WO2011005623A1 - Laundry detergent composition comprising low level of bleach - Google Patents
Laundry detergent composition comprising low level of bleach Download PDFInfo
- Publication number
- WO2011005623A1 WO2011005623A1 PCT/US2010/040506 US2010040506W WO2011005623A1 WO 2011005623 A1 WO2011005623 A1 WO 2011005623A1 US 2010040506 W US2010040506 W US 2010040506W WO 2011005623 A1 WO2011005623 A1 WO 2011005623A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- bleach
- surfactant
- tetraazabicyclo
- alkyl
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3942—Inorganic per-compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38609—Protease or amylase in solid compositions only
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38627—Preparations containing enzymes, e.g. protease or amylase containing lipase
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38645—Preparations containing enzymes, e.g. protease or amylase containing cellulase
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
- C11D3/3907—Organic compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3945—Organic per-compounds
Definitions
- the present invention relates to laundry detergent compositions comprising a detersive surfactant and a peroxide bleach.
- Granular laundry detergent compositions need to have a good fabric-cleaning performance, good dispensing and dissolution profiles.
- Some laundry detergent compositions contain bleaches. Some bleaches are too active and sometimes they may damage the fabric during the wash cycle and/or after storage in a certain environment.
- peroxide bleaches are widely used. Peroxide bleaches, e.g., sodium percarbonate and sodium perborate, are beneficial because they can be applied to various kinds of fabrics. However, using a large amount of peroxide bleach may leave risk of runaway thermal degradation in product, for instance, in hot regions.
- Granular laundry detergent compositions typically contain builders, such as zeolite and phosphate.
- Zeolite builders are water-insoluble and their incorporation in laundry detergent compositions leads to poor dissolution of the laundry detergent composition and can also lead to undesirable residues being deposited on the fabric.
- detergent compositions that contain high levels of zeolite builder form undesirable cloudy wash liquors upon contact with water.
- phosphate builders allegedly do not have favourable environmental profiles and their use in laundry detergent compositions is becoming less common, for example, due to phosphate legislation in many countries.
- percarbonate is a source of carbonate anions in the wash liquor, and these carbonate anions can complex with free calcium cations to form calcium carbonate which is insoluble in water. Such calcium carbonate may precipitate on the fabric surface, and can deteriorate the appearance of the fabric.
- high levels of percarbonate in low builder laundry detergent compositions can have very poor bleach stability profile, due to the lack of an adequate water-sink.
- a catalytic bleach such as a transition metal bleach catalyst and/or an imine bleach catalyst can be incorporated therein without jeopardising the fabric care profile of the product.
- the present invention relates to a laundry detergent composition containing (i) from about lwt% to about 60wt% detersive surfactant; (ii) from about Owt% to about 4wt% zeolite builder; (iii) from about Owt% to about 4wt% phosphate builder; (iv) from about above Owt% to about 7wt% peroxide bleach selected from the group consisting of percarbonate and perborate and (v) balance detergent ingredients.
- the low level of bleach allows good bleaching, improves the stability of the product, and enables the use of a bleach catalyst whilst maintaining good fabric integrity profiles.
- the present invention relates to a solid laundry detersive composition.
- the laundry detergent composition contains a solid laundry detersive composition containing: (i) from about lwt% to about 60wt% detersive surfactant; (ii) from about Owt% to about 4wt% zeolite builder; (iii) from about Owt% to about 4wt% phosphate builder; (iv) from about above Owt% to about 7wt% of a peroxide bleach selected from the group consisting of percarbonate and perborate, and (v) balance additional detergent ingredients.
- the laundry detersive composition is suitable for use in the laundering of fabrics.
- the detergent composition contains a low level of a peroxide bleach.
- the composition contains low levels of peroxide bleaches.
- the composition may contain from about above Owt% to about 7wt%, from about lwt% to about 6wt% or from about lwt% to about 5wt%, or to about 4wt%, or even to about 3wt% peroxide bleach.
- the detergent composition can be in the form of a solid; such as in form of free-flowing particles or in the form of a tablet.
- the detergent composition may be in the form of free- flowing particles such as agglomerates, extrudates, spray-dried particles, noodles, needles, flakes and combinations thereof.
- the composition may be a granular laundry detergent composition.
- the detergent composition in free-flowing particulate form typically has a bulk density of from about 450 g/1 to about 1,000 g/1, preferred low bulk density detergent compositions have a bulk density of from about 550 g/1 to about 650 g/1 and preferred high bulk density detergent compositions have a bulk density of from about 750 g/1 to about 900 g/1.
- the composition is typically contacted with water to give a wash liquor having a pH of from about 7 to about 11, or from about 8 to about 10.5.
- composition may be made by any suitable method including agglomeration, spray- drying, extrusion, mixing, dry-mixing, liquid spray-on, roller compaction, spheronisation or any combination thereof.
- composition herein may contain the following ingredients.
- the composition herein contains from about lwt% to about 60wt%, from about 2wt% to about 55wt%, from 3wt% to about 50wt%, or from about 4wt% to about 40wt% detersive surfactant.
- the detersive surfactant may contain an anionic surfactant, a nonionic surfactant, a cationic surfactant, a zwitterionic surfactant, or any mixture thereof.
- the surfactant may be amphoteric, and have a ionic charge dependant on the pH of a typical wash liquor.
- the composition may contain an anionic surfactant.
- the composition may contain from about lwt% to about 40wt%, from about 2wt% to about 30wt%, or from about 5wt% to about 20wt% anionic surfactant.
- the anionic surfactant can be selected from the group consisting of: Ci O -Ci 8 alkyl benzene sulphonates (LAS), preferably linear C 10 -C 13 alkyl benzene sulphonate; C 10 -C 20 primary, branched-chain, linear-chain and random-chain alkyl sulphates (AS), preferred are linear alkyl sulphates, typically having the following formula:
- M is hydrogen or a cation which provides charge neutrality
- preferred cations include sodium and ammonium cations, wherein x is an integer of at least 7, preferably at least 9, y is an integer of at least 8, preferably at least 9
- Ci 0 -Ci 8 alkyl alkoxy carboxylates mid-chain branched alkyl sulphates as described in more detail in US 6,020,303 and US 6,060,443
- Preferred anionic surfactants are: linear or branched, substituted or unsubstituted, Ci 2-I8 alkyl sulphates; and linear or branched, substituted or unsubstituted, Ci O _i 3 alkylbenzene sulphonates, preferably linear Ci 0-O alkylbenzene sulphonates; and mixtures thereof.
- Another preferred anionic surfactant is a Cg- 24 alkylalkoxylated sulphate, preferably an C ⁇ - 24 alkylethoxylated sulphate, preferably having an average degree of ethoxylation of 1 to 5.
- the anionic surfactant may be structurally modified in such a manner as to cause the anionic surfactant to be more calcium tolerant and less likely to precipitate out of the wash liquor in the presence of free calcium ions.
- This structural modification could be the introduction of a methyl or ethyl moiety in the vicinity of the anionic surfactant's head group, as this can lead to a more calcium tolerant anionic surfactant due to steric hindrance of the head group, which may reduce the anionic surfactant's affinity for complexing with free calcium cations in such a manner as to cause precipitation out of solution.
- the anionic surfactant may be in particulate form, such as an agglomerate, a spray-dried powder, an extrudate, a bead, a noodle, a needle or a flake.
- the anionic surfactant, or at least part thereof, may be in a co-particulate admixture with a non-ionic surfactant, this co-particulate admixture may be in spray-dried form.
- the anionic surfactant may be in agglomerate form; the agglomerate may contain at least about 20wt%, of the agglomerate, of an anionic surfactant, or from about 20wt% to about 65wt%, of the agglomerate, of an anionic surfactant.
- Part of the anionic surfactant may be in the form of a spray-dried powder (e.g. a blown powder), and part of the anionic surfactant may be in the form of a non-spray-dried powder (e.g. an agglomerate, or an extrudate, or a flake).
- a linear alkylbenzene sulphonate may be in a co-particulate admixture with soap; this co-particulate admixture may be in spray-dried form.
- composition herein may contain non-ionic surfactant.
- the composition herein may contain from about 0.1wt% to about 15wt%, from about 0.5wt% to about 10wt%, or from about lwt% to about 6wt% non- ionic surfactant.
- the non-ionic surfactant could be an alkyl polyglucoside and/or an alkyl alkoxylated alcohol.
- the non-ionic surfactant may be a linear or branched, substituted or unsubstituted C 8-I8 alkyl ethoxylated alcohol having an average degree of ethoxylation of from 1 to 10.
