WO2010107494A1 - Expandable tfe copolymers, method of making, and porous, expanded articles thereof - Google Patents
Expandable tfe copolymers, method of making, and porous, expanded articles thereof Download PDFInfo
- Publication number
- WO2010107494A1 WO2010107494A1 PCT/US2010/000811 US2010000811W WO2010107494A1 WO 2010107494 A1 WO2010107494 A1 WO 2010107494A1 US 2010000811 W US2010000811 W US 2010000811W WO 2010107494 A1 WO2010107494 A1 WO 2010107494A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- copolymer
- tfe
- comonomer
- microstructure
- added
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
- C08F214/26—Tetrafluoroethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/16—Making expandable particles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/04—Hollow or tubular parts of organs, e.g. bladders, tracheae, bronchi or bile ducts
- A61F2/06—Blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/16—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/507—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials for artificial blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/56—Porous materials, e.g. foams or sponges
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/04—Macromolecular materials
- A61L31/048—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/146—Porous materials, e.g. foams or sponges
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C55/00—Shaping by stretching, e.g. drawing through a die; Apparatus therefor
- B29C55/005—Shaping by stretching, e.g. drawing through a die; Apparatus therefor characterised by the choice of materials
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F14/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F14/18—Monomers containing fluorine
- C08F14/26—Tetrafluoroethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F210/00—Copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
- C08F214/24—Trifluorochloroethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
- C08F214/26—Tetrafluoroethene
- C08F214/262—Tetrafluoroethene with fluorinated vinyl ethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
- C08F214/26—Tetrafluoroethene
- C08F214/265—Tetrafluoroethene with non-fluorinated comonomers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F259/00—Macromolecular compounds obtained by polymerising monomers on to polymers of halogen containing monomers as defined in group C08F14/00
- C08F259/08—Macromolecular compounds obtained by polymerising monomers on to polymers of halogen containing monomers as defined in group C08F14/00 on to polymers containing fluorine
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29K—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES B29B, B29C OR B29D, RELATING TO MOULDING MATERIALS OR TO MATERIALS FOR MOULDS, REINFORCEMENTS, FILLERS OR PREFORMED PARTS, e.g. INSERTS
- B29K2027/00—Use of polyvinylhalogenides or derivatives thereof as moulding material
- B29K2027/12—Use of polyvinylhalogenides or derivatives thereof as moulding material containing fluorine
- B29K2027/18—PTFE, i.e. polytetrafluorethene, e.g. ePTFE, i.e. expanded polytetrafluorethene
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29K—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES B29B, B29C OR B29D, RELATING TO MOULDING MATERIALS OR TO MATERIALS FOR MOULDS, REINFORCEMENTS, FILLERS OR PREFORMED PARTS, e.g. INSERTS
- B29K2105/00—Condition, form or state of moulded material or of the material to be shaped
- B29K2105/04—Condition, form or state of moulded material or of the material to be shaped cellular or porous
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2207/00—Foams characterised by their intended use
- C08J2207/02—Adhesive
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2327/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers
- C08J2327/02—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment
- C08J2327/12—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
- C08J2327/18—Homopolymers or copolymers of tetrafluoroethylene
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/13—Hollow or container type article [e.g., tube, vase, etc.]
- Y10T428/1352—Polymer or resin containing [i.e., natural or synthetic]
- Y10T428/1376—Foam or porous material containing
Definitions
- the invention relates to fluorocopolymers, as defined herein to denote any fluoropolymer containing tetrafluoroethylene monomer units and at least or more than 1.0% by weight of units of at least one other comonomer,* polymerized to produce an expandable tetrafluoroethylene copolymer of the fine powder type.
- fluorocopolymers as defined herein to denote any fluoropolymer containing tetrafluoroethylene monomer units and at least or more than 1.0% by weight of units of at least one other comonomer,* polymerized to produce an expandable tetrafluoroethylene copolymer of the fine powder type.
- a process of polymerization of these monomers is described, as well as the porous products produced by expansion (stretching under controlled conditions) of the aforesaid copolymers.
- TFE tetrafluoroethylene
- Dispersion polymerization of TFE produces a resin that has come to be known as "fine powder". See, e.g., U.S. Patent 4,016,345 (Holmes, 1977).
- sufficient dispersing agent is introduced into a water carrier such that, upon addition of tetrafluoroethylene monomer in the presence of a suitable polymerization initiator
- the present invention relates, specifically, to the aqueous dispersion polymerization technique, in which the product of the polymerization reaction is the copolymer of the invention dispersed within an aqueous colloidal dispersion.
- tetrafluoroethylene monomer is pressured into an autoclave containing water and polymerization initiators, along with paraffin wax to suppress coagulum formation and an emulsifying agent.
- the reaction mixture is agitated and the polymerization is carried out at suitable temperatures and pressures.
- Polymerization results in the formation of an aqueous dispersion of polymer particles, and the dispersed polymer particles may subsequently be coagulated by techniques known in the art to obtain what has become known as the fine powder form of the polymer.
- Fine powder resins are known to be useful in paste extrusion processes and in stretching (expansion) processes in which the paste-extruded extrudate, after removal of extrusion aid lubricant, is stretched to produce porous, strong products of various cross-sectional shapes such as rods, filaments, sheets, tubes, etc.
- stretching process is disclosed in the pioneering U.S. Pat. No. 3,953,566 (Gore, 1976), assigned commonly with the instant invention.
- copolymer as it has been used in connection with fluoropolymers in the prior art has been inconsistently applied.
- the normal convention of polymer science will be followed, and the term “copolymer” will apply to any fluoropolymer containing more than 1.0% by weight of at least one comonomer in addition to TFE.
- a fluoropolymer containing less than 1.0% comonomer is properly categorized as a "modified” homopolymer (Id.), although the term “copolymer” has been misapplied in the literature when referring, in fact, to "modified” homopolymers.
- the invention provides a true TFE copolymer, of the fine powder type, that is expandable, as defined above, to produce useful, expanded TFE copolymeric products.
- U.S. Patent 4,837,267 discloses a three-component composition termed "core-shell TFE copolymers", which are described as non-melt processible, including chlorotrifluoroethylene (CTFE) monomer residing in the core and having recurring units of a comonomer of perfluoro(n-alkylvinyl) ether of 3-7 carbon atoms (col. 1 , lines 45-55).
- CTFE chlorotrifluoroethylene
- the total comonomer content in the particles is said to be between 0.001 and 2 weight percent.
- the resins are said to be suitable for paste extrusion to produce cable insulation and highly stretchable, unsintered tapes
- This patent describes fluorinated modifying monomers which are capable of copolymerizing with tetrafluoroethylene, such as perfluoropropene, perfluoroalkyl vinyl ether, and halogen- substituted or hydrogen-substituted fluoroolefins.
- the specification cautions that the total amount of the comonomer modifying agent should be so low that the specific properties of the pure polytetrafluoroethylene are retained, that is, there remains no possibility of processing from the melt because of the extremely high melt viscosity for such modified polymers.
- Products disclosed include modified polymer particles having a core of a polymer of "0.05 to 6% by weight" of at least one modifying fluoroolefin comonomer, a first, inner shell, immediately adjacent the core, of TFE units, and a second, outer shell, immediately adjacent the inner shell, of a polymer comprising "0.1 to 15% by weight” o f units of at least one modifying fluoroolefin (col. 3, 1. 5, et seq.).
- Examples of the "three-shell" products provided in this reference for illustration of the principles disclosed therein show that tapes, upon stretching, after removal of lubricant, developed defects or tore completely at relatively modest stretch ratios. For example, the detailed procedure described in Example 31 , at col. 14, 1. 60 to col. 16, 1. 6, produced a product which developed defects at a 4:1 stretch ratio and tore completely at a stretch ratio of 8:1 ('940 patent, Table III).
- PTFE polytetrafluoroethylene
- modified PTFE refers to TFE polymers having such small concentrations of comonomer that the melting point of the resultant polymer is not substantially reduced below that of PTFE.
- concentration of such comonomer is preferably less than 1 weight %, more preferably less than 0.5 weight %.
- the modifying comonomers cited include, for example, hexafluoropropylene (HFP), perfluoro(methyl vinyl ether) (PMVE), perfluoro (propyl vinyl ether) (PPVE), perfluoro ( ethyl vinyl ether) (PEVE), chlorotrifluoroethylene (CTFE), perfluoro-butyl ethylene (PFBE), or other monomer that introduces side groups into the molecule.
- HFP hexafluoropropylene
- PMVE perfluoro(methyl vinyl ether)
- PPVE perfluoro (propyl vinyl ether)
- PEVE perfluoro ( ethyl vinyl ether)
- CTFE chlorotrifluoroethylene
- PFBE perfluoro-butyl ethylene
- copolymer as contrasted with the term “modified homopolymer”, shall mean any fluoropolymer containing more than 1.0% by weight of at least one comonomer in addition to TFE.
- U.S. Patent 6,127,486 (Burger, et. al., 2000) discloses a blend of a fluoropolymer and a "thermoplastic", wherein the "thermoplastic” is said to include a "PTFE copolymer" (col. 4, I. 46).
- the specification instructs that, for the resins described therein, the amount of comonomer is limited such that the [modified] PTFE exhibits properties of "not being processable in the melt.” (Emphasis in original).
