the metastable nuclide 11 B, which spontaneously and nearly instantaneously disintegrates into a 4 He and 7 Li particle, which together possess art average total kinetic energy of 234 MeV.
- These two ionized particles travel about 9 ⁇ m and 5 ⁇ m (7 ⁇ 2 ⁇ m) in opposite directions in soft tissue, respectively.
- the patient's head is irradiated in the general area of the brain tumor with an incident beam or field of epithermal (0.5 eV-10 keV) neutrons.
- the neutrons become progressively thermalized (average energy approximately 0.04 eV) as they penetrate deeper into the head.
- the neutrons become thermalized, they are more readily captured by the boron- 10 concentrated in the tumor cells and/or tumor supporting tissues, since the capture cross section is inversely proportional to the neutron velocity.
- BNCT of malignant brain tumors following the method of the present invention, the patient is first given an infusion of a carborane-containing porphyrin of formula (1), which is highly enriched in boron-10.
- the caxborane-containing porphyrin is then concentrated preferentially in the brain tumor within the effective irradiation volume, which, for brain tumors may be a substantial part of the brain.
- tumors located in most or all of one hemisphere and some or all of the contralateral hemisphere of the brain can accumulate boronated porphyrins,
- the tumor area is then irradiated with thermalized neutrons (primary irradiation), some of which are captured by the boron-10 concentrated in the tumor.
- thermalized neutrons primary irradiation
- the relative probability that the slow-moving thermal neutrons will be captured by the boron-10 nuclide is high compared to the probability of capture by all of the other nuclides normally present in mammalian tissues, provided that boron-10 concentrations in tumor tissues is greater than 30 ⁇ g/g.
- the carborane clusters are highly enriched in boron-10. Specifically, the boron in the carborane cluster is enriched to at least 95 atom% in boron- 10.
- An advantage of the present invention over the prior art for the treatment of cancer is that the boron-containing porphyrins of the present invention selectively accumulate in neoplasms in more preferred ratios than other known boron-containing compounds.
- the porphyrin compounds of the present invention that have been tested in vivo are non-toxic at theoretically therapeutic effective doses.
- the higher selectivity and lower toxicity of the carborane-containing porphyrins of the present invention allow for the selective destruction of tumor tissue with minimal disruption of normal tissues and tissue function when irradiated.
- Another advantage of the carborane-containing porphyrins of the present invention is their increased polarity, imparted by the hydrophilic groups W 1 , W 2 , W, W 4 , W 5 , and W 6 , and/or the ether linkages.
- the greater polarity of such groups render the tetraphenyl porphyrin compounds less lipophilic, which effects a reduction of the amount of an emulsifying co-solvent during administration. Therefore, the microlocalization within the tumor cell may be improved yielding a higher relative biological effect.
- the ether linkages in the carborane-containing porphyrins of the present invention provide more chemical stability than, for example, ester or amide linkages, which can much more easily hydrolyze, thereby causing the loss of the boron functionality.
- the ether linkages possess nearly the same resistance to hydrolysis and other forms of chemical attack as a carbon-carbon linkage.
- carborane-containing porphyrins of the present invention contain in excess of 8 carborane clusters (80 boron atoms).
- the present invention provides for carborane-containing porphyrin molecules containing in excess of 16, 32, or even 64, carborane clusters, which is higher than any carborane- containing porphyrins currently known.
- the carrier may include such commercially available solvents as Cremophor EL, propylene glycol, Tween 80, polyethylene glycol, or liposomes.
- the compound is administered in one or more doses, the last dose being given between about 1 hour and one week prior to the epithermal neutron irradiation.
- the timing of the neutron exposure depends upon the concentration, of the porphyrin in the blood, which decreases more rapidly with time than the porphyrin concentration in the tumor.
- the timing of the neutron exposure also depends on other factors that are well known to those skilled in the art of clinical BNCT. These other factors include the pharmacokinetic behavior of the compound, (e.g., the rate of absorption of the compound into the tumor and into the tumor vasculature) and the rate of excretion from and/or metabolism of the compound in the tumor and various other tissues that absorb the compound.
- the method of treating malignant tumors of the present invention is via PDT
- PDT is a bimodal cancer treatment based on the selective accumulation of a porphyrin in a tumor, followed by irradiation of the tumor with laser red light.
- an electron of the porphyrin is excited from the singlet ground state to a singlet excited state.
- the electron then can either return to the singlet ground state with the emission of light causing fluorescence, or it can change its spin via intersystem crossing to the triplet state.
- the decay of the triplet back down to the ground state singlet it can transfer energy to ground state triplet dioxygen which forms the highly reactive singlet oxygen.