- the non-ionic surfactant not only provides additional greasy soil cleaning performance but may also increase the anionic surfactant activity by making the anionic surfactant less likely to precipitate out of solution in the presence of free calcium cations.
- the weight ratio of anionic surfactant to non-ionic surfactant may be in the range of from about 0.5:1 to about 20:1, from about 0.5:1 to about 10: 1, or from about 0.5:1 to about 6:1.
- the non-ionic surfactant, or at least part thereof can be incorporated into the composition in the form of a liquid spray-on, wherein the non-ionic surfactant, or at least part thereof, in liquid form (e.g. in the form of a hot-melt) is sprayed onto the remainder of the composition.
- the non-ionic surfactant, or at least part thereof may be in particulate form, and the non-ionic surfactant, or at least part thereof, may be dry-added to the remainder of the composition.
- the non-ionic surfactant may be in the form of a co-particulate admixture with a solid carrier material, such as carbonate salt, inorganic sulphate salt, burkeite, silica or any mixture thereof.
- a solid carrier material such as carbonate salt, inorganic sulphate salt, burkeite, silica or any mixture thereof.
- the non-ionic surfactant, or at least part thereof may be in a co-particulate admixture with either an anionic surfactant or a cationic surfactant. However the non-ionic surfactant, or at least part thereof, may not in a co-particulate admixture with both an anionic surfactant and a cationic surfactant.
- the non-ionic surfactant, or at least part thereof may be agglomerated or extruded with either an anionic surfactant or a cationic surfactant.
- the non-ionic surfactant, or at least part thereof may be in spray-dried powder form, optionally the non-ionic surfactant, or at least part thereof, may be spray-dried with an anionic surfactant.
- the non-ionic surfactant, or at least part thereof may be in a co-particulate admixture with soap, this co-particulate admixture may be in non-spray-dried form, such as an extrudate or an agglomerate.
- the composition may contain from about 0wt% to about 6wt%, from about 0.5wt% to about 4wt%, from about lwt% to about 3wt%, or from about lwt% to about 2wt% cationic surfactant.
- Suitable cationic surfactants are alkyl pyridinium compounds, alkyl quaternary ammonium compounds, alkyl quaternary phosphonium compounds, and alkyl ternary sulphonium compounds.
- the cationic surfactant can be selected from the group consisting of: alkoxylate quaternary ammonium (AQA) surfactants as described in more detail in US 6,136,769; dimethyl hydroxyethyl quaternary ammonium compounds as described in more detail in US 6,004,922; polyamine cationic surfactants as described in more detail in WO 98/35002, WO 98/35003, WO 98/35004, WO 98/35005, and WO 98/35006; cationic ester surfactants as described in more detail in US 4,228,042, US 4,239,660, US 4,260,529 and US 6,022,844; amino surfactants as described in more detail in US 6,221,825 and WO 00/47708,
- R is a linear or branched, substituted or unsubstituted C 6-I8 alkyl or alkenyl moiety
- R 1 and R 2 are independently selected from methyl or ethyl moieties
- R 3 is a hydroxyl, hydroxymethyl or a hydroxyethyl moiety
- X is an anion which provides charge neutrality
- preferred anions include halides (such as chloride), sulphate and sulphonate.
- Preferred cationic surfactants are mono-Cg-io alkyl mono-hydroxyethyl di-methyl quaternary ammonium chloride, mono-Cio- 12 alkyl mono-hydroxyethyl di-methyl quaternary ammonium chloride and mono-Cio alkyl mono-hydroxyethyl di-methyl quaternary ammonium chloride.
- the cationic surfactant may provide additional greasy soil cleaning performance. However, the cationic surfactant may increase the tendency of the anionic surfactant to precipitate out of solution.
- the cationic surfactant and the anionic surfactant may be present in the composition in the form of separate particles. This minimises any effect that the cationic surfactant may have on the undesirable precipitation of the anionic surfactant, and also ensures that upon contact with water, the resultant wash liquor is not cloudy.
- the weight ratio of anionic surfactant to cationic surfactant may be in the range of from about 5:1 to about 25: 1, from about 5:1 to about 20:1, from about 6:1 to about 15:1, from about 7:1 to about 10:1, or from about 8:1 to about 9:1.
- composition may contain one or more detergent builders or builder systems.
- Builders include, for example, zeolite builder, phosphate builder.
- the composition contains from about Owt% to about 4wt%, from about Owt% to about 3wt%, or from about Owt% to about 2wt%.
- the composition may be free from deliberately added zeolite builder, i.e., may be substantially free from zeolite builder. This may be preferred if it is desirable for the composition to be very highly soluble. In addition, this is highly preferred if the composition, upon contact with water, is to form a transparent wash liquor.
- Zeolite builders include zeolite A, zeolite X, zeolite P and zeolite MAP.
- the composition contains from about Owt% to about 4wt%, from about Owt% to about 3wt%, from about Owt% to about 2wt%, or from about Owt% to about lwt% phosphate builder.
- the composition may be free from deliberately added phosphate builder, i.e., may be substantially free from phosphate builder. This may be preferred if it is desirable for the composition to have a very good environmental profile.
- Phosphate builders typically include sodium tripolyphosphate and sodium hydroxyethylidene Diphosphonate.
- composition herein may contain other builders in addition to the zeolite builder and/or phosphate builder as described above.
- water-soluble adjunct builders may be used.
- Adjunct builders may include sodium carbonate, citric acid and/or water-soluble salts thereof including sodium citrate; sulphamic acid and/or water-soluble salts thereof; polymeric polycarboxylates such as co-polymers of acrylic acid and maleic acid, or polyacrylate.
- the composition may contain very low levels of water-insoluble builders, such as zeolite A, zeolite X, zeolite P, and zeolite MAP, whilst containing relatively high levels of water-soluble adjunct builders, such as sodium carbonate, sulphamic acid and citric acid.
- the weight ratio of sodium carbonate to zeolite builder may be at least about 5:1, at least about 10:1, at least about 15:1, at least about 20:1 or at least about 25:1.
- the composition contains balance detergent ingredients, such as enzymes, silicate salts, polymeric carboxylates, solid dispersants, inorganic sulphate salts, bleaches, bleach activators, transition metal bleach catalysts, imine bleach catalysts, chelants, and other detergent ingredients.
- detergent ingredients such as enzymes, silicate salts, polymeric carboxylates, solid dispersants, inorganic sulphate salts, bleaches, bleach activators, transition metal bleach catalysts, imine bleach catalysts, chelants, and other detergent ingredients.
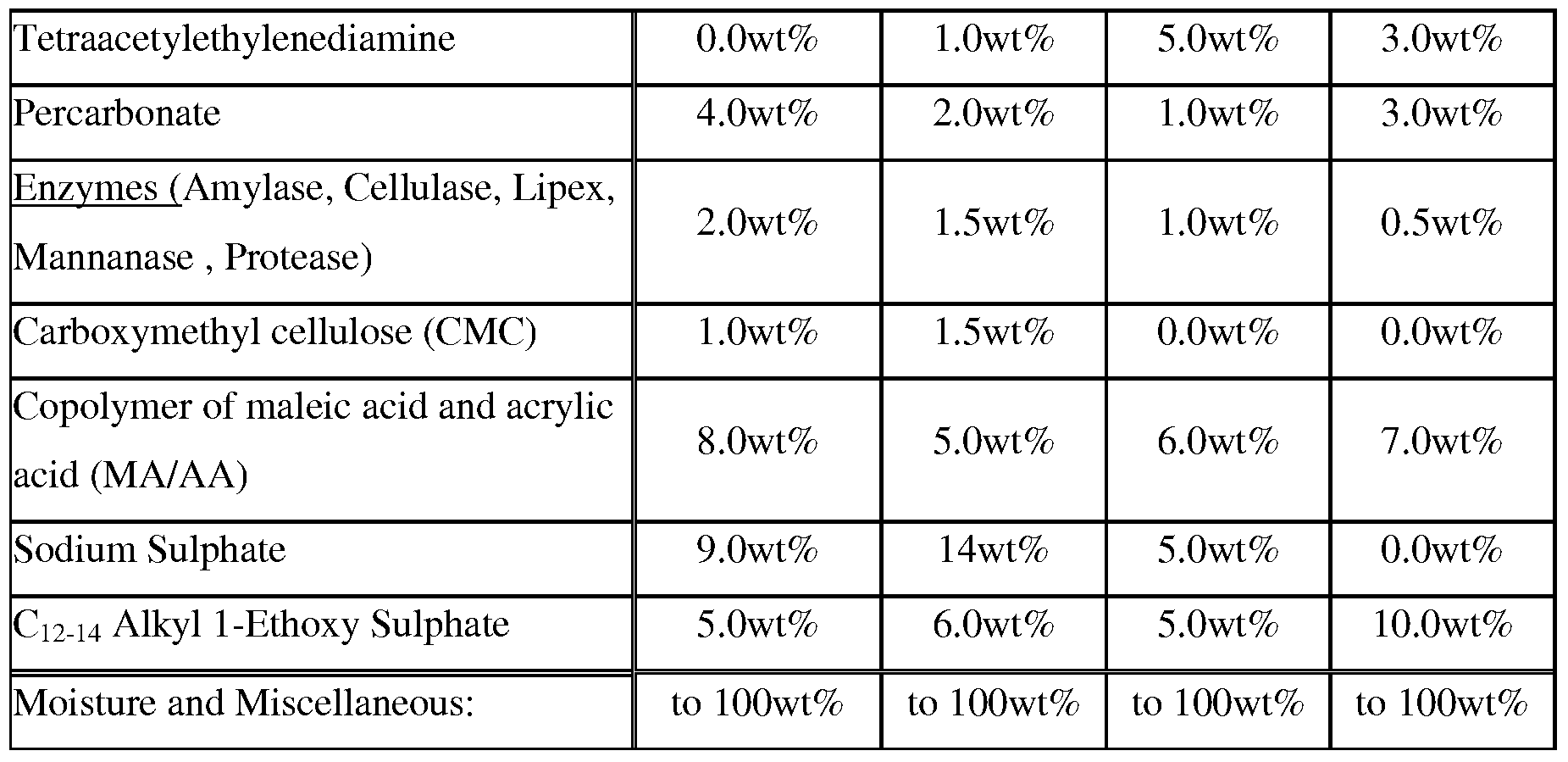
- composition herein contains enzymes, e.g., protease, amylase, lipase, cellulose, mannanase, pectate lyase, bleaching enzymes, and the like.