- the PTFE is referred to as modified PTFE "in which the comonomers are contained in an amount below 2, preferably 1 wt. % in PTFE.” (CoI. 4, 1. 50)
- No examples are provided of any copolymer having greater than 1.0 weight % of an additional comonomer, and the patent concerns blends of polymers, a different physical form entirely from the true copolymers which form the subject matter of the present invention.
- JP Japanese application
- JP Korean Patent Application
- the modification is said to be primarily performed in order to improve the paste extrudability of fine powders, for example, to reduce extrusion pressure, and the content of polymerization units based on comonomers, while less than 0.5 wt%, is "still comparatively high in substantial terms" (0.1 wt% or higher). Consequently, the product has substantially no melt moldability and possesses markedly reduced crystallinity.
- the reference describes "another drawback", that such modified PTFE becomes less heatresistant because of the structure of the comonomers introduced.
- An object of the present invention is to provide a PTFE product that has excellent extrudability, can be uniformly stretched, and yields high- strength porous articles.
- This objective is then said to be obtained by limiting the introduction of polymerization units based on comonomers copolymerizable with TFE to an amount that has no discernible effect on processibility.
- a porous polymer article is provided, obtained by a process in which a powder composed of the aforementioned modified PTFE is paste-extruded and then stretched at a temperature of 25O 0 C or higher. This reference, however, specifically cautions against polymerization in which the amount of copolymerized monomer exceeds certain limits.
- the application states, again quoting directly:
- the content of the polymerization units based on fluorinated comonomer in the present invention must be rigorously controlled because of considerations related to stretchability.
- the content of the units in the PTFE must fall within a range of 0.005 to 0.05 mol%.
- a content above 0.05 mol% brings about a slight reduction in polymer crystallinity, results in a lower paste extrusion pressure, and has a markedly adverse effect on stretchability.
- a content below 0.005 mol% makes it substantially more difficult to improve the physical properties of a stretched article or to obtain other modification effects.
- a range of 0.01 to 0.04 mol% is particularly preferred.
- Example 4 of this Asahi reference in which a "high” content (by applicant's definition), 0.42wt%, of perfluorobutylethylene comonomer was employed, the paste extrusion pressure was desirably low, and "excellent" extrudability was obtained. However, a test specimen, on stretching, broke. The specification discloses, at this "high” level of comonomer concentration of 0.42 wt%, "...breakage occurred during stretching, and it was impossible to obtain a porous article.” (p. 12, ⁇ 0050).
- the present invention is directed to true TFE copolymers, all containing in excess of 1.0 weight percent comonomer units, all of which are expandable to form porous expanded articles, to a process for their manufacture, and to the expanded articles produced thereby.
- No known prior art reference discloses or suggests such porous, expanded copolymeric articles or the resins from which they are produced.
- a process is provided for the copolymerization of an expandable tetrafluoroethylene (TFE) copolymer of the fine powder type, the copolymer containing 99.0% or less by weight tetrafluoroethylene (TFE) monomer units and at least, or greater than, 1.0% by weight, of units of at least one other comonomer, that is, other than tetrafluoroethylene.
- the other comonomer is an ethylenically unsaturated comonomer having a sufficiently high reactivity ratio to TFE to enable polymerization therewith.
- the process includes the steps of copolymerizing the TFE monomer and the at least one other monomer in a pressurized reactor by feeding 99.0% or less by weight of the TFE monomer into the reactor, feeding at least or greater than 1.0% by weight of the other comonomer into the pressurized reactor, wherein percentages are based upon total weight of monomers fed, initiating polymerization of the monomers with a free radical initiator, and stopping the feeding of the other monomer at a point in time in the polymerization reaction prior to completion of the reaction.
- excess comonomer is removed (evacuated) from the reactor, as needed, prior to completion of the reaction.
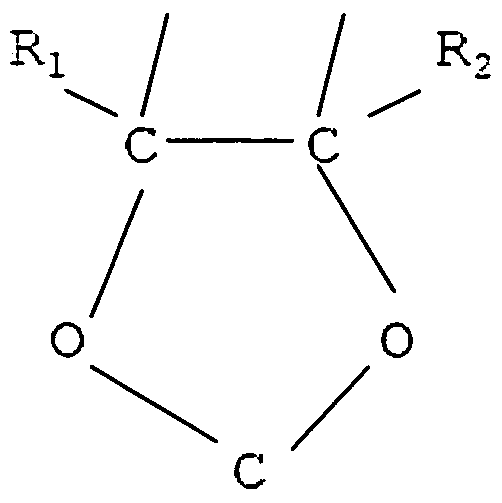
- R 1 and R 2 F or a 1-3 carbon alkyl group containing at least one fluorine, and X, Y may be F 10and/or H; a fluorodioxole of the general formula:
- R f is a perfluoroalkyl carbon of 1-5 atoms, and Ri 1 R 2 may be F and/or CF 3 ;
- R 1 , R 2 may be F and/or a perfluoroalkyl carbon of 1-5 atoms.
- the at least one other comonomer may be a perfluoroalkyl ethylene monomer such as a monomer selected from the group perfluorobutylethylene (PFBE), perfluorohexylethylene (PFHE) and perfluoro-octylethylene (PFOE), or it may be a perfluoroalkyl vinyl ether monomer such as a monomer selected from the group consisting of perfluoro(methyl vinyl ether) (PMVE), perfluoro(ethyl vinyl ether) (PEVE), and perfluoro(propyl vinyl ether) (PPVE). More than one other comonomer may be fed into the pressurized reactor, to produce multicomponent copolymers, i.e., terpolymers, etc.
- the monomer feeds may be introduced as a precharge in the polymerization, or the at least one other comonomer may be introduced incrementally or intermittently during the reaction.
- the process in one embodiment preferably includes stopping the feeding of the at least one other comonomer at less than 90% of the reaction completion.
- Higher concentrations of comonomer in the copolymer produced are achieved by feeding the at least one other comonomer at higher concentration levels, such as at least 1.5% by weight, at least 2.0% by weight, and exceeding 5.0% by weight of the at least one other comonomer to the reactor.
- the aforesaid process produces an expandable tetrafluoroethylene (TFE) copolymer of the fine powder type containing 99.0% or less by weight of polymerized tetrafluoroethylene (TFE) monomer units and at least, or greater than, 1.0% by weight, of polymerized comonomer units of the at least one other comonomer fed into the reaction, based on total weight of polymer produced.
- TFE tetrafluoroethylene
- This true copolymer is expandable to a porous, expanded copolymeric material having a microstructure characterized by nodes 1 interconnected by fibrils 2, as shown in Fig. 1 , described more fully below. Further views of alternative unique node, 1, and fibril, 2, microstructures are shown in Figs. 2 and 3.
- the copolymer produced may include more than one other polymerized comonomer, and the comonomer content in the copolymer always exceeds 1.0% by weight, may exceed 1.5% by weight polymerized units of the other comonomer and, indeed, may exceed 5.0 weight % of polymerized units of the other comonomer(s).
- copolymer materials are produced which exhibit unique adhesion characteristics which cannot be achieved in PTFE homopolymers. That is, the copolymer can be adhered to itself or other materials after subjecting it to lower temperature and/or shorter time and/or lower pressure than what is required for adhering PTFE homopolymer to itself. For example, as described later herein with respect to room temperature adhesion testing, this adhesion, or bonding, can be achieved at temperatures at or below about 29O 0 C with these unique copolymers (hence, at lower temperatures than required for PTFE homopolymers).
- the copolymer of the invention is produced in the form of fine particles dispersed within an aqueous medium which may be coagulated using known techniques to produce fine powder resins.
- Porous, expanded TFE copolymer materials having a microstructure of nodes interconnected by fibrils are further provided according to the invention.
- These porous, expanded copolymeric materials can be produced in the form of shaped articles such as sheets or films, tubes, rods, and continuous filaments, and these articles are generally strong, that is, their matrix tensile strengths in at least one direction exceed 5,000 psi. Matrix tensile strengths in at least one direction can, for certain products, exceed 30,000 psi, thus providing extremely strong, porous, true copolymeric expanded TFE articles useful in many applications.
- the copolymer of the invention is produced in the form of fine particles dispersed within an aqueous medium which may be coagulated using known techniques to produce fine powder resins.
- Porous, expanded TFE copolymer materials having a microstructure of nodes interconnected by fibrils are further provided according to the invention.
- These porous, expanded copolymeric materials can be produced in the form of shaped articles such as sheets or films, tubes, rods, and continuous filaments, and these articles are generally strong, that is, their matrix tensile strengths in at least one direction exceed 5,000 psi. Matrix tensile strengths in at least one direction can, for certain products, exceed 30,000 psi., thus providing extremely strong, porous, true copolymeric expanded TFE articles useful in many applications.
- such expanded TFE materials may be compressed or otherwise processed to achieve a reduction in porosity utilizing processing techniques known in the art.
- the copolymer of the present invention can be used in a wide variety of medical and commercial devices.
- Medical devices include the incorporation of the inventive copolymer into long and short term implantable devices, as well as in disposable, or single use, supplies and devices. These devices include, but are not limited to, vascular grafts (to repair, replace, bypass or augment a blood vessel or another vascular graft), other shunting conduits, surgical and laparoscopic sheets and patches, endoluminal prostheses (e.g., stent-g rafts), components of cell containment devices, and substrates for drug delivery, catheters, space filling or augmentation devices, joint spacers, surface coatings for devices, lenses, work surface or clean room surface coatings, seals, gaskets, blood contact surfaces, bags, containers and fabric liners.
