WO2010076123A2 - A non-freezing dentifrice composition - Google Patents
A non-freezing dentifrice composition Download PDFInfo
- Publication number
- WO2010076123A2 WO2010076123A2 PCT/EP2009/066506 EP2009066506W WO2010076123A2 WO 2010076123 A2 WO2010076123 A2 WO 2010076123A2 EP 2009066506 W EP2009066506 W EP 2009066506W WO 2010076123 A2 WO2010076123 A2 WO 2010076123A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- weight
- dentifrice composition
- polyethyleneglycol
- dentifrice
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q11/00—Preparations for care of the teeth, of the oral cavity or of dentures; Dentifrices, e.g. toothpastes; Mouth rinses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/345—Alcohols containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/86—Polyethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/20—Chemical, physico-chemical or functional or structural properties of the composition as a whole
- A61K2800/24—Thermal properties
- A61K2800/242—Exothermic; Self-heating; Heating sensation
Definitions
- the present invention relates to dentifrice compositions.
- Thermal-warming agents are heat-generating substances, i.e. exothermic agents, (e.g. those which generate heat upon hydration). Consumer satisfaction with a product is likely to be high if some type of sensory signal exists to remind the consumer that the product is working; and it is inter-alia for this reason that warm ing agents are included i n oral-care compositions; more particularly in dentifrices.
- thermal warming agents are synthetic Zeolites, Sodium carbonate, Calciu m chloride, Magnesium sulphate, and Polyols such as Polyethyleneglycol (commonly and hereinafter referred to as PEG), Glycerol and Sorbitol.
- chemo-warming agents include Vanillin and related compounds, Pepper extract, Capsicum extract and Zingiber extract.
- Dentifrices having a combination of two warming agents are also known in the art.
- WO 1 997/049374 (Henkel KGAA) discloses heating toothpastes having 20-85 weight% polyol of the Ethylene glycol, Propylene glycol or Glycerine group and 1-20 weight% of a dehydrated salt selected from Magnesium sulphate, Magnesium chloride or Calcium chloride.
- a further problem identified by the present inventors is that while using PEG, if the amount of water in the composition is not properly controlled, then either the water-soluble actives such as Zinc sulphate and Sodium fluoride, which are usually present in the compositions are poorly solubilised, or if too much water is included , then a large part of PEG gets hydrated in the container (tube) itself, which in effect reduces the amount of active material responsible for warming sensation by de-activating it before actual use.
- the water-soluble actives such as Zinc sulphate and Sodium fluoride
- the dentifrice composition includes a selective combination of Polyethyleneglycol (or a derivative thereof), Glycerine and water.
- It is an object of the present invention to provide a dentifrice composition comprising Polyethyleneglycol:
- the present invention provides a dentifrice composition comprising:
- the molecular mass of Polyethyleneglycol is in the range of 400 to 600 g/mol.
- weight% used throughout the specification means percentage by weight.
- the degree of warming should be such that upon normal use of the dentifrice, the temperature of the dentifrice/saliva mixture and the resultant foam generated in the oral cavity reaches, within 30 seconds, for example, a value of
- the difference between the initial temperature and the increased temperature should be no more than 5 0 C, and more preferably should be in the range of 0.4 to 2 0 C.
- the present inventors have found that by the choice of the amount of Polyethyleneglycol, or the derivative thereof; the d egree of wa rm i n g i n th e ora l cavity ca n be control led . Th e a mou nt of
- Polyethyleneglycol or the derivative thereof is in the range of 50 to 65 weight%, more preferably in the range of 50 to 56 weight%. It is to be noted that if the dentifrice contains optional ingredients which absorb heat upon hydration or solution; the amount of PEG or the derivative thereof will be at the higher end of these ranges to compensate for this heat absorbed and to still provide the self-heating effect.
- the molecular mass of Polyethyleneglycol is in the range of 200 to 600 g/mol, more preferably in the range of 300 to 600 g/mol. It is particularly preferred that the molecular mass is in the range of 400 to 600 g/mol. It has been observed that PEG having molecular mass 600 g/mol is the least bitter of all.
- the composition may include a derivative thereof.
- a preferred derivative of Polyethyleneglycol is Polyethyleneglycol mono methyl ether, further preferably having molecular mass 550 g/mol.