- enzymes e.g., protease, amylase, lipase, cellulose, mannanase, pectate lyase, bleaching enzymes, and the like.
- the composition herein may contain a protease, for instance, the composition contains at least about 11 mg, at least about 15 mg, at least about 20 mg, or at least about 30 mg of active protease per 100 g of composition.
- the protease may contain a calcium binding site.
- the protease may show improved stability and/or activity in the presence of high levels of free calcium cations present in the wash liquor.
- the cleaning performance of the composition is improved, and any reduction in the cleaning performance of the composition due to the low levels of, or lack of, zeolite builders and phosphate builders, which lead to a reduction in the anionic surfactant activity, is mitigated by the increased stability and/or activity of the protease.
- Preferred proteases include: subtilisins from Bacillus [e.g. subtilis, lentus, licheniformis, amyloliquefaciens (BPN, BPN'), alcalophilus] that are sold under the trade names Esperase® * Alcalase®, Everlase® and Savinase® supplied by Novozymes; proteases supplied by Genencor under the tradenames FN2®, FN3® and FN4®; and BLAP and/or variants thereof.
- Suitable proteases are described in more detail in EP 130 756, WO 91/06637, WO 95/10591 and WO 99/20726.
- the composition may also contain amylase, for example, in an amount of at least about 4 mg, at least about 6 mg, at least about 10 mg, at least about 15mg, at least about 20 mg, or about 30 mg of active amylase per 100 g of the total composition.
- the amylase may contain a calcium binding site. Analogous to the protease, the amylase may also show improved stability and/or activity, especially stability, in the presence of high levels of free calcium cations present in the wash liquor. Thus, incorporation of amylase into the composition may improve the cleaning performance.
- amylases include: amylases supplied by Novo Industries A/A under the tradenames Natalase®, Duramyl®, Termamyl®, Ban®, Fungamyl®; amylases supplied by Genencor under the tradename Purafect Ox Am®; and mixtures thereof.
- the amylase can be an ⁇ -amylase or a ⁇ -amylase. Suitable amylases are described in more detail in WO 94/02597 and WO 96/23873.
- the composition may also contain lipase, for example, in an amount of at least about 5 mg, at least about 7 mg, at least about 10 mg, at least about 15 mg, at least about 20 mg, or at least about 30 mg of active lipase per 100 g of composition.
- the lipase may contain a calcium binding site. Analogous to both the protease and the amylase, the lipase may also show improved stability and/or activity, especially activity, in the presence of high levels of free calcium cations present in the wash liquor.
- the incorporation of lipase into the composition improves the cleaning performance.
- Preferred lipases include those produced by Pseudomonas and Chromobacter groups. Preferred lipases are supplied by Novozymes under the tradenames. Lipolase®, Lipolase Ultra®, Lipoprime® and Lipex®. Other suitable lipases are cutinases and esterases.
- the composition may also contain other enzymes such as: cellulases, including bacterial or fungal cellulases such as cellulases produced by Humicola insolens, and in particular cellulases supplied by Novo Industries A/A under the tradenames Carezyme®, Endo A®, other suitable cellulases are the EGIII cellulases from Trichoderma longibrachiatum; carbohydrases, including mannanase such as that described in more detail in US 6,060,299, pectate lyase such as that described in more detail in WO 99/27083, cyclomaltodextringlucanotransferase such as that described in more detail in WO 96/33267, xyloglucanase such as that described in more detail in WO 99/02663; bleaching enzymes such as peroxidases, laccases, oxygenases (e.g. catechol 1,2 dioxygenase), lipoxygenase that is described in more detail in
- the composition may also contain other enzymes such as: cellulases, including bacterial or fungal cellulases such as cellulases produced by Humicola insolens, and in particular cellulases supplied by Novo Industries A/A under the tradenames Carezyme®, Endo A®, other suitable cellulases are the EGIII cellulases from Trichoderma longibrachiatum; carbohydrases, including mannanase such as that described in more detail in US 6,060,299, pectate lyase such as that described in more detail in WO 99/27083, cyclomaltodextringlucanotransferase such as that described in more detail in WO 96/33267, xyloglucanase such as that described in more detail in WO 99/02663; bleaching enzymes such as peroxidases, laccases, oxygenases (e.g. catechol 1,2 dioxygenase), lipoxygenase that is described in more detail in
- the composition herein may further contain silicate salt.
- the detergent composition may contain less than about 10wt%, from about Owt% to about 5wt%, or less than about 4wt%, or less than about 2wt% silicate salt. It may even be preferred for the detergent about composition to be free from silicate salt.
- Silicate salts include water-insoluble silicates.
- Silicate salts include amorphous silicates and crystalline layered silicates (e.g. SKS-6).
- a preferred silicate salt is sodium silicate.
- the composition may contain at least about lwt%, at least about 2wt%, at least about 3wt%, at least about 4wt%, or at least 5wt% polymeric carboxylates.
- Polymeric carboxylates include: polyacrylates, preferably having a weight average molecular weight of from about 1,000 Da to about 20,000 Da; co-polymers of maleic acid and acrylic acid, preferably having a molar ratio of maleic acid monomers to acrylic acid monomers of from about 1:1 to about 1: 10 and a weight average molecular weight of from about 10,000 Da to about 200,000 Da, or preferably having a molar ratio of maleic acid monomers to acrylic acid monomers of from about 0.3: 1 to about 3:1 and a weight average molecular weight of from about 1,000Da to about 50,000Da.
- a soil dispersant having the formula: bis((C 2 H 5 ⁇ )(C 2 H 4 ⁇ )n)(CH 3 )-N + -C x H 2 ⁇ -N + -(CH3)-
- the composition contains at least about lwt%, or at least about 2wt%, or at least about 3wt% soil dispersants, typically having the above described formulae.
- the composition herein contains from about Owt% to about 10wt%, from about Owt% to about 8wt%, from about Owt% to about 6wt%, or from about Owt% to about lwt% inorganic inorganic sulphate salt.
- the composition may be free from deliberately added inorganic sulphate salt, i.e., may be substantially free from inorganic sulphate salt.
- Sulphate inorganic sulphate salts include sodium sulphate, magnesium sulphate, potassium sulphate, ammonium hydrogen sulphate, ammonium sulphate, and the like.
- the inorganic sulphate salt is sodium sulphate.
- the detergent composition contains a peroxide bleach.
- the peroxide bleach may contain percarbonate and/or perborate, typically from about above Owt%, or from lwt% to about 7wt%, or 5wt%, or to 4wt%, or to 3wt% in total composition.
- the percarbonate may be sodium percarbonate and the perborate may be sodium perborate.
- the composition contains a bleach activator.
- Suitable bleach activators are compounds which when used in conjunction with a hydrogen peroxide source leads to the in situ production of the peracid corresponding to the bleach activator.