- vascular grafts to repair, replace, bypass or augment a blood vessel or another vascular graft
- other shunting conduits surgical and laparoscopic sheets and patches
- Fig. 1 is a SEM photomicrograph of an expanded sheet of a copolymeric resin produced according the invention herein, taken at 200X magnification, showing the node 1 and fibril 2 microstructure of this material, the respective nodal intersections being interconnected by the multiplicity of fibrils 2;
- Fig. 2 is a SEM photomicrograph of the expanded beading specimen of the copolymeric resin produced in Example 6, taken at 200X magnification, showing the node 1 and fibril 2 microstructure of this material, the respective nodal intersections being interconnected by the multiplicity of fibrils 2;
- Fig. 3 is another SEM photomicrograph of the expanded sheet specimen of the copolymeric resin produced in Example 6, taken at 20,00OX magnification, showing a node 1 and fibril 2 microstructure;
- Figure 4 includes differential scanning calorimetry (DSC) scans showing the melt transition temperature peaks of the materials of Examples 10, 12 and 13, as well as that of a comparative PTFE homopolymer.
- DSC differential scanning calorimetry
- a process for the polymerization of a true tetrafluoroethylene (TFE) copolymer of the fine powder type is provided, wherein the copolymer contains polymerized comonomer units of at least one comonomer other than TFE in concentrations of at least or exceeding 1.0 weight percent, and which can exceed 5.0 weight percent, wherein the copolymer is expandable, that is, the copolymer may be expanded to produce strong, useful, expanded TFE copolymeric articles having a microstructure of nodes interconnected by fibrils.
- TFE true tetrafluoroethylene
- the copolymer of this invention is produced by a polymerization process wherein the copolymerization reaction is started by a suitable initiator, after which initiator addition is stopped, allowing the reaction to slow down and proceed to completion, at a point between 15% and 90% of the progression of the reaction toward completion.
- the initiator addition is stopped at about the mid-point of the reaction, i.e., at 20-60% to completion.
- Substantially non-telogenic dispersing agents are used.
- Ammonium perfluoro octanoic acid (APFO or "C-8") is one acceptable dispersing agent.
- Programmed addition (precharge and pumping) is known and is preferred. Attention must be paid to ingredient purity to achieve the desired properties in polymerizations as described herein. Ionic impurities, which can increase ionic strength, in addition to soluble organic impurities, which can cause chain transfer or termination, must be minimized. It is clearly important to employ ultra pure water in all such polymerization reactions.
- the break strength associated with an extruded and expanded (stretched) TFE polymeric beading produced from a particular resin is directly related to that resin's general suitability for expansion, and various methods have been employed to measure break strength.
- the following procedure was used to produce and test expanded beading specimens made from the copolymers of this invention, the data for which are reported hereinbelow.
- 113.4 g of fine powder resin is blended together with 130 cc/lb (24.5 g) of Isopar® K.
- the blend is aged for about 2 hours at 22°C in a constant temperature water bath.
- a 1-in. diameter cylindrical preform is made by applying about 270 psig of preforming pressure for about 20 seconds.
- the preform is inspected to ensure it is crack free.
- An extruded beading is produced by extruding the preformed, lubricated resin through a 0.100 in. diameter die having a 30 degree included inlet angle.
- the extruder barrel is 1-in. in diameter and the ram rate of movement is 20 in./min.
- the -extruder barrel and die are at room temperature, maintained at 23 0 C, plus or minus 1.5°C.
- the Isopar K is removed from the beading by drying it for about 25 minutes at 225-230 0 C. Approximately the first and last 8 ft. of the extruded beading are discarded to eliminate end effects.
- section of the extruded beading is expanded by stretching at 290 0 C to a final length of 50 in. (expansion ratio of 25:1) and at an initial rate of stretch of 100% per second, which is a constant rate of 2 in. per second.
- Approximately a 1 ft. length from near the center of the expanded beading is removed, and the maximum break load of the removed sample held at room temperature (23°C plus or minus 1.5°C) is measured using an I nstron® tensile tester using an initial sample length of 2 in and a crosshead speed of 12 in/min. Measurements in duplicate are obtained and reported as the average value for the two samples.
- This procedure is similar to that described in U.S. Patent 6,177,533B1.
- the expansion here is carried out at 290 0 C instead of 300°C.
- Core-shell resin structures containing polymerized monomers additional to TFE have been known for some time. See, e.g., U.S. Patents 4,576,869 (Malhotra), 6,541.589B1 (Baillie) and 6,841 ,594B2 (Jones).
- the resins produced according to the present invention are all true copolymers, i.e., comonomer content exceeding 1.0 weight percent, verified using solid state NMR spectroscopy, as well as mass balance and detection of residual monomer in the gas phase of the polymerization batch, through gas chromatography.
- compositions are all expandable to a stretch ratio of at least 25:1 , to form expanded copolymeric articles having their unique node, 1, and fibril, 2, microstructure as shown in Fig. 1, verifiable through SEM examination, as demonstrated below. Further views of alternative unique node, 1, and fibril, 2, microstructures are shown in Figs. 2 and 3.
- Characterization of copolymer materials can be performed via standard analytical techniques available in the art including, but not limited to, DCS, NMR (including fluorine, proton, carbon and other known NMR techniques), TGA, IR, FTIR, Raman spectroscopy, and other suitable techniques.
- DSC Differential Scanning Calorimetry
- the sample was loaded into the pan and the weight was recorded to 0.01 mg precision, with samples ranging from 5.00mg to lO.OOmg. These values were entered into the Thermal Advantage control software for the Q2000.
- the lid was placed on the pan and crimped using a standard press.
- a similar pan for reference was prepared, with the exception of the sample article, and its weight was also entered into the software.
- the pan containing the sample article was loaded onto the sample sensor in the Q2000 and the empty pan was loaded onto the reference sensor.
- the samples were then equilibrated at -5O 0 C and ramped at 20°C/min to 400 0 C. Data were analyzed using Universal Analysis 2000 V.3.9A from TA Instruments.
- the reactor was heated to 83 C and agitated at 60 rpm. Subsequently, 0.8 MPa of VDF was added followed by addition of TFE until the pressure reached 2.8 MPa. At this time, KMNO4 in a Dl water solution (0.063g/L) was injected at 80ml_/min until approximately 2 kg of TFE wa s added. After addition of the 2nd Kg of TFE, the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa. The KMnO4 was added at 20mL ⁇ nin for the 3rd Kg of TFE and further reduced to IOmUmin for the 4th Kg of TFE. After the 4th Kg of TFE was added, KMnO4 was no longer added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 14.3Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 44.73 Kg containing 32.6% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.296 microns and the standard specific gravity was 2.156.
- the VDF concentration in the copolymer was measured to be 3.48 mol% (2.26 wt %).
- the break strength of the beading was 6.6 lbs.
- the matrix tensile strength of the specimen was measured to be 37,299 psi.
- TrFE trifluoroethylene
- KMNO4 in a Dl water solution 0.1 g/L was injected at 80ml_/min until approximately 0.5 kg of TFE was consumed.
- the rate was reduced to 40 mL/min until a second Kg of TFE was consumed.
- the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa.
- the KMnO4 was again added at 40mL/min for the next 0.5 Kg of TFE and continued until 4 Kg of TFE was consumed. After 4 Kg of TFE was consumed, KMnO4 was no longer added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 45.74 Kg containing 35.8% solids.
- the dispersion was coagulated with Nitric acid and dried at 170 C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.283 microns and the standard specific gravity was 2.213.
- the trifluoroethylene concentration in the copolymer was measured to be 3.2 mol% (2.6 wt %).
- the break strength of the beading specimen was 7.24 lbs.
- the matrix tensile strength of the specimen was measured to be 28,602 psi.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 42.76 Kg containing 29.0% solids.
- the dispersion was coagulated with Nitric acid and dried at 17O 0 C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.263 microns and the standard specific gravity was 2.157.
- the VDF concentration in the copolymer was measured to be 4.30 mol% (2.80 wt %).
- the PFBE concentration in the copolymer was measured to be 0.03 mol % (0.07 wt %), yielding a total copolymer concentration in the composition of 2.87 wt%.
- the break strength of the beading specimen was 13.6 lbs.
- the matrix tensile strength of the specimen was measured to be 44,878 psi.
- EXAMPLE 4 To a 50-liter, horizontal polymerization reactor equipped with a 3-bladed agitator was added 1.5 Kg wax , 28 Kg of deionized (Dl) water, 18g of ammonium perfluorooctanoic acid (APFO) and 5g of succinic acid dissolved in about 5Og of Dl water. The reactor and contents were heated above the melting point of the wax. The reactor was repeatedly evacuated and pressurized (to about 1 Atm or less) with TFE until the oxygen level was reduced to 20 ppm or less. The contents were briefly agitated at about 60 rpm between evacuation and purge cycles to ensure that the water was deoxygenated.
- Dl deionized
- APFO ammonium perfluorooctanoic acid
- the KMnO4 was again added at 40ml_/min until an additional 0.5 Kg of TFE was consumed and reduced to 20ml_/min until 4Kg of TFE was consumed. After the 4th Kg of TFE wa s added, KMnO4 was no longer added.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.240 microns and the standard specific gravity was 2.159.
- the VDF concentration in the copolymer was measured to be 3.50 mol% (2.20 wt %).
- the PFOE concentration in the copolymer was measured to be 0.03 mol % (0.16 wt %), yielding a total copolymer concentration in the composition of 2.36 wt %.
- the break strength of the beading specimen was 14.1 lbs.
- the matrix tensile strength of the specimen was measured to be 48,236 psi.