- a combination of PEG of different molecular masses in the abovementioned range, or a combination of PEG and a derivative of PEG may also advantageously be used.
- the dentifrice composition includes 2.5 to 15 weight% Glycerine, which serves as a humectant.
- Preferred level of Glycerine is in the range of 8 to 10 weight%.
- the dentifrice composition must be low in water, which means that it preferably contains 5 to 7 weight% water, more preferably 7% free water, which limit is exclusive of any free water present in the ingredients of the dentifrice.
- the composition includes 0.1 to 0.5 weight% binder selected from Carrageenan or Sodium carboxy methyl Cellulose. It is further preferred that the composition includes 0.1 to 0.4 weight% Sod iu m carboxy methyl Cell ulose (hereinafter referred to as SCMC), as the binder. SCMC is particularly preferred as it gives desired viscosity profile at relatively lower usage level. A combination of Carrageenan and SCMC is also particularly preferred. Synthetic polymers such as polyacrylates and carboxyvinyl polymers such as CARBOPOLTM may also be used.
- the viscosity of the dentifrice composition preferably is in the range of 160000 to 300000 cP, more preferably in the range of 1 80000 to 300000 cP, and most preferably in the range of 180000 to 220000 cP.
- the viscosity is measured at 25 0 C with a BROOKFIELDTM Viscometer using T-bar D-spindle at 5 rpm.
- the composition can be in the form of an opaque chalk/silica based toothpaste or a transparent gel; it is preferred that the dentifrice composition is a gel, as gel- based dentifrices have higher consumer appeal and acceptance.
- the composition may also be a suitable combination of a paste and a gel in the form of dual- compositions, available generally in core-&-sheath, deep-stripe an d co-extruded formats.
- the balance of the dentifrice composition of the invention preferably includes conventional dentifrice ingredients. Such ingredients include cosmetically acceptable carriers like alcohol systems. Small amounts of surfactants, such as anionic, non-ionic or amphoteric surfactants may also be included.
- ingredients may be particulate abrasive materials including agglomerated particulate abrasive materials such as silicas, aluminas, calcium carbonates (both natural and synthetic), di-calcium phosphates, calcium pyrophosphates, hydroxyapatites, trimetaphosphates, and insoluble hexametaphosphates, usually in amounts between 5 to 60 weight%.
- agglomerated particulate abrasive materials such as silicas, aluminas, calcium carbonates (both natural and synthetic), di-calcium phosphates, calcium pyrophosphates, hydroxyapatites, trimetaphosphates, and insoluble hexametaphosphates, usually in amounts between 5 to 60 weight%.
- Flavours such as peppermint and spearmint oils may also preferably be included. It is also preferred to use preservatives, opacifying agents, colouring agents, pH-adjusting agents and sweetening agents.
- the dentifrice composition contains an anti-bacterial agent selected from Copper, Zinc or Stannous salts such as Zinc citrate, Zinc sulphate, Sodium Zinc citrate and Stannous pyrophosphate.
- an anti-bacterial agent selected from Copper, Zinc or Stannous salts such as Zinc citrate, Zinc sulphate, Sodium Zinc citrate and Stannous pyrophosphate.
- anti-bacterial agents which may be included are quaternary ammonium compounds such as cetylpyridinium chloride; bis-biguanides such as chlorhexidine, chlorhexidine digluconate, hexetidine, octenidine, alexidine; TRICLOSANTM and other halogenated bisphenolic compounds such as 2,2' methylenebis-(4-chloro-6-bromophenol).
- a particularly preferred antibacterial agent is Zinc sulphate.
- the composition may also include antioxidants.
- Preferred antioxidants for use in dentifrice compositions are those that are compatible with other components of the composition and are not hazardous to health.
- Preferred antioxidants include ascorbic acid, erythorbic acid, ascorbyl palmitate, thiodipropionic acid, calcium ascorbate, dilauryldithiopropionate, gum guaiac, sodium ascorbate, Butylated hydroxyl Toluene, Butylated hydroxyl Anisole, and tocopherols. Mixtures and combinations of antioxidants can be used. If present, the antioxidant is added in a level effective to reduce or mitigate discoloration that would otherwise result from oxidation of the components of the dentifrice compositions.