- Various non limiting examples of bleach activators are disclosed in U.S. Patent 4,915,854, issued April 10, 1990 to Mao et al, and U.S. Patent 4,412,934.
- NOBS nonanoyloxybenzene sulfonate
- TAED tetraacetylethylenediamine
- Another suitable bleach activator is decanoyloxybenzenecarboxylic acid (DOBA).
- a leaving group is any group that is displaced from the bleach activator as a consequence of the nucleophilic attack on the bleach activator by the hydroperoxide anion.
- a preferred leaving group is oxybenzenesulfonate.
- bleach activators of the above formulae include (6-octanamido- caproyl)oxybenzenesulfonate, (6-nonanamidocaproyl)oxybenzenesulfonate, (6-decanamido- caproyl)oxybenzenesulfonate, and mixtures thereof as described in U.S. Patent 4,634,551, incorporated herein by reference.
- Another class of bleach activators contains the benzoxazin-type activators disclosed by Hodge et al in U.S. Patent 4,966,723, issued October 30, 1990, incorporated herein by reference.
- a highly preferred activator of the benzoxazin-type is:
- Still another class of preferred bleach activators includes the acyl lactam activators, especially acyl caprolactams and acyl valerolactams of the formulae:
- lactam activators include benzoyl caprolactam, octanoyl caprolactam, 3,5,5-trimethylhexanoyl caprolactam, nonanoyl caprolactam, decanoyl caprolactam, undecenoyl caprolactam, benzoyl valerolactam, octanoyl valerolactam, decanoyl valerolactam, undecenoyl valerolactam, nonanoyl valerolactam, 3,5,5- trimethylhexanoyl valerolactam and mixtures thereof. See also U.S. Patent 4,545,784, issued to Sanderson, October 8, 1985, incorporated herein by reference, which discloses acyl caprolactams, including benzoyl caprolactam, adsor
- Highly preferred bleach activators are nonanoyloxybenzene sulfonate (NOBS) and/or tetraacetylethylenediamine (TAED) .
- NOBS nonanoyloxybenzene sulfonate
- TAED tetraacetylethylenediamine
- the weight ratio of bleach activator to source of hydrogen peroxide present in the laundry detergent composition is at least 0.5:1, at least 0.6:1, at least 0.7:1, 0.8:1, preferably at least 0.9: 1, or 1.0:1.0, or even 1.2: 1 or higher.
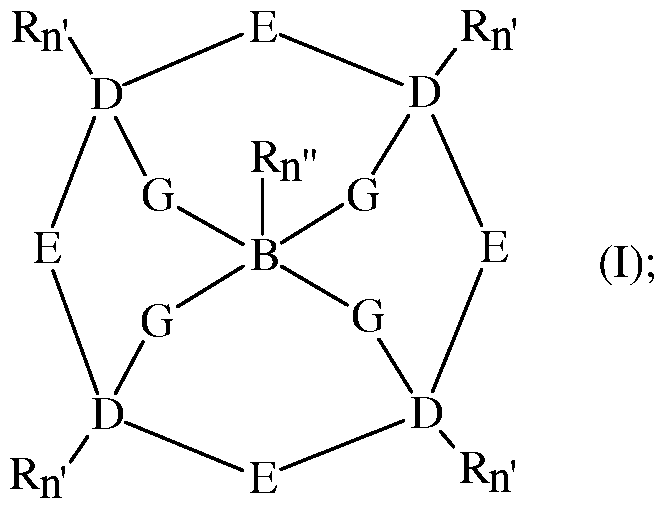
- the composition may contain a transition metal bleach catalyst.
- the transition metal bleach catalyst typically contains a transition metal ion, preferably selected from transition metal selected from the group consisting of Mn(II), Mn(III), Mn(IV), Mn(V), Fe(II), Fe(III), Fe(IV), Co(I), Co(II), Co(III), Ni(I), Ni(II), Ni(III), Cu(I), Cu(II), Cu(III), Cr(II), Cr(III), Cr(IV), Cr(V), Cr(VI), V(III), V(IV), V(V), Mo(IV), Mo(V), Mo(VI), W(IV), W(V), W(VI), Pd(II), Ru(II), Ru(III), and Ru(IV), more preferably Mn(II), Mn(III), Mn(IV), Fe(II), Fe(III), Cr(II), Cr(III), Cr(IV), Cr(V), and Cr(VI).
- the transition metal bleach catalyst typically contains a ligand, preferably a macropolycyclic ligand, more preferably a cross-bridged macropolycyclic ligand.
- the transition metal ion is preferably coordinated with the ligand.
- the ligand contains at least four donor atoms, at least two of which are bridgehead donor atoms.
- the cross-bridged macropolycyclic ligand is coordinated by four or five donor atoms to the same transition metal and contains:
- an organic macrocycle ring containing four or more donor atoms selected from N and optionally O and S, at least two of these donor atoms being N (preferably at least 3, more preferably at least 4, of these donor atoms are N), separated from each other by covalent linkages of 2 or 3 non-donor atoms, two to five (preferably three to four, more preferably four) of these donor atoms being coordinated to the same transition metal in the complex;
- a cross-bridging chain which covalently connects at least 2 non-adjacent N donor atoms of the organic macrocycle ring, said covalently connected non-adjacent N donor atoms being bridgehead N donor atoms which are coordinated to the same transition metal in the complex, and wherein said cross-bridged chain contains from 2 to about 10 atoms (preferably the cross-bridged chain is selected from 2, 3 or 4 non-donor atoms, and 4-6 non-donor atoms with a further, preferably N, donor atom); and
- non-macropolycyclic ligands preferably selected from the group consisting of H 2 O, ROH, NR 3 , RCN, OH “ , OOH “ , RS “ , RO “ , RCOO “ , OCN “ , SCN “ , N 3 “ , CN “ , F “ , Cl “ , Br “ , I “ , O 2 “ , NO 3 “ , NO 2 “ , SO 4 2” , SO 3 2” , PO 4 3” , organic phosphates, organic phosphonates, organic sulfates, organic sulfonates, and aromatic N donors such as pyridines, pyrazines, pyrazoles, imidazoles, benzimidazoles, pyrimidines, triazoles and thiazoles with R being H, optionally substituted alkyl, optionally substituted aryl.