- KMnO4 was again added at 80ml_/min until an additional 1 Kg of TFE was consumed at which time it was reduced to 40ml_/min until 4Kg of TFE was consumed. After the fourth Kg of TFE was consumed the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa. An additional amount of KMnO4 was added at 10ml_/min until the fifth Kg of TFE was consumed. After the consumption of the fifth Kg of TFE, no more KMnO4 was added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 48.8 Kg containing 34.5% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.234 microns and the standard specific gravity was 2.151.
- the VDF concentration in the copolymer was measured to be 3.15 mol% (2.04 wt %), and the PFBE concentration in the copolymer was measured to be 0.03 mol % (0.07 wt %), yielding a total copolymer concentration in the composition of 2.11 wt %.
- the break strength of the beading specimen was 8.6 lbs.
- the matrix tensile strength of the specimen was 10 measured to be 31 ,342 psi.
- TFE was added until the pressure reached 2.8 MPa.
- KMnO4 in a Dl water solution (0.063g/L) was injected at 80ml_/min until approximately 1 kg of TFE was added.
- the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with 0.8 MPa of VDF followed by addition of TFE until the pressure reached 2.8 MPa.
- the KMnO4 was again added at 80mL/min until an additional 2 Kg of TFE was consumed at which time it was reduced to 40ml_/min until 4Kg of TFE was consumed.
- the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa.
- An additional amount of KMnO4 was added at 40ml_/min until the fifth Kg of TFE was consumed. After the consumption of the fifth Kg of TFE, no more KMnO4 was added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 46.86 Kg containing 35.0% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.265 microns and the standard specific gravity was 2.158.
- the VDF concentration in the copolymer was measured to be 3.35 mol% (2.17 wt %).
- the break strength of the beading specimen was 6.6 lbs.
- An SEM of the microstructure of the beading specimen is shown in Figure 2.
- the matrix tensile strength of the specimen was measured to be 26,053 psi.
- the copolymer material formed in this example was then blended with lsopar K (Exxon Mobil Corp., Fairfax, VA) in the proportion of 0.196 g/g of fine powder.
- the lubricated powder was compressed into a cylinder to form a pellet and placed into an oven set at 49 0 C for approximately 12 hours.
- the compressed and heated pellet was ram extruded to produce a tape approximately 16.0 cm wide by 0.73 mm thick.
- the extruded tape was then rolled down between compression rolls to a thickness of 0.256 mm.
- the tape was then transversely stretched to approximately 56 cm wide (i.e., at a ratio of 3.5: 1 ) and dried at a temperature of 25O 0 C.
- the dry tape was longitudinally expanded between banks of rolls over a heated plate set to a temperature of 345 0 C.
- the speed ratio between the second bank of rolls and the first bank of rolls was 10:1.
- the width of the expanded tape was 12.1 cm.
- the copolymer was measured to be 4.2 mol% (3.5 wt %), and the PFBE concentration in the copolymer was measured to be 0.03 mol % (0.07 wt %), yielding a total copolymer concentration in the composition of 3.57 wt %.
- the break strength of the beading specimen was 3.48 lbs.
- the matrix tensile strength of the specimen was measured to be 13,382 psi.
- the reactor was heated to 83° C and agitated at 60 rpm. Subsequently, TFE was added until the pressure reached 2.8 MPa. At this time, KMnO4 in a Dl water solution (0.063g/L) was injected at 80mUmin unt i I approximately 1 kg of TFE was added. At this time the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with 0.8 MPa of TrFE followed by addition of TFE until the pressure reached 2.8 MPa. The KMnO4 was again added at 80ml_/min until an additional 3 Kg of TFE was consumed.
- the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa.
- An additional amount of KMnO4 was added at 40mL/min until the fifth Kg of TFE was consumed. After the consumption of the fifth Kg of TFE, no more KMnO4 was added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 47.22 Kg containing 34.8% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C .
- the raw dispersion particle size (RDPS) of the polymer particle was 0.276 microns and the standard specific gravity was 2.219.
- the matrix tensile strength of the specimen was measured to be 15,329 psi.
- the reactor was heated to 83° C and agitated at 6 0 rpm. Subsequently, TFE was added until the pressure reached 2.8 MPa. At this time, KMnO4 in a Dl water solution (0.063g/L) was injected at ⁇ OmUmin until approximately 1 kg of TFE was added. At this time the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with 1.2 Kg of HFP followed by addition of TFE until the pressure reached 1.9 MPa. The KMnO4 was again added at 80ml_/min until an additional three Kg of TFE was consumed.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 48.54 Kg containing 30.4% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.302 microns and the standard specific gravity was 2.157.
- the HFP concentration in the copolymer was measured to be 0.77 mol% (1.25 wt%).
- the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa.
- An additional amount of KMnO4 was added at 40mL/min until the fifth Kg of TFE was consumed. After the consumption of the fifth Kg of TFE, no more KMnO4 was added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 46.9 Kg containing 33.1 % solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.227 microns and the s tandard specific gravity was 2.217.
- the matrix tensile strength of the specimen was measured to be 34,178 psi.
- EXAMPLE 10 To a 50-liter, horizontal polymerization reactor equipped with a 3-bladed agitator was added 1.5 Kg wax, 28 Kg of deionized (Dl) water, 18g of ammonium perfluorooctanoic acid (APFO), 0.2g FeSO4 and 5g of succinic acid dissolved in about 5Og of Dl water. The reactor and contents were heated above the melting point of the wax. The reactor was repeatedly evacuated and pressurized (to about 1 Atm or less) with TFE until the oxygen level was reduced to 20 ppm or less. The contents were briefly agitated at about 60 rpm between evacuation and purge cycles to ensure that the water was deoxygenated.
- Dl deionized
- APFO ammonium perfluorooctanoic acid
- FeSO4 succinic acid
- the reactor was heated to 83° C and agitated at 60 rpm. Subsequently, 0.81 MPa of CTFE was added followed by addition of TFE until the pressure reached 2.8 MPa. At this time, a solution containing 3g ammonium persulfate and 3g sodium hydrosulfite in 200OmL of Dl water was injected at 40mL/min until 2Kg of TFE was consumed. After addition of the second Kg of TFE, the pressure in the reactor was reduced to 50 Kpa using vacuum and pressurized with fresh TFE to 2.8 MPa. Additional initiator solution was again added at 20ml_/Min until a total of 2.5Kg of TFE was consumed. At this time the rate was reduced to 10mL/min. After 3Kg of total TFE was consumed no more initiator was added.
- the polymerization reaction was then allowed to continue and the reaction stopped after 16Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 48.07 Kg containing 35.0% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C .
- the raw dispersion particle size (RDPS) of the polymer particle was 0.245 microns and the standard specific gravity was 2.228.
- the CTFE concentration in the copolymer was measured to be 3.9 mol% (4.5 wt %).
- the break strength of the beading specimen was 7.6 lbs.
- the matrix tensile strength of the specimen was measured to be 23,991 psi. Adhesion testing was performed, and the results are reported in Table 2.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.178 microns and the standard specific gravity was 2.247.
- the CTFE concentration in the copolymer was measured to be 3.1 mol % (3.70 wt %) and the PFBE concentration in the polymer was measured to be 0.03 mol % (0.07 wt %), yielding a total copolymer concentration in the composition of 3.77 wt %.
- the reactor was repeatedly evacuated and pressurized ( to about 1 Atm or less) with TFE until the oxygen level was reduced to 20 ppm or less.
- the contents were briefly agitated at about 60 rpm between evacuation and purge cycles to ensure that the water was deoxygenated.
- the reactor was heated to 83° C, and agitated at 60 rpm. Subsequently, 2.0
- the raw dispersion particle size (RDPS) of the polymer particle was 0.321 microns and the standard specific gravity was 2.137.
- the VDF concentration in the copolymer was measured to be 11.8 mol % (7.90 wt %).
- the break strength of the beading specimen was 10.53 lbs.
- the matrix tensile strength of the specimen was measured to be 37,000 psi.
- Adhesion testing was performed, and the results are reported in Table 2.
- a DSC scan for this material is included in Figure 4, which shows a first melt transition for the material at about 185 0 C.
- the reactor was heated to 83 0 C and agitated at 60 rpm. Subsequently, 2.0 MPa of VDF was added followed by addition of TFE until the pressure reached 2.8 MPa. At this time, KMnO 4 in a Dl water solution (0.1 g/L) was injected at 80mUmin until was approximately 4 kg of TFE were added. The KMnO 4 was added at 40ml_/min during the next 2 Kg TFE addition. After 5 Kg of TFE was consumed an additional 20Og of initiator solution was added. The total amount of KMnO 4 solution added was 3.375 Kg.
- the polymerization reaction was then allowed to continue and the reaction stopped after 9Kg of TFE had been added to the reactor.
- the weight of the dispersion produced was 40.18 Kg containing 19.6% solids.
- the dispersion was coagulated with Nitric acid and dried at 170° C.
- the raw dispersion particle size (RDPS) of the polymer particle was 0.339 microns.
- the VDF concentration in the copolymer was measured to be 23.8 mol% (16.7 wt %).
- the Break strength of the beading specimen was 8.62 lbs.
- the matrix tensile strength of the specimen was measured to be 23,511 psi. Adhesion testing was performed, and the results are reported in Table 2.