- the levels range from about 0.01 to 1 weight%, based on the total weight of the dentifrice composition.
- Polymeric compounds which can enhance the delivery of active ingredients such as anti-bacterial agents can also be included . Examples of such polymers are copolymers of polyvinylmethylether with maleic anhydride and other similar delivery enhancing polymers.
- Anti-caries agents such as sod i u m- and stannous fluoride, aminefluorides, monosodiumfluorophosphate, casein, plaque buffers such as urea, pyruvates, arginine, small peptides, and calcium glycerophosphate may also be included. It is particularly preferred that the anticaries agent is Sodium fluoride.
- the addition of the fluoride containing agent will preferably be at such an amount to provide free fluoride ion at from 100 to 2000 ppm, preferably from 900 to 1500 ppm.
- vitamins such as Vitamin C; and plant extracts.
- Desensitising agents such as potassium tartrate, potassium citrate, potassium chloride, potassium bicarbonate, potassium oxalate, potassium nitrate, calcium phosphates as well as strontium salts, bleaching agents such as peroxy compounds e.g. sodium percarbonate, potassium peroxydiphosphate, effervescing systems such as sodium bicarbonate/citric acid systems, colour change systems, anti-bad breath ingredients may also be included.
- the composition may also include one or more of breath strips, sparkles, large silica particles, granules, beads, and flavour encapsulates for enhanced sensory benefits or for visual appeal.
- Buffers and salts to buffer the pH and ionic strength of the compositions may also be included.
- the heating effect provided by the dentifrice composition according to the invention is generally linked to the efficacy of the product by the consumers.
- composition-A having the formulation as given in Table- 1 below was prepared by the procedure as given in Example-2 below.
- the method of preparation involved two stages:
- Stage-1 Pre-hvdration of SCMC Glycerine and SCMC were separately weighed. SCMC was then gradually added to Glycerine under stirring at room temperature, while ensuring that no lumps were formed, and the powder completely dispersed into the liquid phase giving a clear viscous dispersion. Stirring was discontinued after the addition of SCMC was complete. The resultant clear dispersion was checked for the presence of any un- dissolved matter before proceeding further. This premix was added to pre-weighed water under stirring, until the addition was completed and the mixture was completely homogenized . This pre-hydrated mix was used for preparation of gel-based toothpaste as per the procedure in stage-2. Pre-hydration stage takes about 25 to 30 minutes.
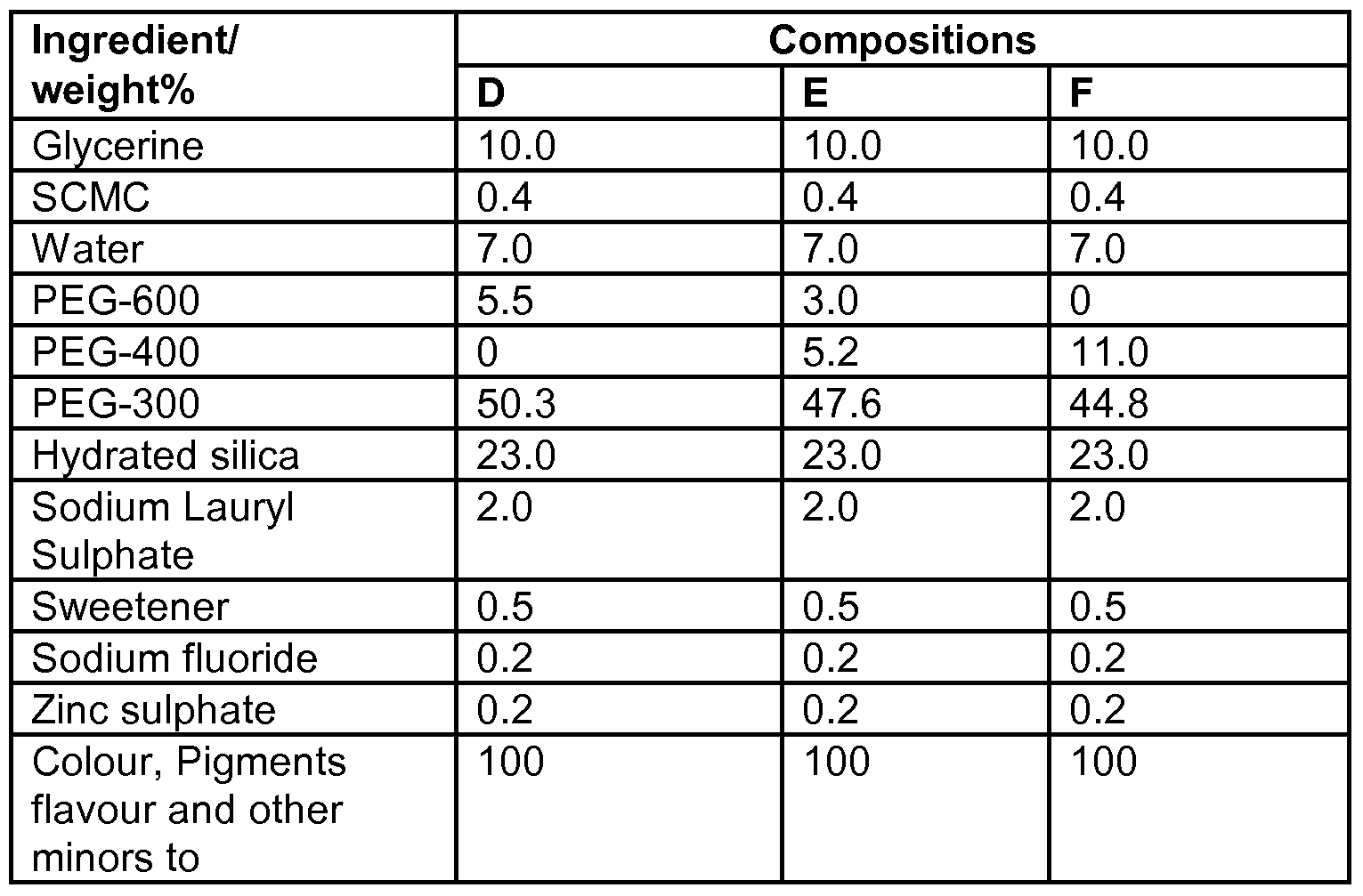
- Dentifrice compositions including PEG-400 and PEG monomethyl ether-550
- compositions were then filled in standard collapsible tubes and the tubes were immersed in ice maintained at -9 0 C, for 30 minutes. After this, it was checked whether the tubes could be manually squeezed or not, and whether the compositions had solidified inside the tubes or not.
- 1 g of each of the compositions at 22.7 0 C was mixed with 1 g water at 37 0 C to observe the increase in temperature. The increase in temperature was recorded for all the compositions. The results have been shown in Table-4 below.
- composition-I the effect of various binders on the viscosity of the compositions was studied by preparing three compositions, in which the formulations were similar to that of Composition-A in Table-1 , except for the change in the binders. Details of the variations in the individual compositions are given in Table-5 below.
- the formulation of the three compositions G, H and I were similar to that of Composition-A in Table-1 , except for the replacement of the binder.
- Composition-I an equivalent amount of water was reduced from Composition-A, as the amount of binder in Composition-I was 0.5 weight%, as against 0.4 weight% in Composition-A.
- viscosity of all the compositions was measured after storing them at 25 0 C and 50% Relative humidity for 14 days. The viscosity was measured at 25 0 C with a BROOKFIELDTM Viscometer using T-bar D-spindle at 5 rpm. This data is also included in Table-5 below.
- Composition-A, Composition-G, and Composition-I had viscosity in the range of 160000 to 300000 cP, which provides for relatively higher physical stability. This viscosity allows for a relatively smoother extrusion of the dentifrice composition from a tube.