- a suitable transition metal bleach catalyst contains a complex of a transition metal and a macropolycyclic rigid ligand, preferably a cross-bridged macropolycyclic ligand, wherein:
- said transition metal is selected from the group consisting of Mn(II), Mn(III), Mn(IV), Mn(V), Fe(II), Fe(III), Fe(IV), Co(I), Co(II), Co(III), Ni(I), Ni(II), Ni(III), Cu(I), Cu(II), Cu(III), Cr(II), Cr(III), Cr(IV), Cr(V), Cr(VI), V(III), V(IV), V(V), Mo(IV), Mo(V), Mo(VI), W(IV), W(V), W(VI), Pd(II), Ru(II), Ru(III), and Ru(IV);
- said macropolycyclic rigid ligand is coordinated by at least four, preferably four or five, donor atoms to the same transition metal and contains:
- an organic macrocycle ring containing four or more donor atoms preferably at least 3, more preferably at least 4, of these donor atoms are N
- donor atoms preferably at least 3, more preferably at least 4, of these donor atoms are N
- covalent linkages of at least one, preferably 2 or 3, non-donor atoms, two to five (preferably three to four, more preferably four) of these donor atoms being coordinated to the same transition metal in the complex
- linking moiety preferably a cross-bridging chain, which covalently connects at least 2 (preferably non-adjacent) donor atoms of the organic macrocycle ring, said covalently connected (preferably non-adjacent) donor atoms being bridgehead donor atoms which are coordinated to the same transition metal in the complex, and wherein said linking moiety (preferably a cross-bridged chain) contains from 2 to about 10 atoms (preferably the cross- bridged chain is selected from 2, 3 or 4 non-donor atoms, and 4-6 non-donor atoms with a further donor atom), including for example, a cross-bridge which is the result of a Mannich condensation of ammonia and formaldehyde; and
- non-macropolycyclic ligands preferably monodentate ligands, such as those selected from the group consisting of H 2 O, ROH, NR 3 , RCN, OH “ , OOH “ , RS “ , RO “ , RCOO “ , OCN “ , SCN “ , N 3 “ , CN “ , F “ , Cl “ , Br “ , I “ , O 2 “ , NO 3 “ , NO 2 “ , SO 4 2” , SO 3 2” , PO 4 3” , organic phosphates, organic phosphonates, organic sulfates, organic sulfonates, and aromatic N donors such as pyridines, pyrazines, pyrazoles, imidazoles, benzimidazoles, pyrimidines, triazoles and thiazoles with R being H, optionally substituted alkyl, optionally substituted aryl (specific examples of monodentate ligands, such as those selected from the
- Suitable cross-bridged macropolycyclic ligands include:
- each "E” is the moiety (CR n ) a -X-(CR n ) a ', wherein -X- is selected from the group consisting of O, S, NR and P, or a covalent bond, and preferably X is a covalent bond and for each E the sum of a + a' is independently selected from 1 to 5, more preferably 2 and 3;
- each "R” is independently selected from H, alkyl, alkenyl, alkynyl, aryl, alkylaryl (e.g., benzyl), and heteroaryl, or two or more R are covalently bonded to form an aromatic, heteroaromatic, cycloalkyl, or heterocycloalkyl ring;
- each "D” is a donor atom independently selected from the group consisting of N, O, S, and P, and at least two D atoms are bridgehead donor atoms coordinated to the transition metal (in the preferred embodiments, all donor atoms designated D are donor atoms which coordinate to the transition metal, in contrast with heteroatoms in the structure which are not in D such as those which may be present in E; the non-D heteroatoms can be non-coordinating and indeed are non-coordinating whenever present in the preferred embodiment);
- B is a carbon atom or "D” donor atom, or a cycloalkyl or heterocyclic ring;
- n is an integer independently selected from 1 and 2, completing the valence of the carbon atoms to which the R moieties are covalently bonded;
- each "n"' is an integer independently selected from 0 and 1, completing the valence of the D donor atoms to which the R moieties are covalently bonded;
- each "n”" is an integer independently selected from 0, 1, and 2 completing the valence of the B atoms to which the R moieties are covalently bonded;
- each "a” and “a'” is an integer independently selected from 0-5, preferably a + a' equals 2 or 3, wherein the sum of all "a” plus “a”' in the ligand of formula (I) is within the range of from about 6 (preferably 8) to about 12, the sum of all "a” plus “a”' in the ligand of formula (II) is within the range of from about 8 (preferably 10) to about 15, and the sum of all "a” plus "a”' in the ligand of formula (III) is within the range of from about 10 (preferably 12) to about 18;
- a suitable cross-bridged macropolycyclic ligand is selected from the group consisting of:
- each "R” is independently selected from H, alkyl, alkenyl, alkynyl, aryl, alkylaryl (e.g., benzyl) and heteroaryl, or two or more R are covalently bonded to form an aromatic, heteroaromatic, cycloalkyl, or heterocycloalkyl ring;
- n is an integer independently selected from 0, 1 and 2, completing the valence of the carbon atoms to which the R moieties are covalently bonded;
- each "b” is an integer independently selected from 2 and 3;
- each "a” is an integer independently selected from 2 and 3.
- Suitable transition metal bleach catalysts include: Dichloro-5,12-dimethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-4,10-dimethyl-l,4,7,10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Diaquo-5,12-dimethyl- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Manganese(II) Hexafluorophosphate; Aquo-hydroxy-5,12- dimethyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(III) Hexafluorophosphate; Diaquo- 4, 10-dimethyl- 1 ,4,7, 10-tetraazabicyclo[5.5.2]tetradecane Manganese(II) Hexafluorophosphate; Diaquo-5,12-dimethyl-l
- Trifluoromethanesulfonate Chloro-5,12,17-trimethyl-l,5,8,12,17- pentaazabicyclo[6.6.5]nonadecane Manganese(II) Hexafluorophosphate; Chloro-4,10,15-trimethyl- 1,4,7, 10,15-pentaazabicyclo[5.5.5]heptadecane Manganese(II) Hexafluorophosphate; Chloro- 5,12,17-trimethyl-l,5,8,12,17-pentaazabicyclo[6.6.5]nonadecane Manganese(II) Chloride; Chloro- 4,10,15-trimethyl-l,4,7,10,15-pentaazabicyclo[5.5.5]heptadecane Manganese(II) Chloride; and any mixture thereof.
- transition metal bleach catalysts are described in U.S. 5,580,485, U.S. 4,430,243; U.S. 4,728,455; U.S. 5,246,621; U.S. 5,244,594; U.S. 5,284,944; U.S. 5,194,416; U.S. 5,246,612; U.S. 5,256,779; U.S. 5,280,117; U.S. 5,274,147; U.S. 5,153,161; U.S. 5,227,084; U.S. 5,114,606; U.S. 5,114,611, EP 549,271 Al; EP 544,490 Al; EP 549,272 Al; and EP 544,440 A2.
- a suitable transition metal bleach catalyst is a manganese-based catalyst, for example disclosed in U.S. 5,576,282.
- Suitable cobalt bleach catalysts are described, for example, in U.S. 5,597,936 andU.S. 5,595,967. Such cobalt catalysts are readily prepared by known procedures, such as taught for example in U.S. 5,597,936, and U.S. 5,595,967.
- a suitable transition metal bleach catalyst is a transition metal complex of ligand such as bispidones described in WO 05/042532 Al. Transition metal bleach catalysts provide robust cleaning profiles, especially under dilute wash conditions, and especially against beta-carotene, squalene and unsaturated triglyceride soils.
- the composition may contain an imine bleach catalyst.
- Suitable imine bleach catalysts include, but are not limited to: iminium cations and polyions; iminium zwitterions; N-sulphonyl imines; N-phosphonyl imines; N-acyl imines; perfluoroimines; and mixtures thereof.
- Suitable iminium cations and polyions include, but are not limited to, N-methyl-3,4- dihydroisoquinolinium tetrafluoroborate, prepared as described in Tetrahedron (1992), 49(2), 423-38 (see, for example, compound 4, p. 433); N-methyl-3,4-dihydroisoquinolinium p-toluene sulphonate, prepared as described in U.S. Pat. 5,360,569 (see, for example, Column 11, Example 1); and N-octyl-3,4-dihydroisoquinolinium p-toluene sulphonate, prepared as described in U.S. Pat. 5,360,568 (see, for example, Column 10, Example 3).
- Suitable iminium zwitterions include, but are not limited to, N-(3-sulfopropyl)-3,4- dihydroisoquinolinium, inner salt, prepared as described in U.S. Pat. 5,576,282 (see, for example, Column 31, Example II); N-[2-(sulphooxy)dodecyl]-3,4-dihydroisoquinolinium, inner salt, prepared as described in U.S. Pat.
- Suitable N-sulphonyl imine oxygen transfer catalysts include, but are not limited to, 3- methyl-l,2-benzisothiazole 1,1 -dioxide, prepared according to the procedure described in the Journal of Organic Chemistry (1990), 55(4), 1254-61.
- Suitable N-phosphonyl imine oxygen transfer catalysts include, but are not limited to, [R- (E)]-N-[(2-chloro-5-nitrophenyl)methylene]-P-phenyl-P-(2,4,6-trimethylphenyl)- phosphinic amide, which can be made according to the procedures described in the Journal of the Chemical Society, Chemical Communications (1994), (22), 2569-70.
- Suitable N-acyl imine oxygen transfer catalysts include, but are not limited to, [N(E)J-N- (phenylmethylene)acetamide, which can be made according to the procedures described in Polish Journal of Chemistry (2003), 77(5), 577-590.
- Suitable perfluoroimine oxygen transfer catalysts include, but are not limited to, (Z)- 2,2,3,3,4,4,4-heptafluoro-N-(nonafluorobutyl)butanimidoyl fluoride, which can be made according to the procedures described in Tetrahedron Letters (1994), 35(34), 6329-30.
- Suitable cyclic sugar ketone oxygen transfer catalysts include, but are not limited to, l,2:4,5-di-O-isopropylidene-D-erythro-2,3-hexodiuro-2,6-pyranose as prepared in U.S. Pat. 6,649,085 (Column 12, Example 1).
- the imine bleach catalyst contains an iminium and/or carbonyl functional group and is typically capable of forming an oxaziridinium and/or dioxirane functional group upon acceptance of an oxygen atom, especially upon acceptance of an oxygen atom from a peroxyacid and/or salt thereof.
- the imine bleach catalyst contains an oxaziridinium functional group and/or is capable of forming an oxaziridinium functional group upon acceptance of an oxygen atom, especially upon acceptance of an oxygen atom from a peroxyacid and/or salt thereof.
- the imine bleach catalyst contains a cyclic iminium functional group, preferably wherein the cyclic moiety has a ring size of from five to eight atoms (including the nitrogen atom), preferably six atoms.
- the imine bleach catalyst contains an aryliminium functional group, preferably a bi-cyclic aryliminium functional group, preferably a 3,4-dihydroisoquinolinium functional group.