- Biomolecules that react most readily with singlet oxygen include unsaturated lipids and alpha amino-acid residues, both of which are major constituents of biological membranes. Beyond a certain reversible or repairable threshold, damage to membranes, especially to endothelial cell membranes, can lead to local vascular thrombosis and shutdown of blood circulation.
- the patient is first given an injection or infusion of a photosensitizing carborane-containing porphyrin of formula (1). Fiber-optic probes are then used to illuminate the tumor tissue.
- the PDT photosensitizers have optical absorbance peaks at sufficiently long wavelengths for maximum penetration to the depth of the tumor.
- the therapeutic treatment of malignant tumors is augmented by the use of SPECT or PET.
- SPECT the patient is first given an infusion or injection of a compound of formula (1) wherein M is a gamma-emitting radioactive metal ion.
- M is a gamma-emitting radioactive metal ion.
- the patient's head is then scanned noninvasively and the radionuclide concentration, and hence indirectly, the average boron concentration, in each pixel or voxel representing brain or brain tumor tissue is imaged. Contour lines representing zones of equal boron- 10 concentration can thereby be drawn on each image of the brain.
- SPECT of the brain is at least one order of magnitude more sensitive to isotopic tracers than is conventional radiography or computerized tomography
- SPECT results can be analyzed to provide quantitative information either in defined volumes or voxels of the brain images, in the concentrations of boron relevant to BNCT treatment planning and implementation, SPECT scanning can indicate the presence of a tumor in the patient, as well as its location in the brain or elsewhere in the body. SPECT scanning is noninvasive, fast, and convenient.
- the positron emitting PET-imageable radioisotope Cu-64 is more readily available than is Cu-67, used in SPECT. Because of the much greater availability of Cu-64, we have carried out preclinical PET studies using a Cu-64 labeled porphyrin.
- the therapeutic treatment of malignant tumors is augmented by the use of MRI.
- MRI Magnetic resonance Imaging
- a patient is first given an infusion or injection of a solution containing a carborane-containing porphyrin of formula (I) chelated to a suitable paramagnetic metal ion.
- the patient's head is then scanned and the paramagnetic metal ion concentration, and thus, boron concentration in the brain is imaged and quantified.
- MRl utilizing the compounds of the present invention may permit rapid enhanced targeting and treatment planning for neutron irradiation in BNCT before, during and after infusion when the boronated compound is being redistributed in blood, tumor, and healthy tissue.
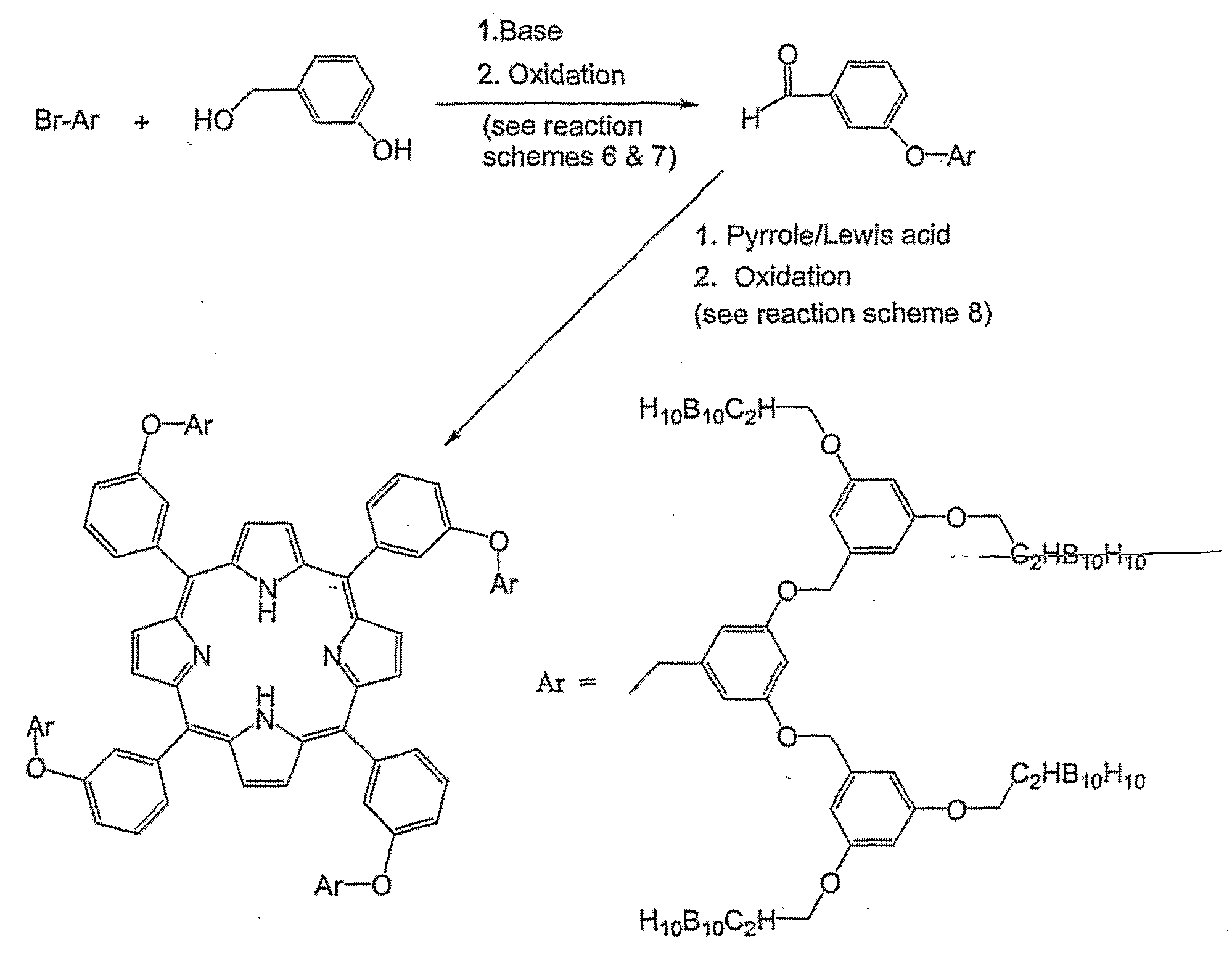
- the carborane-containing porphyrins of the present invention are synthesized through a series of separate steps. Provided below is first, a summary of the synthetic steps required for the preparation of the preferred carborane-containing porphyrins of the present invention.