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020117024590A KR101705920B1 (en) | 2009-03-20 | 2010-03-17 | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
| CA2754020A CA2754020C (en) | 2009-03-20 | 2010-03-17 | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
| RU2011142289/04A RU2523455C2 (en) | 2009-03-20 | 2010-03-17 | Expandable copolymers tfe, method of their obtaining, and porous, expanded products from these copolymers |
| EP10723366.0A EP2408827B1 (en) | 2009-03-20 | 2010-03-17 | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
| BRPI1013661A BRPI1013661A2 (en) | 2009-03-20 | 2010-03-17 | tfe expandable copolymers, method for producing them and expanded porous articles made therewith |
| JP2012500788A JP5756079B2 (en) | 2009-03-20 | 2010-03-17 | Stretchable TFE copolymer, production method thereof and porous stretched article |
| CN201080013571.9A CN102395611B (en) | 2009-03-20 | 2010-03-17 | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/408,153 | 2009-03-20 | ||
| US12/408,153 US9040646B2 (en) | 2007-10-04 | 2009-03-20 | Expandable TFE copolymers, methods of making, and porous, expanded articles thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010107494A1 true WO2010107494A1 (en) | 2010-09-23 |
Family
ID=42318683
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/000811 WO2010107494A1 (en) | 2009-03-20 | 2010-03-17 | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
Country Status (9)
| Country | Link |
|---|---|
| US (4) | US9040646B2 (en) |
| EP (1) | EP2408827B1 (en) |
| JP (1) | JP5756079B2 (en) |
| KR (1) | KR101705920B1 (en) |
| CN (1) | CN102395611B (en) |
| BR (1) | BRPI1013661A2 (en) |
| CA (1) | CA2754020C (en) |
| RU (1) | RU2523455C2 (en) |
| WO (1) | WO2010107494A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013163179A (en) * | 2011-06-22 | 2013-08-22 | Daikin Industries Ltd | Porous polymer film and production method for porous polymer film |
| WO2016018969A1 (en) * | 2014-07-29 | 2016-02-04 | W.L. Gore & Associates, Inc. | Articles produced from vdf-co-(tfe or trfe) polymers |
| WO2016099914A1 (en) * | 2014-12-19 | 2016-06-23 | W.L. Gore & Associates, Inc. | Dense articles formed from tetrafluroethylene core shell copolymers and methods of making the same |
| WO2016099913A1 (en) * | 2014-12-19 | 2016-06-23 | W.L. Gore & Associates, Inc. | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
| US9650479B2 (en) | 2007-10-04 | 2017-05-16 | W. L. Gore & Associates, Inc. | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
Families Citing this family (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9040646B2 (en) | 2007-10-04 | 2015-05-26 | W. L. Gore & Associates, Inc. | Expandable TFE copolymers, methods of making, and porous, expanded articles thereof |
| EP3753534A1 (en) | 2008-09-29 | 2020-12-23 | Edwards Lifesciences CardiAQ LLC | Heart valve |
| EP2341871B1 (en) | 2008-10-01 | 2017-03-22 | Edwards Lifesciences CardiAQ LLC | Delivery system for vascular implant |
| CN102282301B (en) | 2009-01-16 | 2014-07-30 | Zeus工业品公司 | Electrospinning of ptfe with high viscosity materials |
| US20130268062A1 (en) | 2012-04-05 | 2013-10-10 | Zeus Industrial Products, Inc. | Composite prosthetic devices |
| US8257640B2 (en) | 2009-08-07 | 2012-09-04 | Zeus Industrial Products, Inc. | Multilayered composite structure with electrospun layer |
| CA2961053C (en) | 2009-04-15 | 2019-04-30 | Edwards Lifesciences Cardiaq Llc | Vascular implant and delivery system |
| US8579964B2 (en) | 2010-05-05 | 2013-11-12 | Neovasc Inc. | Transcatheter mitral valve prosthesis |
| WO2012083107A2 (en) * | 2010-12-17 | 2012-06-21 | 3M Innovative Properties Company | Fluorine-containing polymer comprising a sulfinate-containing molecule |
| US9554897B2 (en) | 2011-04-28 | 2017-01-31 | Neovasc Tiara Inc. | Methods and apparatus for engaging a valve prosthesis with tissue |
| US9308087B2 (en) | 2011-04-28 | 2016-04-12 | Neovasc Tiara Inc. | Sequentially deployed transcatheter mitral valve prosthesis |
| US9345573B2 (en) | 2012-05-30 | 2016-05-24 | Neovasc Tiara Inc. | Methods and apparatus for loading a prosthesis onto a delivery system |
| US10583002B2 (en) | 2013-03-11 | 2020-03-10 | Neovasc Tiara Inc. | Prosthetic valve with anti-pivoting mechanism |
| US9681951B2 (en) | 2013-03-14 | 2017-06-20 | Edwards Lifesciences Cardiaq Llc | Prosthesis with outer skirt and anchors |
| US9572665B2 (en) | 2013-04-04 | 2017-02-21 | Neovasc Tiara Inc. | Methods and apparatus for delivering a prosthetic valve to a beating heart |
| EP3075768A4 (en) * | 2013-11-29 | 2017-06-21 | Daikin Industries, Ltd. | Biaxially-oriented porous film |
| TWI631144B (en) * | 2013-11-29 | 2018-08-01 | 大金工業股份有限公司 | Porous body, polymer electrolyte membrane, filter material for filter and filter unit |
| CN103665240B (en) * | 2013-12-11 | 2015-09-30 | 中昊晨光化工研究院有限公司 | The preparation method of polytetrafluoroethyldispersion dispersion resin |
| JP6353686B2 (en) * | 2014-04-10 | 2018-07-04 | 三菱重工業株式会社 | Re-entry machine manufacturing method |
| US20160032069A1 (en) * | 2014-07-29 | 2016-02-04 | W. L. Gore & Associates, Inc. | Porous Articles Formed From Polyparaxylylene and Processes For Forming The Same |
| US9932429B2 (en) * | 2014-07-29 | 2018-04-03 | W. L. Gore & Associates, Inc. | Method for producing porous articles from alternating poly(ethylene tetrafluoroethylene) and articles produced therefrom |
| US10294339B2 (en) * | 2015-01-20 | 2019-05-21 | Daikin Industries, Ltd. | Method for producing modified molded article of fluororesin |
| CN106883337B (en) * | 2015-12-15 | 2019-10-01 | 浙江汉丞科技有限公司 | Dispersion resin can be copolymerized by the high molecular weight tetrafluoroethene of extruding |
| US11479647B2 (en) * | 2016-09-28 | 2022-10-25 | Daikin Industries. Ltd. | Film including a fluoropolymer |
| CN110494516B (en) * | 2017-04-11 | 2022-11-04 | 索尔维特殊聚合物意大利有限公司 | Process for making fluoropolymers |
| CN113038976A (en) | 2018-09-06 | 2021-06-25 | 比奥莫迪克斯有限公司 | Medical tubular device |
| WO2022261579A1 (en) | 2021-06-11 | 2022-12-15 | W. L. Gore & Associates, Inc. | High temperature insulative composites and articles thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4837267A (en) * | 1988-03-21 | 1989-06-06 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene copolymers |
| US6156453A (en) * | 1995-04-10 | 2000-12-05 | Daikin Industries, Ltd. | Water-repelling agent for batteries and battery |
| EP1201689A1 (en) * | 2000-10-30 | 2002-05-02 | Asahi Glass Co., Ltd. | Tetrafluoroethylene polymer for stretching |
| EP1746130A1 (en) * | 2005-07-21 | 2007-01-24 | Solvay Solexis S.p.A. | Fine fluoropolymer powders |
| WO2009045423A1 (en) * | 2007-10-04 | 2009-04-09 | Gore Enterprise Holdings, Inc | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
Family Cites Families (147)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3196194A (en) * | 1964-06-04 | 1965-07-20 | Pennsylvania Fluorocarbon Co I | Fep-fluorocarbon tubing process |
| DE1795078C3 (en) * | 1968-08-07 | 1974-12-19 | Farbwerke Hoechst Ag, Vormals Meister Lucius & Bruening, 6000 Frankfurt | Aqueous polytetrafluoroethylene dispersions, process for their preparation and their use |
| US3655611A (en) | 1968-08-09 | 1972-04-11 | Allied Chem | Cold flow resistant homogeneous polymers of tetrafluoroethylene and hexafluoropropene and process for preparing them |
| CA962021A (en) | 1970-05-21 | 1975-02-04 | Robert W. Gore | Porous products and process therefor |
| JPS4922472A (en) | 1972-06-22 | 1974-02-27 | ||
| US4016345A (en) | 1972-12-22 | 1977-04-05 | E. I. Du Pont De Nemours And Company | Process for polymerizing tetrafluoroethylene in aqueous dispersion |
| US4129618A (en) | 1974-05-16 | 1978-12-12 | Imperial Chemical Industries Limited | Tetrafluoroethylene polymers |
| GB1460535A (en) | 1974-05-16 | 1977-01-06 | Ici Ltd | Tetrafluoroethylene polymers |
| US4036802A (en) * | 1975-09-24 | 1977-07-19 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene copolymer fine powder resin |