- the imine functional group is a quaternary imine functional group and is typically capable of forming a quaternary oxaziridinium functional group upon acceptance of an oxygen atom, especially upon acceptance of an oxygen atom from a peroxyacid and/or salt thereof.
- the imine bleach catalyst has a chemical structure corresponding to the following chemical formula
- n and m are independently from 0 to 4, preferably n and m are both 0; each R is independently selected from a substituted or unsubstituted radical selected from the group consisting of hydrogen, alkyl, cycloalkyl, aryl, fused aryl, heterocyclic ring, fused heterocyclic ring, nitro, halo, cyano, sulphonato, alkoxy, keto, carboxylic, and carboalkoxy radicals; and any two vicinal R 1 substituents may combine to form a fused aryl, fused carbocyclic or fused heterocyclic ring; each R 2 is independently selected from a substituted or unsubstituted radical independently selected from the group consisting of hydrogen, hydroxy, alkyl, cycloalkyl, alkaryl, aryl, aralkyl, alkylenes, heterocyclic ring, alkoxys, arylcarbonyl groups, carboxyalkyl groups and amide groups
- each Y is independently selected from the group consisting of O, S,
- each R is independently selected from the group consisting of alkyl, aryl and heteroaryl, said moieties being substituted or unsubstituted, and whether substituted or unsubsituted said moieties having less than 21 carbons;
- each G is independently selected from the group consisting of CO, SO 2 , SO, PO and PO 2 ;
- R 9 and R 10 are independently selected from the group consisting of H and Ci-C 4 alkyl;
- R 11 and R 12 are independently selected from the group consisting of H and alkyl, or when taken together may join to form a carbonyl;
- b O or 1;
- y is an integer from 1 to 6;
- k is an integer from O to 20;
- R 6 is H, or an alkyl, aryl or heteroaryl moiety; said moieties being substituted or unsubstituted; and
- X if present, is a suitable
- the imine bleach catalyst has a structure corresponding to general formula below:
- R » 13 is a branched alkyl group containing from three to 24 carbon atoms (including the branching carbon atoms) or a linear alkyl group containing from one to 24 carbon atoms; preferably R 13 is a branched alkyl group containing from eight to 18 carbon atoms or linear alkyl group containing from eight to eighteen carbon atoms; preferably R 13 is selected from the group consisting of 2-ethylhexyl, 2-propylheptyl, 2-butyloctyl, 2-pentylnonyl, 2-hexyldecyl, n- dodecyl, n-tetradecyl, n-hexadecyl, n-octadecyl, iso-nonyl, iso-decyl, iso-tridecyl and iso- pentadecyl; preferably R 13 is selected from the group consisting of 2-butyloctyl, 2-
- the imine bleach catalyst has a structure corresponding to general formula below or mixtures thereof.
- G is selected from -O-, -CH 2 O-, -(CH 2 ) 2 -, and -CH 2 -.
- R 1 is selected from H or Ci-C 4 alkyl. Suitable Ci-C 4 alkyl moieties include, but are not limited to methyl, ethyl, iso-propyl, and tert-butyl.
- Each R 2 is independently selected from C 4 -Cs alkyl, benzyl, 2-methylbenzyl, 3- methylbenzyl, 4-methylbenzyl, 4-ethylbenzyl, 4-iso-propylbenzyl and 4-tert-butylbenzyl.
- Suitable C 4 -Cs alkyl moieties include, but are not limited to n-butyl, n-pentyl, cyclopentyl, n- hexyl, cyclohexyl, cyclohexylmethyl, n-heptyl and octyl.
- G is selected from -O- and -CH 2 -.
- R 1 is selected from H, methyl, ethyl, iso-propyl, and tert-butyl.
- Each R 2 is independently selected from C 4 -C 6 alkyl, benzyl, 2-methylbenzyl, 3-methylbenzyl, and 4-methylbenzyl.
- G is -CH 2 -, R 1 is H and each R 2 is independently selected from n-butyl, n-pentyl, n-hexyl, benzyl, 2-methylbenzyl, 3-methylbenzyl, and 4- methylbenzyl.
- the composition may contain a chelant.
- Suitable chelants include diethylene triamine pentaacetate, diethylene triamine penta(methyl phosphonic acid), ethylene diamine-N'N'- disuccinic acid, ethylene diamine tetraacetate, ethylene diamine tetra(methylene phosphonic acid) and hydroxyethane di(methylene phosphonic acid).
- a preferred chelant is ethylene diamine-N'N'-disuccinic acid (EDDS) and/or hydroxyethane diphosphonic acid (HEDP).
- EDDS ethylene diamine-N'N'-disuccinic acid
- HEDP hydroxyethane diphosphonic acid
- the ethylene diamine-N'N'-disuccinic acid is in S'S' enantiomeric form.
- the composition may contain a detergent ingredient selected from: suds suppressing systems such as silicone based suds suppressors; brighteners; photobleach; filler salts; fabric- softening agents such as clay, silicone and/or quaternary ammonium compounds; flocculants such as polyethylene oxide; dye transfer inhibitors such as polyvinylpyrrolidone, poly 4- vinylpyridine N-oxide and/or co-polymer of vinylpyrrolidone and vinylimidazole; fabric integrity components such as hydrophobically modified cellulose and oligomers produced by the condensation of imidazole and epichlorhydrin; soil dispersants and soil anti-redeposition aids such as polycarboxylates, alkoxylated polyamines and ethoxylated ethyleneimine polymers; and anti-redeposition components such as carboxymethyl cellulose and polyesters.
- suds suppressing systems such as silicone based suds suppressors; brighteners; photobleach; filler salts; fabric- softening agents such as
Abstract
A laundry detergent composition containing (i) from about 1wt% to about 60wt% detersive surfactant; (ii) from about 0wt% to about 4wt% zeolite builder; (iii) from about 0wt% to about 4wt% phosphate builder, (iv) from about above 0wt% to about 7wt% peroxide bleach selected from the group consisting of percarbonate and perborate and (v) balance detergent ingredients. The low level of bleach allows good bleaching (e.g., the use of a catalyst), while it maintains good fabric integrity profiles.
Description
LAUNDRY DETERGENT COMPOSITION COMPRISING
LOW LEVEL OF BLEACH
FIELD OF THE INVENTION
The present invention relates to laundry detergent compositions comprising a detersive surfactant and a peroxide bleach.
BACKGROUND OF THE INVENTION
Granular laundry detergent compositions need to have a good fabric-cleaning performance, good dispensing and dissolution profiles. Some laundry detergent compositions contain bleaches. Some bleaches are too active and sometimes they may damage the fabric during the wash cycle and/or after storage in a certain environment. Among bleaches, peroxide bleaches are widely used. Peroxide bleaches, e.g., sodium percarbonate and sodium perborate, are beneficial because they can be applied to various kinds of fabrics. However, using a large amount of peroxide bleach may leave risk of runaway thermal degradation in product, for instance, in hot regions.
Granular laundry detergent compositions typically contain builders, such as zeolite and phosphate. Zeolite builders are water-insoluble and their incorporation in laundry detergent compositions leads to poor dissolution of the laundry detergent composition and can also lead to undesirable residues being deposited on the fabric. In addition, detergent compositions that contain high levels of zeolite builder form undesirable cloudy wash liquors upon contact with water. Further, phosphate builders allegedly do not have favourable environmental profiles and their use in laundry detergent compositions is becoming less common, for example, due to phosphate legislation in many countries.
If zeolite and phosphate builders are reduced or removed from a laundry detersive composition, percarbonate is a source of carbonate anions in the wash liquor, and these carbonate anions can complex with free calcium cations to form calcium carbonate which is insoluble in water. Such calcium carbonate may precipitate on the fabric surface, and can deteriorate the appearance of the fabric.
In addition, high levels of percarbonate in low builder laundry detergent compositions can have very poor bleach stability profile, due to the lack of an adequate water-sink.
In addition, there is a need to improve the fabric care profile of the laundry detergent composition so that a catalytic bleach, such as a transition metal bleach catalyst and/or an imine bleach catalyst can be incorporated therein without jeopardising the fabric care profile of the product.
The inventors have overcome these problems by formulating a laundry detergent composition as defined by the claims.
SUMMARY OF THE INVENTION
The present invention relates to a laundry detergent composition containing (i) from about lwt% to about 60wt% detersive surfactant; (ii) from about Owt% to about 4wt% zeolite builder; (iii) from about Owt% to about 4wt% phosphate builder; (iv) from about above Owt% to about 7wt% peroxide bleach selected from the group consisting of percarbonate and perborate and (v) balance detergent ingredients. The low level of bleach allows good bleaching, improves the stability of the product, and enables the use of a bleach catalyst whilst maintaining good fabric integrity profiles.