- the synthetic summary provides general methods for synthesizing compounds of the invention, and is thereby meant to encompass numerous ways for synthesizing each compound. For example, different starting materials may be used to synthesize the same product, and each starting material may require a different set of reaction conditions such as temperature, reaction time, solvents, and extraction and purification procedures.
- the coupling agent is any compound, mixture, or sequence of compounds capable of coupling a phenol or thiophenol and an alkyl halide to produce an ether. Some coupling agents may not require reflux conditions or a polar non-prot ⁇ c solvent.
- the coupling agent is a mixture of potassium carbonate and potassium iodide (K 2 CO 3 /KI).
- the acid catalyst may be any Bronsted-Lowry (proton donating) acid that does not interfere with conversion of the alcohol to the ester product.
- the acid catalyst is sulfuric acid, H 2 SO 4 .
- the borane cluster is any cluster comprising at least three boron atoms within a cage structure.
- the borane cluster can be decaborane, B 10 H 14 .
- the borane cluster reacts with the triple bond of the propargyl starting material to form the carboranyl product
- Z represents the carborane -C 2 HB 10 H 10 .
- Z represents any carborane cluster comprising at least two carbon atoms and at least three boron atoms, or at least one carbon atom and at least five boron atoms, within a cage structure
- the carborane cluster maybe -C 2 HB 9 H 10 or -C 2 HB 10 H 10 , wherein -C 2 HB 9 H 10 is nido ortho-, meta-, or para-carborane, and -C 2 HB 10 H 10 is closo ortho-, meta-, or para-carborane.
- the protecting acid is any acid, acid mixture, or sequence of acid additions capable of converting the ester into the alcohol product.
- the protonating acid is concentrated HCl.
- the protic solvent may be, for example, an alcohol such as methanol.
- the halogenating agent is any agent capable of converting the hydroxy substituent of the starting material to a halogen.
- the halogenating agent is a 1:1 mixture of carbon tetrabromide and triphenylphosphine, wherein D becomes a bromide.
- the reaction is performed preferably in an ether solvent, such as tetrahydrofuran (THF).
- Solvent C is preferably a polar, aprotic solvent such as acetone.
- the coupling agent is any compound, mixture, or sequence of compounds capable of coupling a phenol or thiophenol and an alkyl halide to produce an ether. Some coupling agents may not require reflux conditions or a polar, aprotic solvent.
- the coupling agent is a mixture of potassium carbonate and potassium iodide (K 2 CO 3 /KI).
- the oxidant is any oxidizing compound capable of selectively converting a primary alcohol to an aldehyde, preferably 2,3- dichloro-5,6-dicyano-1.4-benzoquinone (DDQ) or pyridinium chlorochromate (PCC).
- Solvent D is a non-polar aprotic solvent, preferably dlchloromethane.
- the coupling system preferably comprises a Lewis acid (electron acceptor) such as boron trifluoride (BF 3 ) or trifluoroacetic acid (TFA) to form the intermediate porphyrinogen from the pyrrole, and benzaldehyde and an oxidizing agent such as 2,3-dichloro-5,6-dicyano-1.4-benzoquinone (DDQ) to oxidize the porphyrinogen to porphyrin.