- Composition-H had viscosity outside the above range.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009801535792A CN102271651A (en) | 2008-12-30 | 2009-12-07 | A non-freezing dentifrice composition |
| EP09764265A EP2370048A2 (en) | 2008-12-30 | 2009-12-07 | A non-freezing dentifrice composition |
| BRPI0918684A BRPI0918684A2 (en) | 2008-12-30 | 2009-12-07 | toothpaste composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN2720MU2008 | 2008-12-30 | ||
| IN2720/MUM/2008 | 2008-12-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010076123A2 true WO2010076123A2 (en) | 2010-07-08 |
| WO2010076123A3 WO2010076123A3 (en) | 2010-11-25 |
Family
ID=42237379
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/066506 WO2010076123A2 (en) | 2008-12-30 | 2009-12-07 | A non-freezing dentifrice composition |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP2370048A2 (en) |
| CN (1) | CN102271651A (en) |
| BR (1) | BRPI0918684A2 (en) |
| WO (1) | WO2010076123A2 (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007019719A1 (en) | 2005-08-15 | 2007-02-22 | Givaudan Sa | Cooling compounds |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3689637A (en) * | 1969-07-11 | 1972-09-05 | Lever Brothers Ltd | Dentifrice composition |
| US4490353A (en) * | 1983-07-13 | 1984-12-25 | Colgate-Palmolive Company | Antiplaque dentifrice with improved fluoride stability |
| US4623537A (en) * | 1984-06-20 | 1986-11-18 | Union Carbide Corporation | Oral hygiene compositions |
| US4837008A (en) * | 1985-04-09 | 1989-06-06 | Peroxydent Group | Periodontal composition and method |
| US4891211A (en) * | 1988-06-29 | 1990-01-02 | Church & Dwight Co., Inc. | Stable hydrogen peroxide-releasing dentifice |
| US5614174A (en) * | 1994-11-14 | 1997-03-25 | Colgate Palmolive Company | Stabilized dentifrice compositions containing reactive ingredients |
| WO1997002802A1 (en) * | 1995-07-10 | 1997-01-30 | Unilever N.V. | Self-heating dentifrice |
| MXPA04005066A (en) * | 2001-11-28 | 2004-08-19 | Procter & Gamble | Dentifrice compositions. |
-
2009
- 2009-12-07 EP EP09764265A patent/EP2370048A2/en not_active Withdrawn
- 2009-12-07 BR BRPI0918684A patent/BRPI0918684A2/en not_active Application Discontinuation
- 2009-12-07 WO PCT/EP2009/066506 patent/WO2010076123A2/en active Application Filing
- 2009-12-07 CN CN2009801535792A patent/CN102271651A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007019719A1 (en) | 2005-08-15 | 2007-02-22 | Givaudan Sa | Cooling compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0918684A2 (en) | 2015-12-01 |
| WO2010076123A3 (en) | 2010-11-25 |
| CN102271651A (en) | 2011-12-07 |
| EP2370048A2 (en) | 2011-10-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TW585778B (en) | Oral composition comprising porous calcium carbonate | |
| AU773109B2 (en) | Starch thickened non-aqueous dentifrices | |
| JP5320776B2 (en) | Toothpaste products | |
| JP5625647B2 (en) | Dentifrice composition | |
| EP2438901B1 (en) | Dentifrice | |
| JP2006347987A (en) | Composition for oral cavity and method for enhancing foamability of composition for oral cavity | |
| JP2003292426A (en) | Dentifrice composition | |
| JP4985907B2 (en) | Toothpaste composition | |
| JP3854719B2 (en) | Oral composition | |
| JP2001031542A (en) | Composition for oral cavity | |
| EP3076926B1 (en) | Oral care whitening compositions containing fatty amphiphiles | |
| EP2370048A2 (en) | A non-freezing dentifrice composition | |
| JP2009120552A (en) | Method for manufacturing toothpaste agent composition | |
| JP5516796B2 (en) | Dentifrice composition | |
| MX2007001031A (en) | Oral composition. | |
| JP4131732B2 (en) | Oral composition | |
| KR102440320B1 (en) | Perfume beads and toothpaste comprising the same, and the preparation method thereof | |
| JP5716649B2 (en) | Dentifrice composition | |
| JP2018203714A (en) | Oral composition | |
| JP7363815B2 (en) | Oral composition | |
| JP5470838B2 (en) | Dentifrice preparation | |
| JP7000823B2 (en) | Toothpaste composition | |
| JP3482323B2 (en) | Oral composition | |
| JP5720416B2 (en) | Gel oral composition and method for producing the same | |
| JP6007696B2 (en) | Dentifrice composition and method for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980153579.2 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09764265 Country of ref document: EP Kind code of ref document: A2 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2009764265 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12011501302 Country of ref document: PH |
|
| NENP | Non-entry into the national phase in: |
Ref country code: DE |
|
| ENP | Entry into the national phase in: |
Ref document number: PI0918684 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110630 |