These and other objects, advantages and features of the invention will be more fully understood and appreciated by reference to the written specification.
DETAILED DESCRIPTION OF THE INVENTION LAUNDRY DETERGENT COMPOSITION
The present invention relates to a solid laundry detersive composition. The laundry detergent composition contains a solid laundry detersive composition containing: (i) from about lwt% to about 60wt% detersive surfactant; (ii) from about Owt% to about 4wt% zeolite builder; (iii) from about Owt% to about 4wt% phosphate builder; (iv) from about above Owt% to about 7wt% of a peroxide bleach selected from the group consisting of percarbonate and perborate, and (v) balance additional detergent ingredients.
The laundry detersive composition is suitable for use in the laundering of fabrics. The detergent composition contains a low level of a peroxide bleach.
Accordingly, it may be preferred if the composition contains low levels of peroxide bleaches. The composition may contain from about above Owt% to about 7wt%, from about lwt% to about 6wt% or from about lwt% to about 5wt%, or to about 4wt%, or even to about 3wt% peroxide bleach.
The detergent composition can be in the form of a solid; such as in form of free-flowing particles or in the form of a tablet. The detergent composition may be in the form of free- flowing particles such as agglomerates, extrudates, spray-dried particles, noodles, needles, flakes and combinations thereof. The composition may be a granular laundry detergent composition. The detergent composition in free-flowing particulate form typically has a bulk density of from about 450 g/1 to about 1,000 g/1, preferred low bulk density detergent compositions have a bulk density of from about 550 g/1 to about 650 g/1 and preferred high bulk density detergent compositions have a bulk density of from about 750 g/1 to about 900 g/1. During the laundering process, the composition is typically contacted with water to give a wash liquor having a pH of from about 7 to about 11, or from about 8 to about 10.5.
The composition may be made by any suitable method including agglomeration, spray- drying, extrusion, mixing, dry-mixing, liquid spray-on, roller compaction, spheronisation or any combination thereof.
The composition herein may contain the following ingredients.
SURFACTANT
The composition herein contains from about lwt% to about 60wt%, from about 2wt% to about 55wt%, from 3wt% to about 50wt%, or from about 4wt% to about 40wt% detersive surfactant. The detersive surfactant may contain an anionic surfactant, a nonionic surfactant, a cationic surfactant, a zwitterionic surfactant, or any mixture thereof. The surfactant may be amphoteric, and have a ionic charge dependant on the pH of a typical wash liquor.
ANIONIC SURFACTANT
The composition may contain an anionic surfactant. Preferably the composition may contain from about lwt% to about 40wt%, from about 2wt% to about 30wt%, or from about 5wt% to about 20wt% anionic surfactant. The anionic surfactant can be selected from the group consisting of: CiO-Ci8 alkyl benzene sulphonates (LAS), preferably linear C10-C13 alkyl benzene sulphonate; C10-C20 primary, branched-chain, linear-chain and random-chain alkyl sulphates (AS), preferred are linear alkyl sulphates, typically having the following formula:
CH3(CH2)XCH2-OSO3 " M+,
wherein, M is hydrogen or a cation which provides charge neutrality, preferred cations include sodium and ammonium cations, wherein x is an integer of at least 7, preferably at least 9, highly preferred are linear or branched, substituted or unsubstituted Ci2-Ci8 alkyl sulphate; CI0-CI8 secondary (2,3) alkyl sulphates, typically having the following formulae:
OSO3 " M+ OSO3 " M+
I I
CH3(CH2)X(CH)CH3 or CH3(CH2)y (CH)CH2CH3 wherein, M is hydrogen or a cation which provides charge neutrality, preferred cations include sodium and ammonium cations, wherein x is an integer of at least 7, preferably at least 9, y is an integer of at least 8, preferably at least 9; Ci0-Ci8 alkyl alkoxy carboxylates; mid-chain branched alkyl sulphates as described in more detail in US 6,020,303 and US 6,060,443; modified alkylbenzene sulphonate (MLAS) as described in more detail in WO 99/05243, WO 99/05242, WO 99/05244, WO 99/05082, WO 99/05084, WO 99/05241, WO 99/07656, WO 00/23549, and WO 00/23548; methyl ester sulphonate (MES); alpha-olefin sulphonate (AOS) and mixtures thereof. Preferred anionic surfactants are: linear or branched, substituted or unsubstituted, Ci2-I8 alkyl sulphates; and linear or branched, substituted or unsubstituted, CiO_i3 alkylbenzene sulphonates, preferably linear Ci0-O alkylbenzene sulphonates; and mixtures thereof.
Another preferred anionic surfactant is a Cg-24 alkylalkoxylated sulphate, preferably an Cδ-24 alkylethoxylated sulphate, preferably having an average degree of ethoxylation of 1 to 5.
The anionic surfactant may be structurally modified in such a manner as to cause the anionic surfactant to be more calcium tolerant and less likely to precipitate out of the wash liquor in the presence of free calcium ions. This structural modification could be the introduction of a methyl or ethyl moiety in the vicinity of the anionic surfactant's head group, as this can lead to a more calcium tolerant anionic surfactant due to steric hindrance of the head group, which may reduce the anionic surfactant's affinity for complexing with free calcium cations in such a manner as to cause precipitation out of solution. Other structural modifications include the introduction of functional moieties, such as an amine moiety, in the alkyl chain of the anionic surfactant; this can lead to a more calcium tolerant anionic surfactant because the presence of a functional group in the alkyl chain of an anionic surfactant may minimise the undesirable physicochemical property of the anionic surfactant to form a smooth crystal
structure in the presence of free calcium ions in the wash liquor. This may reduce the tendency of the anionic surfactant to precipitate out of solution.
The anionic surfactant may be in particulate form, such as an agglomerate, a spray-dried powder, an extrudate, a bead, a noodle, a needle or a flake. The anionic surfactant, or at least part thereof, may be in a co-particulate admixture with a non-ionic surfactant, this co-particulate admixture may be in spray-dried form. The anionic surfactant, or at least part thereof, may be in agglomerate form; the agglomerate may contain at least about 20wt%, of the agglomerate, of an anionic surfactant, or from about 20wt% to about 65wt%, of the agglomerate, of an anionic surfactant. Part of the anionic surfactant may be in the form of a spray-dried powder (e.g. a blown powder), and part of the anionic surfactant may be in the form of a non-spray-dried powder (e.g. an agglomerate, or an extrudate, or a flake). A linear alkylbenzene sulphonate may be in a co-particulate admixture with soap; this co-particulate admixture may be in spray-dried form.
NON-IONIC SURFACTANT
The composition herein may contain non-ionic surfactant. The composition herein may contain from about 0.1wt% to about 15wt%, from about 0.5wt% to about 10wt%, or from about lwt% to about 6wt% non- ionic surfactant.
The non-ionic surfactant can be selected from the group consisting of: Ci2-Ci8 alkyl ethoxylates, such as, NEODOL® non-ionic surfactants from Shell; CO-CI2 alkyl phenol alkoxylates wherein the alkoxylate units are ethyleneoxy units, or propyleneoxy units, or a mixture thereof; Ci2-Ci8 alcohol and CO-CI2 alkyl phenol condensates with ethylene oxide/propylene oxide block polymers such as Pluronic from BASF; C14-C22 mid-chain branched alcohols, BA, as described in more detail in US 6,150,322; C14-C22 mid-chain branched alkyl alkoxylates, BAEx, wherein x = from 1 to 30, as described in more detail in US 6,153,577, US 6,020,303 and US 6,093,856; alkylpoly saccharides as described in more detail in US 4,565,647, specifically alkylpolyglycosides as described in more detail in US 4,483,780 and US 4,483,779; polyhydroxy fatty acid amides as described in more detail in US 5,332,528, WO 92/06162, WO 93/19146, WO 93/19038, and WO 94/09099; ether capped poly(oxyalkylated) alcohol surfactants as described in more detail in US 6,482,994 and WO 01/42408; and mixtures thereof.
The non-ionic surfactant could be an alkyl polyglucoside and/or an alkyl alkoxylated alcohol. The non-ionic surfactant may be a linear or branched, substituted or unsubstituted C8-I8 alkyl ethoxylated alcohol having an average degree of ethoxylation of from 1 to 10.
The non-ionic surfactant not only provides additional greasy soil cleaning performance but may also increase the anionic surfactant activity by making the anionic surfactant less likely to precipitate out of solution in the presence of free calcium cations. The weight ratio of anionic surfactant to non-ionic surfactant may be in the range of from about 0.5:1 to about 20:1, from about 0.5:1 to about 10: 1, or from about 0.5:1 to about 6:1.