- Solvent E is a nonpolar aprotic solvent, preferably dichloromethane.
- M is selected from the group consisting of vanadium (V), manganese (Mn), iron (Fe), ruthenium (Ru), technetium (Tc), chromium (Cr), platinum (Pt), cobalt (Co), nickel (Ni), copper (Cu), zinc (Zn), germanium (Ge), indium. (In), tin (Sn), yttrium (Y), gold (Au), barium (Ba), tungsten (W), and gadolinium (Gd).
- M is copper (Cu) or nickel (Ni).
- the metal salt used contains the metal ion M chelated to the porphyrin.
- M metal ion M chelated to the porphyrin.
- copper acetate i.e., Cu(OAc) 2 .H 2 O
- Solvent F is any solvent or solvent mixture capable of at least partially solubilizing the porphyrin and metal salt, and that does not interfere with incorporating the metal into the porphyrin.
- porphyrin VIII has the following structure:
- Porphyrin VIII shown above In this case, a, b, c, and d are 1, Y 1 , Y 2 , Y 3 , and Y 4 are represented by formula (2); D is Z, wherein Z is the -C 2 HB 10 H 10 carborane; X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1; m is 2; Y 1 , Y 2 , Y 3 , and Y 4 are in the meta position on the phenyl ring; the - ⁇ 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5 positions on the phenyl ring, and e, f, g, h, k, 1, u, and w are 0.
- Porphyrin A an 8-carborane-containing porphyrin containing hydrophilic groups W 1 , W 2 , W 3 , and W 4 :
- a, b, c, d, e, f, g, and h. are 1 ; k, l, u, and w are 0; Y 1 , Y 2 , Y 3 , and Y 4 (i.e., O-Ar) are in the para position of each phenyl ring, the hydrophilic groups W 1 , W 2 , W 3 , and W 4 are in the meta position of each phenyl ring, wherein W 1 , W 2 , W 3 , and W 4 are methoxy; the -X 2 -(CR 3 R 4 ) -D substituents are in the 3 and 5 positions of the Ar phenyl rings; D is Z, wherein Z is the -C 2 HB 10 H 10 carborane.
- Porphyrin B a 12-carborane-containing porphyrin:
- k, l, u, and w are 1; a, b, c, and d are 1, Y 1 , Y 2 , Y 3 , and
- Y 4 are represented by formula (2); X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1; and m is 2; Y 1 , Y 2 , Y 3 , and Y 4 (i.e., O-Ar) are in the meta position of each phenyl ring; Y 5 , Y 6 , Y 7 , and Y 8 (in this case, O-CH 2 -C 2 HB 10 H 10 ) are in the para position of each phenyl ring; the -X 2 -(CR 3 R 4 ) p -D substituents are in the 3 and 5 positions of the Ar phenyl rings; D is Z, wherein Z is the -C 2 HB 10 H 10 carborane; X a is O; R a and R b are H; v is 1, and e, f, g, and h are 0.
- Porphyrin C an 8-carborane-containing porphyrin:
- a, b, c, and d are 2; Y 1 , Y 2 , Y 3 , and Y 4 (i.e., O-Ar) are represented by formula (2); X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1 ; m is 2; Y 1 , Y 2 , Y 3 , and Y 4 are in the two meta positions on each phenyl ring; the -X 2 -(CR 3 R 4 ) p - D substituents are in the 3 and 5 positions on the Ar phenyl rings; D is Z, wherein Z is the -C 2 HB 10 H 10 carborane; k, 1, u, and w are 0; and e, f, g, and h are 0.
- Porphyrin D a 16-carborane containing porphyrin:
- a, b, c, and d are 1; e, f, g, h, k, 1, u, and w are 0; Y 1 , Y 2 , Y 3 , and Y 4 are represented by formula (2); X 1 and X 2 are O; R 1 , R 2 , R 3 and R 4 are H; n and p are 1; m is 2; Y 1 , Y 2 , Y 3 , and Y 4 (i.e., O-Ar) are in the meta positions on each phenyl ring; the - ⁇ 2 -(CR 3 R 4 ) -D substituents are in the 3 and 5 positions on the phenyl ring; D is according to formula (3), wherein X 3 is O, R 10 and R 11 are H, s is 1 , D' is Z, wherein Z is the -C 2 HB 10 H 10 carborane; q is 2, and
- the product had a melting point of 79-80°C and gave the following proton nuclear magnetic resonance ( 1 H NMR) spectrum in ppm (in CDCl 3 solvent): 2.52 (triplet, 2H, alkynyl); 2.15 (broad singlet, 1H, hydroxyl); 4,65 (doublet, 4H, ArOCH 2 ); 4.60 (singlet, 2H, ArCH 2 ); 6.52 (singlet, 1H, aryl); 6.60 (singlet, 2H, aryl).