| USRE31907E (en) | 1975-09-24 | 1985-06-04 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene copolymer fine powder resin |
| US4194041A (en) | 1978-06-29 | 1980-03-18 | W. L. Gore & Associates, Inc. | Waterproof laminate |
| DE2949907A1 (en) | 1979-12-12 | 1981-06-19 | Hoechst Ag, 6230 Frankfurt | FLUOROPOLYMERS WITH SHELL-MODIFIED PARTICLES AND METHOD FOR THE PRODUCTION THEREOF |
| JPS5692943U (en) | 1979-12-20 | 1981-07-24 | ||
| US4518650A (en) | 1980-07-11 | 1985-05-21 | E. I. Du Pont De Nemours And Company | Protective clothing of fabric containing a layer of highly fluorinated ion exchange polymer |
| US4469744A (en) | 1980-07-11 | 1984-09-04 | E. I. Du Pont De Nemours And Company | Protective clothing of fabric containing a layer of highly fluorinated ion exchange polymer |
| US4381384A (en) | 1981-08-17 | 1983-04-26 | E. I. Du Pont De Nemours And Company | Continuous polymerization process |
| JPS5982144U (en) | 1982-11-27 | 1984-06-02 | 松下電工株式会社 | Architectural corner parts |
| US4792594A (en) | 1982-12-13 | 1988-12-20 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene copolymers |
| US4770927A (en) | 1983-04-13 | 1988-09-13 | Chemical Fabrics Corporation | Reinforced fluoropolymer composite |
| US5230937A (en) | 1983-04-13 | 1993-07-27 | Chemfab Corporation | Reinforced fluoropolymer composite |
| JPS6042446A (en) | 1983-04-28 | 1985-03-06 | イ−・アイ・デユポン・デ・ニモアス・アンド・カンパニ− | Modified polytetrafluoroethylene composition manufactured from dispersion solution |
| JPS60251041A (en) * | 1985-03-26 | 1985-12-11 | ダイキン工業株式会社 | Laminated rubber plug |
| US4576869A (en) | 1984-06-18 | 1986-03-18 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene fine powder and preparation thereof |
| GB2168981B (en) | 1984-12-27 | 1988-07-06 | Asahi Chemical Ind | Porous fluorine resin membrane and process for preparation thereof |
| US4742122A (en) | 1985-10-25 | 1988-05-03 | E. I. Du Pont De Nemours And Company | Melt-processible tetrafluoroethylene/perfluoroolefin copolymers and processes for preparing them |
| US4675380A (en) | 1985-10-25 | 1987-06-23 | E. I. Du Pont De Nemours And Company | Melt-processible tetrafluoroethylene/perfluoroolefin copolymer granules and processes for preparing them |
| JPS62106910A (en) * | 1985-11-01 | 1987-05-18 | Agency Of Ind Science & Technol | Ultraviolet-transmitting film and its production |
| JPS62109846A (en) * | 1985-11-06 | 1987-05-21 | Daikin Ind Ltd | Aqueous dispersion of fluorine-containing copolymer particle and organosol composition of said particle |
| JPS62279920A (en) * | 1986-05-28 | 1987-12-04 | Daikin Ind Ltd | Porous heat-shrinkable tetrafluoroethylene polymer pipe and its manufacture |
| JPS6356532A (en) | 1986-08-27 | 1988-03-11 | Daikin Ind Ltd | Modified polytetrafluoroethylene fine powder and production thereof |
| US4824511A (en) | 1987-10-19 | 1989-04-25 | E. I. Du Pont De Nemours And Company | Multilayer circuit board with fluoropolymer interlayers |
| US4904726A (en) | 1987-12-31 | 1990-02-27 | E. I. Dupont Denemours And Company | Modified polytetrafluoroethylene resins and blends thereof |
| US4952636A (en) | 1987-12-31 | 1990-08-28 | E. I. Du Pont De Nemours And Company | Modified polytetrafluoroethylene resins and blends thereof |
| US4879362A (en) * | 1987-12-31 | 1989-11-07 | E. I. Dupont De Nemours And Company | Modified polytetrafluoroethylene resins |
| US4952630A (en) | 1987-12-31 | 1990-08-28 | E. I. Du Pont De Nemours And Company | Modified polytetrafluoroethylene resins and blends thereof |
| JP2729837B2 (en) | 1988-07-25 | 1998-03-18 | 旭化成工業株式会社 | Polytetrafluoroethylene filament and method for producing the same |
| JP2611400B2 (en) * | 1988-12-12 | 1997-05-21 | ダイキン工業株式会社 | Aqueous fluorinated polymer dispersion and fluorinated polymer organosol composition |
| JPH078926B2 (en) | 1989-12-07 | 1995-02-01 | ダイキン工業株式会社 | Method for producing polytetrafluoroethylene multilayer porous membrane |
| JPH03191100A (en) | 1989-12-21 | 1991-08-21 | Fuji Photo Film Co Ltd | Production of support for printing plate |
| EP0587588B1 (en) | 1991-06-04 | 1998-07-08 | Donaldson Company, Inc. | Fluid treated polytetrafluoroethylene products and their manufacture |
| JP3303408B2 (en) * | 1992-07-09 | 2002-07-22 | ダイキン工業株式会社 | Fluorine-containing resin composite fine particles |
| US5374473A (en) | 1992-08-19 | 1994-12-20 | W. L. Gore & Associates, Inc. | Dense polytetrafluoroethylene articles |
| WO1994005712A1 (en) | 1992-08-28 | 1994-03-17 | E.I. Du Pont De Nemours And Company | Low-melting tetrafluoroethylene copolymer and its uses |
| US5374683A (en) | 1992-08-28 | 1994-12-20 | E. I. Du Pont De Nemours And Company | Low-melting tetrafluoroethylene copolymer and its uses |
| US5547761A (en) | 1992-08-28 | 1996-08-20 | E. I. Du Pont De Nemours And Company | Low melting tetrafluoroethylene copolymer and its uses |
| WO1995014719A1 (en) | 1992-08-28 | 1995-06-01 | E.I. Du Pont De Nemours And Company | Low-melting tetrafluoroethylene copolymer and its uses |
| US5266639A (en) | 1992-08-28 | 1993-11-30 | E. I. Du Pont De Nemours And Company | Low-melting tetrafluorethylene copolymer and its uses |
| JP3172983B2 (en) | 1993-09-20 | 2001-06-04 | ダイキン工業株式会社 | Aqueous dispersion of vinylidene fluoride polymer and process for producing the same |
| CN1084766C (en) | 1994-03-02 | 2002-05-15 | 纳幕尔杜邦公司 | Non-chalking release/wear coating |
| DE69500430T2 (en) | 1994-04-01 | 1998-02-26 | Toagosei Co | Aqueous, fluorine-based paint composition and manufacturing process |
| US5523346A (en) | 1994-06-10 | 1996-06-04 | W. L. Gore & Associates, Inc. | Seeded microemulsion polymerization for the production of small polymer particles |
| DE4424466A1 (en) | 1994-07-12 | 1996-01-18 | Hoechst Ag | Tetrafluoroethylene-ethylene copolymer with a core-shell particle structure |
| JP2992464B2 (en) | 1994-11-04 | 1999-12-20 | キヤノン株式会社 | Covering wire for current collecting electrode, photovoltaic element using the covering wire for current collecting electrode, and method of manufacturing the same |
| US5599614A (en) | 1995-03-15 | 1997-02-04 | W. L. Gore & Associates, Inc. | Integral composite membrane |
| US5547551A (en) | 1995-03-15 | 1996-08-20 | W. L. Gore & Associates, Inc. | Ultra-thin integral composite membrane |
| USRE37701E1 (en) | 1994-11-14 | 2002-05-14 | W. L. Gore & Associates, Inc. | Integral composite membrane |
| USRE37307E1 (en) | 1994-11-14 | 2001-08-07 | W. L. Gore & Associates, Inc. | Ultra-thin integral composite membrane |
| US6133389A (en) | 1995-02-06 | 2000-10-17 | E. I. Du Pont De Nemours And Company | Amorphous tetrafluoroethylene-hexafluoropropylene copolymers |
| DE69632693T2 (en) | 1995-02-06 | 2005-06-16 | E.I. Du Pont De Nemours And Co., Wilmington | Hexafluoropropene containing amorphous copolymers |
| EP0830249B1 (en) | 1995-06-07 | 1999-10-13 | W.L. GORE & ASSOCIATES (UK) LTD | Porous composite |
| EP0835272B1 (en) | 1995-06-30 | 2000-08-30 | E.I. Du Pont De Nemours And Company | Modified polytetrafluoroethylene fine powder |
| US5922468A (en) | 1995-07-13 | 1999-07-13 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene polymer dispersion composition |
| DE69601335T2 (en) * | 1995-08-17 | 1999-08-12 | Du Pont | Tetrafluoroethylene terpolymer |
| US5889104A (en) | 1996-01-11 | 1999-03-30 | W. L. Gore & Associates, Inc. | Low dielectric constant material for use as an insulation element in an electronic device |
| US5756620A (en) | 1995-11-15 | 1998-05-26 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene polymer for improved paste extrusion |
| JP3941128B2 (en) | 1995-12-18 | 2007-07-04 | ダイキン工業株式会社 | Powder coating composition |
| GB9606818D0 (en) | 1996-03-30 | 1996-06-05 | Gore W L & Ass Uk | Granular-type modified polytetrafluoroethlyene dispersions and fused articles prepared therefrom (Case A) |
| US5922425A (en) * | 1996-05-28 | 1999-07-13 | Minnesota Mining And Manufacturing Company | Multi-layer compositions and articles comprising fluorine-containing polymer |
| WO1998007450A2 (en) * | 1996-08-14 | 1998-02-26 | Rtc, Inc. | Membranes suitable for medical use |