- the aqueous solution was extracted with 50 mL x 2 of dichloromethane, and the organic phase was washed with water (50 mL x 2) and then dried with anhydrous sodium sulfate.
- the desired product was purified using a pad of silica in a sintered glass funnel, the pad of silica then washed with 200 mL dichloromethane.
- the dichloromethane of the filtrate was removed by rotary evaporation, leaving a yellow oil, which solidified upon standing, 7.2g of product was obtained, which corresponds to a 96% yield.
- the product had a melting point of 65-66°C and gave the following proton nuclear magnetic resonance ( 1 H NMR) spectrum in ppm (in CDCI 3 solvent): 2.11 (singlet, 3H, CH 3 ); 2.54 (triplet, 2H, alkynyl); 4.67 (doublet, 4H, ArOCH 2 ); 5.05 (singlet, 2H, ArCH 2 ); 6.58 (singlet, 1H, aryl); 6.61 (singlet, 2H, aryl).
- the product had a melting point of 122-123°C and gave the following proton nuclear magnetic resonance ( 1 H NMR) spectrum in ppm (in CDCI 3 solvent): 2.12 (singlet, 3H, CH 3 ); 4.06 (singlet, 2H, CCHB 10 H 10 ); 4.39 (singlet, 4H, ArOCH 2 ); 5.01 (singlet, 2H, ArCH 2 ); 6.32 (singlet, 1H, aryl); 6.52 (singlet, 2H, aryl).
- the product had a melting point of 267-269°C and gave the following proton nuclear magnetic resonance ( 1 H NMR) spectrum in ppm (in CDCI 3 solvent): 2.54 (broad singlet, 1H hydroxyl); 4.04 (singlet, 2H, CCHB 10 H 10 ); 4.40 (singlet, 4H, ArOCH 2 ); 4.65 (singlet, 2H, ArCH 2 ); 6.28 (singlet, 1H, aryl); 6.54 (singlet, 2H, aryl).
- 1 H NMR proton nuclear magnetic resonance
- the product had a melting point of 230-232°C and gave the following proton nuclear magnetic resonance ( 1 H NMR) spectrum in ppm (in CDCl 3 solvent): 4.02 (singlet, 2H, CCHB 10 H 10 ); 4.37 (singlet, 2H, CH 2 Br); 439 (singlet, 4H, ArOCH 2 ); 6.26 (singlet, 1H, aryl); 6.55 (singlet, 2H, aryl).
- K 2 CO 3 0.210 grams (1.5 millimoles), and KI, 0.25 grams (1,5 millimoles), were placed in a 50 mL round-bottomed flask. 3,5-dicarboranyl- methoxylbenzyl bromide (V), 0.410 grams (0.80 millimoles), 3-hydroxybenzylalcohol, 0.100 grams (0.80 millimoles), and 20 mL acetone, were then added. The mixture was reftuxed under an argon atmosphere for 24 bours. The solvent was removed by rotary evaporation, leaving an organic residue. The residue was extracted with 10 mL of dichloromethane, and the organic phase was washed with water in a separatory funnel. The organic phase was then dried over anhydrous potassium carbonate. The dichloromethane was removed by rotary evaporation, leaving a white solid. 0.430 grams of product was obtained, which corresponds to a 96% yield.
- the product had a melting point of 259-261°C and gave the following proton nuclear magnetic resonance ( 1 H MMR) spectrum in ppm (in CDCI 3 solvent): 1.70 (singlet, 1H, hydroxyl); 4,04 (singlet, 2H, CCHB 10 H 10 ); 4.40 (singlet, 4H, CH 2 CCHB 10 H 10 ); 4.67 (singlet, 2H, ArCH 2 OH); 5.00 (singlet 2H, ArCH 2 Oar); 6.31 (singlet.
- 1 H MMR proton nuclear magnetic resonance
- the product had a melting point of 263-265°C and gave the following proton nuclear magnetic resonance ( 1 H HMR) spectrum in ppm (in CDCI 3 solvent): 4.04 (singlet, 2H, CCBB 10 H 10 ); 4.42 (singlet; 4H, ArOCH 2 ); 5,00 (singlet, 2H, ArCH 2 O); 6,33 (singlet, 1H, aryl); 6.61 (singlet, 2H, aryl); 7.23 (singlet, 1H, aryl); 7.44 (multiplet, 1H, aryl); 7.50 (multiplet, 2H, aryl); 9,98 (singlet, 1H, CHO).