| US5866711A (en) | 1996-09-13 | 1999-02-02 | E. I. Du Pont De Nemours And Company | Fluorocyanate and fluorocarbamate monomers and polymers thereof |
| US5969067A (en) | 1996-09-13 | 1999-10-19 | E.I. Dupont De Nemours And Company | Phosphorus-containing fluoromonomers and polymers thereof |
| DE19638416C1 (en) | 1996-09-19 | 1997-11-13 | Gore W L & Ass Gmbh | Microporous fluoro-polymer blend with thermoplastic to increase strength |
| US6114452A (en) | 1996-11-25 | 2000-09-05 | E. I. Du Pont De Nemours And Company | Perfluoroelastomer composition having excellent heat stability |
| US5936060A (en) | 1996-11-25 | 1999-08-10 | E. I. Du Pont De Nemours And Company | Perfluoroelastomer composition having improved processability |
| RU2117459C1 (en) * | 1996-12-23 | 1998-08-20 | Закрытое акционерное общество "Научно-производственный комплекс "Экофлон" | Implanted hollow prosthesis |
| JPH10243976A (en) | 1997-03-04 | 1998-09-14 | Atsushi Kawano | Combustible medical waste processing container |
| US6689833B1 (en) | 1997-04-09 | 2004-02-10 | E. I. Du Pont De Nemours And Company | Fluoropolymer stabilization |
| RU2128024C1 (en) * | 1997-08-07 | 1999-03-27 | Закрытое акционерное общество "Научно-производственный комплекс "Экофлон" | Implanted hollow prosthesis and method of its manufacture |
| US5854382A (en) * | 1997-08-18 | 1998-12-29 | Meadox Medicals, Inc. | Bioresorbable compositions for implantable prostheses |
| US6103361A (en) | 1997-09-08 | 2000-08-15 | E. I. Du Pont De Nemours And Company | Patterned release finish |
| US6140410A (en) | 1997-09-09 | 2000-10-31 | E. I. Du Pont De Nemours And Company | Fluoropolymer composition |
| US6312814B1 (en) | 1997-09-09 | 2001-11-06 | E. I. Du Pont De Nemours And Company | Fluoropolymer laminate |
| US6166138A (en) | 1997-09-09 | 2000-12-26 | E. I. Du Pont De Nemours And Company | Fluoropolymer composition |
| DE69835649T2 (en) | 1997-10-15 | 2007-09-13 | E.I. Dupont De Nemours And Co., Wilmington | Copolymers of maleic acid or its anhydride and fluorinated olefins |
| DE69823051T2 (en) | 1997-10-24 | 2005-03-17 | Daikin Industries, Ltd. | AQUEOUS RESIN DISPERSION COMPOSITION |
| JP3669172B2 (en) | 1997-12-25 | 2005-07-06 | 旭硝子株式会社 | Tetrafluoroethylene copolymer, production method thereof and use thereof |
| JP3613024B2 (en) | 1997-12-26 | 2005-01-26 | 旭硝子株式会社 | Tetrafluoroethylene copolymer for stretching and its use |
| US6025092A (en) | 1998-02-13 | 2000-02-15 | E. I. Du Pont De Nemours And Company | Fluorinated ionomers and their uses |
| US6197904B1 (en) | 1998-02-26 | 2001-03-06 | E. I. Du Pont De Nemours And Company | Low-melting tetrafluoroethylene copolymer |
| US6232372B1 (en) | 1998-03-18 | 2001-05-15 | E. I. Du Pont De Nemours And Company | Multicomponent particles of fluoropolymer and high temperature resistant non-dispersed polymer binder |
| US6191208B1 (en) | 1998-05-20 | 2001-02-20 | Dupont Dow Elastomers L.L.S. | Thermally stable perfluoroelastomer composition |
| GB2337991A (en) | 1998-06-04 | 1999-12-08 | Gore & Ass | Fine powder type PTFE material |
| WO2000001741A1 (en) | 1998-07-07 | 2000-01-13 | Daikin Industries, Ltd. | Process for producing fluoropolymer |
| US6281296B1 (en) | 1998-08-10 | 2001-08-28 | Dupont Dow Elastomers L.L.C. | Curable perfluoroelastomer composition |
| JP4362258B2 (en) | 1998-08-10 | 2009-11-11 | デュポン パフォーマンス エラストマーズ エルエルシー | Curable perfluoroelastomer composition |
| US6248435B1 (en) | 1998-09-01 | 2001-06-19 | E. I. Du Pont De Nemours And Company | Heat transfer release finish |
| US6177533B1 (en) | 1998-11-13 | 2001-01-23 | E. I. Du Pont De Nemours And Company | Polytetrafluoroethylene resin |
| US6136933A (en) | 1998-11-13 | 2000-10-24 | E. I. Du Pont De Nemours And Company | Process for polymerizing tetrafluoroethylene |
| US7049380B1 (en) * | 1999-01-19 | 2006-05-23 | Gore Enterprise Holdings, Inc. | Thermoplastic copolymer of tetrafluoroethylene and perfluoromethyl vinyl ether and medical devices employing the copolymer |
| US6416698B1 (en) | 1999-02-18 | 2002-07-09 | E. I. Du Pont De Nemours And Company | Fluoropolymer finishing process |
| US6291054B1 (en) | 1999-02-19 | 2001-09-18 | E. I. Du Pont De Nemours And Company | Abrasion resistant coatings |
| US6518349B1 (en) | 1999-03-31 | 2003-02-11 | E. I. Du Pont De Nemours And Company | Sprayable powder of non-fibrillatable fluoropolymer |
| US6403213B1 (en) | 1999-05-14 | 2002-06-11 | E. I. Du Pont De Nemours And Company | Highly filled undercoat for non-stick finish |
| US6395848B1 (en) | 1999-05-20 | 2002-05-28 | E. I. Du Pont De Nemours And Company | Polymerization of fluoromonomers |
| US6429258B1 (en) | 1999-05-20 | 2002-08-06 | E. I. Du Pont De Nemours & Company | Polymerization of fluoromonomers |
| US6806332B2 (en) | 1999-11-12 | 2004-10-19 | North Carolina State University | Continuous method and apparatus for separating polymer from a high pressure carbon dioxide fluid stream |
| US6914105B1 (en) | 1999-11-12 | 2005-07-05 | North Carolina State University | Continuous process for making polymers in carbon dioxide |
| US6770404B1 (en) | 1999-11-17 | 2004-08-03 | E. I. Du Pont De Nemours And Company | Ultraviolet and vacuum ultraviolet transparent polymer compositions and their uses |
| US6824930B1 (en) | 1999-11-17 | 2004-11-30 | E. I. Du Pont De Nemours And Company | Ultraviolet and vacuum ultraviolet transparent polymer compositions and their uses |
| CA2399049A1 (en) | 2000-02-02 | 2001-08-09 | Robert D. Lousenberg | Linear copolymers of fluorocarbon-hydrocarbon monomers synthesized in carbon dioxide |
| US6638999B2 (en) | 2000-02-08 | 2003-10-28 | Dupont Dow Elastomers Llc. | Curable perfluoroelastomer composition |
| TW552435B (en) | 2000-06-12 | 2003-09-11 | Asahi Glass Co Ltd | Plastic optical fiber |
| JP4320506B2 (en) | 2000-09-25 | 2009-08-26 | 旭硝子株式会社 | Polytetrafluoroethylene composition, method for producing the same, and granulated product |
| US6746773B2 (en) * | 2000-09-29 | 2004-06-08 | Ethicon, Inc. | Coatings for medical devices |
| RU2271367C2 (en) * | 2000-10-30 | 2006-03-10 | Асахи Гласс Компани, Лимитед | Stretchable tetrafluoroethylene polymer |
| US6582628B2 (en) | 2001-01-17 | 2003-06-24 | Dupont Mitsui Fluorochemicals | Conductive melt-processible fluoropolymer |
| JP4042390B2 (en) | 2001-03-26 | 2008-02-06 | 旭硝子株式会社 | Method for producing tetrafluoroethylene polymer having excellent strength |
| US6761964B2 (en) | 2001-04-02 | 2004-07-13 | E. I. Du Pont De Nemours And Company | Fluoropolymer non-stick coatings |
| JP5329013B2 (en) | 2001-04-12 | 2013-10-30 | 旭硝子株式会社 | Method for producing high strength tetrafluoroethylene polymer |
| US6541589B1 (en) * | 2001-10-15 | 2003-04-01 | Gore Enterprise Holdings, Inc. | Tetrafluoroethylene copolymer |
| JP4058668B2 (en) | 2001-12-27 | 2008-03-12 | 旭硝子株式会社 | Hard porous molded body of fluororesin |
| BR0215427B1 (en) | 2002-01-04 | 2012-02-22 | dispersion, fluoropolymer powder, coating composition, substrate, self-supporting film and batch process. | |
| BR0215428A (en) | 2002-01-04 | 2004-12-14 | Du Pont | Aqueous dispersion, aqueous dispersion coating composition, coated substrates and self-supporting film |
| US6833418B2 (en) | 2002-04-05 | 2004-12-21 | 3M Innovative Properties Company | Dispersions containing perfluorovinyl ether homopolymers and use thereof |
| US6822059B2 (en) | 2002-04-05 | 2004-11-23 | 3M Innovative Properties Company | Dispersions containing bicomponent fluoropolymer particles and use thereof |
| US6921606B2 (en) | 2002-04-16 | 2005-07-26 | Gore Enterprise Holdings, Inc. | Composite films for electrochemical devices |
| US6870020B2 (en) | 2002-04-30 | 2005-03-22 | E. I. Du Pont De Nemours And Company | High vinyl ether modified sinterable polytetrafluoroethylene |
| ITMI20021561A1 (en) * | 2002-07-16 | 2004-01-16 | Ausimont Spa | COPOLYMERS OF TFE |
| JP2006504844A (en) | 2002-10-31 | 2006-02-09 | スリーエム イノベイティブ プロパティズ カンパニー | Emulsifier-free aqueous emulsion polymerization to produce copolymers of fluorinated olefins and hydrocarbon olefins |