- the product gave the following proton-decoupled 13 C NMR spectrum in ppm (in CDCI 3 solvent); 58.0 (CH 2 CCHB 10 H 10 ); 69.5 (-CCHB 10 H 10 ); 69.7 (ArCH 2 OAr); 71.2 (-CCHB 10 H 10 ); 102.0 (aryl); 107.4 (aryl); 112.8 (aryl); 122.4 (aryl); 124.8 (aryl); 130.6 (aryl); 138.1 (aryl); 138.4 (aryl); 140.3 (aryl); 158.6 (aryl); 192.1 (CHO).
- the mass spectrum (FAB) showed a parent ion peak of 558.0 that matched the molecular weight of the compound.
- the mass spectrum showed a parent ion peak of 2418.3 that matched the molecular weight of the compound.
- the ultraviolet-visible absorbance specrtrum of the product (dichloromethane) showed the following peaks in nanometers of wavelength: 420, 516, 550, 589, and 645.
- the solid was re-dissolved in dichloromethane and purified using a silica pad eluting with a 1 : 1 solvent mixture of hexane and dichloromethane.
- the solvents were removed by rotary evaporation, leaving the red copper porphyrin compound, 57 milligrams of product, which corresponds to a 92% yield.
- the mass spectrum (FAB) showed a parent ion peak of 2479.9 that matched the molecular weight of the compound.
- the ultraviolet-visible absorbance spectrum of the product showed the following peaks in nanometers of wavelength (in dichloromethane solvent): 416, 539.
- Porphyrin compound (IX) was emulsified in 9% Cremophor EL and 18% propylene glycol in saline to give a porphyrin concentration of approximately 23 mg/mL.
- CCM Cremophor EL
- PRG propylene glycol
- the porphyrin was dissolved in tetrahydrofuran (THF) (1,5% of the total volume) and then heated to 40°C for 15 min. CRM (9% of total volume) was then added and the mixture was heated to 60°C for 2 hours, which removed most of the THF.
- mice bearing subcutaneously implanted EMT-6 mammary carcinomas implanted on the dorsal thorax were given a total dose of 87 milligrams porphyrin compound (IX) per kilogram body weight (30 mg B/kg) in 3 intraperitoneal (i.p.) injections over a period of 8 hours.
- the blood was first analyzed for hematologic parameters that indicate toxicity before it was analyzed for boron.
- Table 1 below shows the average boron concentrations for different types of tissue in five of the BALB/c mice in milligram of porphyrin compound (IX) per kilogram body weight.
- mice Weight changes and hematologic parameters in mice given 87 mg/kg porphyrin (IX) (30 mg B/kg) or solvent only (9% Cremophor and 18% propylene glycol in saline) at 2 or 4 days after the last injection. Values are reported as median (and range).
- the results of the preliminary biodistribution study showed that the tumor boron concentrations are adequate for therapy, particularly at the two-day time-point.
- the tumor to blood boron ratios are quite high, which are more than 80:1, and tumor to brain ratios are even higher.
- the platelet data indicate a small but significant decrease in the porphyrin-administered mice at the 2-day time point compared to the solvent-only mice. However, by the 4-day time point, the platelet counts have rebounded to a level greater than those from the solvent-only group.
- the weight data indicate that the level of toxicity is very low if it exists at all since there were no differences between porphyrin- and solvent-injected mice, Thus, it is possible that the doses can be escalated significantly without affecting toxicity.
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2007519285A JP2008504365A (en) | 2004-06-28 | 2005-06-22 | Carboranyl porphyrins and their use |
| NZ552877A NZ552877A (en) | 2004-06-28 | 2005-06-22 | Carboranylporphyrins and uses thereof |
| EP05785045A EP1778082A4 (en) | 2004-06-28 | 2005-06-22 | Carboranylporphyrins and uses thereof |
| AU2005267390A AU2005267390B2 (en) | 2004-06-28 | 2005-06-22 | Carboranylporphyrins and uses thereof |
| CN2005800251742A CN1988848B (en) | 2004-06-28 | 2005-06-22 | Carboranylporphyrins and uses thereof |
| CA002572287A CA2572287A1 (en) | 2004-06-28 | 2005-06-22 | Carboranylporphyrins and uses thereof |
| KR1020077002041A KR20070033006A (en) | 2004-06-28 | 2005-06-22 | Carbonaranilporphyrin and uses thereof |
| US11/606,864 US20070093463A1 (en) | 2004-05-20 | 2006-12-01 | Radiation enhancement agent for X-ray radiation therapy and boron neutron-capture therapy |
| IL180365A IL180365A (en) | 2004-06-28 | 2006-12-26 | Carboranylporphyrins and uses thereof in imaging and in the treatment of cancer |
| NO20070509A NO20070509L (en) | 2004-06-28 | 2007-01-26 | Carboranyl porphyrins and uses thereof |
| HK07113697.2A HK1109039A1 (en) | 2004-06-28 | 2007-12-17 | Carboranylporphyrins and uses thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/878,138 US6989443B2 (en) | 2004-06-28 | 2004-06-28 | Carboranylporphyrins and uses thereof |
| US10/878,138 | 2004-06-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006012140A1 true WO2006012140A1 (en) | 2006-02-02 |
Family
ID=35505984
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2005/022061 WO2006012140A1 (en) | 2004-05-20 | 2005-06-22 | Carboranylporphyrins and uses thereof |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US6989443B2 (en) |
| EP (1) | EP1778082A4 (en) |
| JP (1) | JP2008504365A (en) |
| KR (1) | KR20070033006A (en) |
| CN (1) | CN1988848B (en) |
| AU (1) | AU2005267390B2 (en) |
| CA (1) | CA2572287A1 (en) |
| HK (1) | HK1109039A1 (en) |
| IL (1) | IL180365A (en) |
| NO (1) | NO20070509L (en) |
| NZ (1) | NZ552877A (en) |
| WO (1) | WO2006012140A1 (en) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070093463A1 (en) * | 2004-05-20 | 2007-04-26 | Brookhaven Science Associates, Llc | Radiation enhancement agent for X-ray radiation therapy and boron neutron-capture therapy |
| US8287839B2 (en) * | 2006-12-04 | 2012-10-16 | Brookhaven Science Associates, Llc | Carboranylporphyrins and uses thereof |
| US8444953B2 (en) * | 2007-03-22 | 2013-05-21 | Brookhaven Science Associates, Llc | Symmetric and asymmetric halogen-containing metallocarboranylporphyrins and uses thereof |
| US20080279781A1 (en) * | 2007-05-10 | 2008-11-13 | Brookhaven Science Associates, Llc | Glycosylated Carboranylporphyrins and Uses Thereof |
| JP5745415B2 (en) * | 2009-09-17 | 2015-07-08 | 国立大学法人九州大学 | Porphyrin compounds |
| CN102260269B (en) * | 2010-05-25 | 2013-11-06 | 苏州和健医药科技有限公司 | Dendrimer containing porphyrin or chlorine and its application |
| GB201212409D0 (en) | 2012-07-12 | 2012-08-22 | Morex Dev Partners Llp | Cancer treatment |
| JP6699004B2 (en) * | 2015-08-25 | 2020-05-27 | 住友重機械工業株式会社 | Neutron capture therapy system and method of controlling neutron capture therapy system |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4783529A (en) | 1985-12-03 | 1988-11-08 | Research Corporation Technologies | Rapid synthesis of radiolabeled porphyrin complexes for medical application |
| US5162231A (en) | 1989-10-25 | 1992-11-10 | Cole Dean A | Method of using 5,10,15,20-tetrakis(carboxyphenyl)porphine for detecting cancers of the lung |
| US4959356A (en) | 1989-05-26 | 1990-09-25 | The United States Of America As Represented By The United States Department Of Energy | Porphyrins for boron neutron capture therapy |
| FR2656866B1 (en) | 1990-01-10 | 1992-05-15 | Cis Bio Int | PORPHYRIN DERIVATIVES AND METALLOPORPHYRINS POTENTIALLY COUPLED TO A BIOLOGICALLY ACTIVE MOLECULE, AND PHARMACEUTICAL COMPOSITION CONTAINING THEM. |
| US5149801A (en) | 1990-11-21 | 1992-09-22 | The Regents Of The University Of California | Boronated porphyrin compounds |
| US5654423A (en) | 1990-11-21 | 1997-08-05 | Regents Of The University Of California | Boronated metalloporphyrine and therapeutic methods |
| US5312896A (en) | 1992-10-09 | 1994-05-17 | Sri International | Metal ion porphyrin-containing poly(imide) |
| DE4305523A1 (en) | 1993-02-17 | 1994-08-18 | Diagnostikforschung Inst | Meso-tetraphenylporphyrin complexes, processes for their preparation and pharmaceutical compositions containing them |
| IT232801Y1 (en) * | 1994-03-07 | 2000-01-19 | Imbac Spa | SIDE OF BOX WITH REMOVABLE AND INTERCHANGEABLE SIDE SHAFT |