| JP4466002B2 (en) * | 2002-12-06 | 2010-05-26 | 旭硝子株式会社 | Tetrafluoroethylene copolymer, process for producing the same, and paste extrusion molding |
| US7049365B2 (en) | 2003-01-06 | 2006-05-23 | E. I. Du Pont De Nemours And Company | Fluoropolymer sealant |
| US20040173978A1 (en) | 2003-03-06 | 2004-09-09 | Christopher Bowen | PTFE membranes and gaskets made therefrom |
| EP1621573A4 (en) | 2003-04-16 | 2006-08-02 | Kureha Corp | Porous film of vinylidene fluoride resin and method for producing same |
| WO2005061023A1 (en) | 2003-12-12 | 2005-07-07 | C. R. Bard, Inc. | Implantable medical devices with fluorinated polymer coatings, and methods of coating thereof |
| WO2005061567A1 (en) * | 2003-12-22 | 2005-07-07 | Daikin Industries, Ltd. | Non melt processable polytetrafluoroethylene and fine powder thereof |
| JP2005306033A (en) | 2004-03-26 | 2005-11-04 | Daikin Ind Ltd | Polytetrafluorethylene resin film and its manufacturing process |
| DE602005020754D1 (en) | 2004-03-31 | 2010-06-02 | Cook Inc | TRANSPLANT MATERIAL AND VASCOPY THERAPY WITH EXTRACELLULAR COLLAGEN MATRIX AND ITS MANUFACTURING PROCESS |
| US20050238872A1 (en) | 2004-04-23 | 2005-10-27 | Kennedy Michael E | Fluoropolymer barrier material |
| US8012555B2 (en) | 2004-07-21 | 2011-09-06 | Maztech, Inc. | Fluoroplastic composite elastomer |
| US20060047311A1 (en) | 2004-08-26 | 2006-03-02 | Lutz David I | Expanded PTFE articles and method of making same |
| US20060233990A1 (en) * | 2005-04-13 | 2006-10-19 | Trivascular, Inc. | PTFE layers and methods of manufacturing |
| US7531611B2 (en) * | 2005-07-05 | 2009-05-12 | Gore Enterprise Holdings, Inc. | Copolymers of tetrafluoroethylene |
| US20070027551A1 (en) | 2005-07-29 | 2007-02-01 | Farnsworth Ted R | Composite self-cohered web materials |
| US9650479B2 (en) | 2007-10-04 | 2017-05-16 | W. L. Gore & Associates, Inc. | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
| US9040646B2 (en) * | 2007-10-04 | 2015-05-26 | W. L. Gore & Associates, Inc. | Expandable TFE copolymers, methods of making, and porous, expanded articles thereof |
| JP5473824B2 (en) | 2010-08-05 | 2014-04-16 | ニチアス株式会社 | High density polytetrafluoroethylene tape and method for producing the same |
| US9775933B2 (en) | 2012-03-02 | 2017-10-03 | W. L. Gore & Associates, Inc. | Biocompatible surfaces and devices incorporating such surfaces |
-
2009
- 2009-03-20 US US12/408,153 patent/US9040646B2/en active Active
-
2010
- 2010-03-17 EP EP10723366.0A patent/EP2408827B1/en active Active
- 2010-03-17 KR KR1020117024590A patent/KR101705920B1/en active IP Right Grant
- 2010-03-17 WO PCT/US2010/000811 patent/WO2010107494A1/en active Application Filing
- 2010-03-17 BR BRPI1013661A patent/BRPI1013661A2/en not_active IP Right Cessation
- 2010-03-17 RU RU2011142289/04A patent/RU2523455C2/en not_active IP Right Cessation
- 2010-03-17 JP JP2012500788A patent/JP5756079B2/en active Active
- 2010-03-17 CN CN201080013571.9A patent/CN102395611B/en active Active
- 2010-03-17 CA CA2754020A patent/CA2754020C/en active Active
-
2013
- 2013-03-11 US US13/792,398 patent/US8911844B2/en active Active
- 2013-12-04 US US14/096,089 patent/US9193811B2/en active Active
-
2015
- 2015-10-15 US US14/883,772 patent/US9593223B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4837267A (en) * | 1988-03-21 | 1989-06-06 | E. I. Du Pont De Nemours And Company | Tetrafluoroethylene copolymers |
| US6156453A (en) * | 1995-04-10 | 2000-12-05 | Daikin Industries, Ltd. | Water-repelling agent for batteries and battery |
| EP1201689A1 (en) * | 2000-10-30 | 2002-05-02 | Asahi Glass Co., Ltd. | Tetrafluoroethylene polymer for stretching |
| EP1746130A1 (en) * | 2005-07-21 | 2007-01-24 | Solvay Solexis S.p.A. | Fine fluoropolymer powders |
| WO2009045423A1 (en) * | 2007-10-04 | 2009-04-09 | Gore Enterprise Holdings, Inc | Expandable tfe copolymers, method of making, and porous, expanded articles thereof |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9650479B2 (en) | 2007-10-04 | 2017-05-16 | W. L. Gore & Associates, Inc. | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
| JP2013163179A (en) * | 2011-06-22 | 2013-08-22 | Daikin Industries Ltd | Porous polymer film and production method for porous polymer film |
| WO2016018969A1 (en) * | 2014-07-29 | 2016-02-04 | W.L. Gore & Associates, Inc. | Articles produced from vdf-co-(tfe or trfe) polymers |
| US9441088B2 (en) | 2014-07-29 | 2016-09-13 | W. L. Gore & Associates, Inc. | Articles produced from VDF-co-(TFE or TrFE) polymers |
| US10266670B2 (en) | 2014-07-29 | 2019-04-23 | W. L. Gore & Associates, Inc. | Articles produced from VDF-co-(TFE or TrFE) polymers |
| AU2015296585B2 (en) * | 2014-07-29 | 2019-09-12 | W.L. Gore & Associates, Inc. | Articles produced from VDF-co-(TFE or TrFE) polymers |
| US10472491B2 (en) | 2014-07-29 | 2019-11-12 | W. L. Gore & Associates, Inc. | Articles Produced from VDF-co-(TFE or TrFE) Polymers |
| AU2019272051B2 (en) * | 2014-07-29 | 2021-11-25 | W. L. Gore & Associates, Inc | Articles produced from VDF-co-(TFE or TrFE) polymers |
| WO2016099914A1 (en) * | 2014-12-19 | 2016-06-23 | W.L. Gore & Associates, Inc. | Dense articles formed from tetrafluroethylene core shell copolymers and methods of making the same |
| WO2016099913A1 (en) * | 2014-12-19 | 2016-06-23 | W.L. Gore & Associates, Inc. | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
| AU2015363139B2 (en) * | 2014-12-19 | 2018-03-01 | W.L. Gore & Associates Gmbh | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
| AU2015363140B2 (en) * | 2014-12-19 | 2018-07-26 | W.L. Gore & Associates Gmbh | Dense articles formed from tetrafluroethylene core shell copolymers and methods of making the same |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102395611A (en) | 2012-03-28 |
| US20130189464A1 (en) | 2013-07-25 |
| BRPI1013661A2 (en) | 2016-04-26 |
| US20140094557A1 (en) | 2014-04-03 |
| EP2408827A1 (en) | 2012-01-25 |
| JP5756079B2 (en) | 2015-07-29 |
| US9040646B2 (en) | 2015-05-26 |
| RU2011142289A (en) | 2013-05-10 |
| CN102395611B (en) | 2015-07-08 |
| KR101705920B1 (en) | 2017-02-10 |
| RU2523455C2 (en) | 2014-07-20 |
| US20160039989A1 (en) | 2016-02-11 |
| US9193811B2 (en) | 2015-11-24 |
| CA2754020C (en) | 2016-06-07 |
| US20090258958A1 (en) | 2009-10-15 |
| KR20110138247A (en) | 2011-12-26 |
| CA2754020A1 (en) | 2010-09-23 |
| JP2012520920A (en) | 2012-09-10 |
| EP2408827B1 (en) | 2016-07-06 |
| US8911844B2 (en) | 2014-12-16 |
| US9593223B2 (en) | 2017-03-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9593223B2 (en) | Expandable TFE copolymers, method of making, porous, expanded article thereof | |
| EP2201050B1 (en) | Expandable tfe copolymers, method of making, and porous, expanded articles thereof | |
| US6541589B1 (en) | Tetrafluoroethylene copolymer | |
| US9988506B2 (en) | Dense articles formed tetrafluoroethylene core shell copolymers and methods of making the same | |
| EP3233987B1 (en) | Dense articles formed from tetrafluroethylene core shell copolymers and methods of making the same | |
| EP3233985B1 (en) | Dense articles formed from tetrafluoroethylene core shell copolymers and methods of making the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080013571.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10723366 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2754020 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3734/KOLNP/2011 Country of ref document: IN |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010723366 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010723366 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012500788 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20117024590 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2011142289 Country of ref document: RU Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI1013661 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: PI1013661 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110906 |