| US5563132A (en) | 1994-06-21 | 1996-10-08 | Bodaness; Richard S. | Two-step cancer treatment method |
| US5877165A (en) | 1995-11-02 | 1999-03-02 | Brookhaven Science Associates | Boronated porhyrins and methods for their use |
| US5955586A (en) | 1996-03-22 | 1999-09-21 | Sessler; Jonathan L. | Highly boronated derivatives of texaphyrins |
| US6066628A (en) | 1997-01-09 | 2000-05-23 | Emory University | Non-iron metalloporphyrins and methods of use |
| US6010805A (en) | 1998-05-26 | 2000-01-04 | United States Of America As Represented By The Secretary Of The Air Force | Ion conducting electrolyte material containing a lithium porphyrin complex |
| AU2001274827A1 (en) | 2000-05-09 | 2001-11-20 | The Regents Of The University Of California | Porphyrin-based neutron capture agents for cancer therapy |
| GB2393123B (en) * | 2001-06-06 | 2005-01-12 | Brookhaven Science Ass Llc | Halogenated tetraphenylporphyrins and their uses as radiosensitizers for radiation therapy |
| US6566517B2 (en) | 2001-06-06 | 2003-05-20 | Brookhaven Science Associates, Llc | Metalloporphyrins and their uses as imageable tumor-targeting agents for radiation therapy |
-
2004
- 2004-06-28 US US10/878,138 patent/US6989443B2/en not_active Expired - Fee Related
-
2005
- 2005-06-22 WO PCT/US2005/022061 patent/WO2006012140A1/en active Application Filing
- 2005-06-22 CA CA002572287A patent/CA2572287A1/en not_active Abandoned
- 2005-06-22 CN CN2005800251742A patent/CN1988848B/en not_active Expired - Fee Related
- 2005-06-22 AU AU2005267390A patent/AU2005267390B2/en not_active Ceased
- 2005-06-22 KR KR1020077002041A patent/KR20070033006A/en not_active Application Discontinuation
- 2005-06-22 NZ NZ552877A patent/NZ552877A/en not_active IP Right Cessation
- 2005-06-22 EP EP05785045A patent/EP1778082A4/en not_active Withdrawn
- 2005-06-22 JP JP2007519285A patent/JP2008504365A/en active Pending
-
2006
- 2006-12-26 IL IL180365A patent/IL180365A/en not_active IP Right Cessation
-
2007
- 2007-01-26 NO NO20070509A patent/NO20070509L/en not_active Application Discontinuation
- 2007-12-17 HK HK07113697.2A patent/HK1109039A1/en not_active IP Right Cessation
Non-Patent Citations (1)
| Title |
|---|
| HARTH ET AL: "The Effect of Macromolecular Architecture in Nanomaterials: A comparison of Site Isolation in PorphyrinCore Dendrimers and Their Isomeric Linear Analogues", J. AM. CHEM. SOC., vol. 124, 2002, pages 3926 - 3938, XP008117119 * |
Also Published As
| Publication number | Publication date |
|---|---|
| IL180365A (en) | 2011-09-27 |
| CN1988848B (en) | 2010-12-15 |
| KR20070033006A (en) | 2007-03-23 |
| US6989443B2 (en) | 2006-01-24 |
| US20050287073A1 (en) | 2005-12-29 |
| AU2005267390B2 (en) | 2011-01-20 |
| HK1109039A1 (en) | 2008-05-30 |
| AU2005267390A1 (en) | 2006-02-02 |
| IL180365A0 (en) | 2007-06-03 |
| JP2008504365A (en) | 2008-02-14 |
| CN1988848A (en) | 2007-06-27 |
| NO20070509L (en) | 2007-01-29 |
| EP1778082A1 (en) | 2007-05-02 |
| CA2572287A1 (en) | 2006-02-02 |
| EP1778082A4 (en) | 2010-09-22 |
| NZ552877A (en) | 2010-03-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2005327597B2 (en) | Carboranylporphyrins and uses thereof | |
| US8673265B2 (en) | Carbonylporphyrins and uses thereof | |
| US6951640B2 (en) | Use of novel metalloporphyrins as imageable tumor-targeting agents for radiation therapy | |
| AU2005267390B2 (en) | Carboranylporphyrins and uses thereof | |
| US20070093463A1 (en) | Radiation enhancement agent for X-ray radiation therapy and boron neutron-capture therapy | |
| US20080279781A1 (en) | Glycosylated Carboranylporphyrins and Uses Thereof | |
| US8444953B2 (en) | Symmetric and asymmetric halogen-containing metallocarboranylporphyrins and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2572287 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 180365 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007519285 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580025174.2 Country of ref document: CN Ref document number: 2005267390 Country of ref document: AU Ref document number: 302/KOLNP/2007 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 552877 Country of ref document: NZ Ref document number: 1020077002041 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005785045 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2005267390 Country of ref document: AU Date of ref document: 20050622 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005267390 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020077002041 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005785045 Country of ref document: EP |