The non-ionic surfactant, or at least part thereof, can be incorporated into the composition in the form of a liquid spray-on, wherein the non-ionic surfactant, or at least part thereof, in liquid form (e.g. in the form of a hot-melt) is sprayed onto the remainder of the composition. The non-ionic surfactant, or at least part thereof, may be in particulate form, and the non-ionic surfactant, or at least part thereof, may be dry-added to the remainder of the composition. The non-ionic surfactant, or at least part thereof, may be in the form of a co-particulate admixture with a solid carrier material, such as carbonate salt, inorganic sulphate salt, burkeite, silica or any mixture thereof.
The non-ionic surfactant, or at least part thereof, may be in a co-particulate admixture with either an anionic surfactant or a cationic surfactant. However the non-ionic surfactant, or at least part thereof, may not in a co-particulate admixture with both an anionic surfactant and a cationic surfactant. The non-ionic surfactant, or at least part thereof, may be agglomerated or extruded with either an anionic surfactant or a cationic surfactant. The non-ionic surfactant, or at least part thereof, may be in spray-dried powder form, optionally the non-ionic surfactant, or at least part thereof, may be spray-dried with an anionic surfactant. The non-ionic surfactant, or at least part thereof, may be in a co-particulate admixture with soap, this co-particulate admixture may be in non-spray-dried form, such as an extrudate or an agglomerate.
CATIONIC SURFACTANT
The composition may contain from about 0wt% to about 6wt%, from about 0.5wt% to about 4wt%, from about lwt% to about 3wt%, or from about lwt% to about 2wt% cationic surfactant.
Suitable cationic surfactants are alkyl pyridinium compounds, alkyl quaternary ammonium compounds, alkyl quaternary phosphonium compounds, and alkyl ternary sulphonium
compounds. The cationic surfactant can be selected from the group consisting of: alkoxylate quaternary ammonium (AQA) surfactants as described in more detail in US 6,136,769; dimethyl hydroxyethyl quaternary ammonium compounds as described in more detail in US 6,004,922; polyamine cationic surfactants as described in more detail in WO 98/35002, WO 98/35003, WO 98/35004, WO 98/35005, and WO 98/35006; cationic ester surfactants as described in more detail in US 4,228,042, US 4,239,660, US 4,260,529 and US 6,022,844; amino surfactants as described in more detail in US 6,221,825 and WO 00/47708, specifically amido propyldimethyl amine; and mixtures thereof. Preferred cationic surfactants are quaternary ammonium compounds having the general formula:
(RXR^R^R^N+X" wherein, R is a linear or branched, substituted or unsubstituted C6-I8 alkyl or alkenyl moiety, R1 and R2 are independently selected from methyl or ethyl moieties, R3 is a hydroxyl, hydroxymethyl or a hydroxyethyl moiety, X is an anion which provides charge neutrality, preferred anions include halides (such as chloride), sulphate and sulphonate. Preferred cationic surfactants are mono-Cg-io alkyl mono-hydroxyethyl di-methyl quaternary ammonium chloride, mono-Cio-12 alkyl mono-hydroxyethyl di-methyl quaternary ammonium chloride and mono-Cio alkyl mono-hydroxyethyl di-methyl quaternary ammonium chloride.
The cationic surfactant may provide additional greasy soil cleaning performance. However, the cationic surfactant may increase the tendency of the anionic surfactant to precipitate out of solution. The cationic surfactant and the anionic surfactant may be present in the composition in the form of separate particles. This minimises any effect that the cationic surfactant may have on the undesirable precipitation of the anionic surfactant, and also ensures that upon contact with water, the resultant wash liquor is not cloudy. The weight ratio of anionic surfactant to cationic surfactant may be in the range of from about 5:1 to about 25: 1, from about 5:1 to about 20:1, from about 6:1 to about 15:1, from about 7:1 to about 10:1, or from about 8:1 to about 9:1.
BUILDER
The composition may contain one or more detergent builders or builder systems. Builders include, for example, zeolite builder, phosphate builder.
ZEOLITE BUILDER
The composition contains from about Owt% to about 4wt%, from about Owt% to about 3wt%, or from about Owt% to about 2wt%. The composition may be free from deliberately added zeolite builder, i.e., may be substantially free from zeolite builder. This may be preferred if it is desirable for the composition to be very highly soluble. In addition, this is highly preferred if the composition, upon contact with water, is to form a transparent wash liquor. Zeolite builders include zeolite A, zeolite X, zeolite P and zeolite MAP.
PHOSPHATE BUILDER
The composition contains from about Owt% to about 4wt%, from about Owt% to about 3wt%, from about Owt% to about 2wt%, or from about Owt% to about lwt% phosphate builder. In one embodiment, the composition may be free from deliberately added phosphate builder, i.e., may be substantially free from phosphate builder. This may be preferred if it is desirable for the composition to have a very good environmental profile. Phosphate builders typically include sodium tripolyphosphate and sodium hydroxyethylidene Diphosphonate.
OTHER BUILDERS
The composition herein may contain other builders in addition to the zeolite builder and/or phosphate builder as described above. In one embodiment, water-soluble adjunct builders may be used. Adjunct builders may include sodium carbonate, citric acid and/or water-soluble salts thereof including sodium citrate; sulphamic acid and/or water-soluble salts thereof; polymeric polycarboxylates such as co-polymers of acrylic acid and maleic acid, or polyacrylate. The composition may contain very low levels of water-insoluble builders, such as zeolite A, zeolite X, zeolite P, and zeolite MAP, whilst containing relatively high levels of water-soluble adjunct builders, such as sodium carbonate, sulphamic acid and citric acid.
The weight ratio of sodium carbonate to zeolite builder may be at least about 5:1, at least about 10:1, at least about 15:1, at least about 20:1 or at least about 25:1.
BALANCE DETERGENT INGREDIENTS
The composition contains balance detergent ingredients, such as enzymes, silicate salts, polymeric carboxylates, solid dispersants, inorganic sulphate salts, bleaches, bleach activators, transition metal bleach catalysts, imine bleach catalysts, chelants, and other detergent ingredients.
ENZYME
The composition herein contains enzymes, e.g., protease, amylase, lipase, cellulose, mannanase, pectate lyase, bleaching enzymes, and the like.
The composition herein may contain a protease, for instance, the composition contains at least about 11 mg, at least about 15 mg, at least about 20 mg, or at least about 30 mg of active protease per 100 g of composition. The protease may contain a calcium binding site. The protease may show improved stability and/or activity in the presence of high levels of free calcium cations present in the wash liquor. By incorporating the protease into the composition, the cleaning performance of the composition is improved, and any reduction in the cleaning performance of the composition due to the low levels of, or lack of, zeolite builders and phosphate builders, which lead to a reduction in the anionic surfactant activity, is mitigated by the increased stability and/or activity of the protease. Preferred proteases include: subtilisins from Bacillus [e.g. subtilis, lentus, licheniformis, amyloliquefaciens (BPN, BPN'), alcalophilus] that are sold under the trade names Esperase®* Alcalase®, Everlase® and Savinase® supplied by Novozymes; proteases supplied by Genencor under the tradenames FN2®, FN3® and FN4®; and BLAP and/or variants thereof. Suitable proteases are described in more detail in EP 130 756, WO 91/06637, WO 95/10591 and WO 99/20726.
The composition may also contain amylase, for example, in an amount of at least about 4 mg, at least about 6 mg, at least about 10 mg, at least about 15mg, at least about 20 mg, or about 30 mg of active amylase per 100 g of the total composition. The amylase may contain a calcium binding site. Analogous to the protease, the amylase may also show improved stability and/or activity, especially stability, in the presence of high levels of free calcium cations present in the wash liquor. Thus, incorporation of amylase into the composition may improve the cleaning performance. The amylases include: amylases supplied by Novo Industries A/A under the tradenames Natalase®, Duramyl®, Termamyl®, Ban®, Fungamyl®; amylases supplied by Genencor under the tradename Purafect Ox Am®; and mixtures thereof. The amylase can be an α-amylase or a β-amylase. Suitable amylases are described in more detail in WO 94/02597 and WO 96/23873.
The composition may also contain lipase, for example, in an amount of at least about 5 mg, at least about 7 mg, at least about 10 mg, at least about 15 mg, at least about 20 mg, or at least about 30 mg of active lipase per 100 g of composition. The lipase may contain a calcium binding site. Analogous to both the protease and the amylase, the lipase may also show
improved stability and/or activity, especially activity, in the presence of high levels of free calcium cations present in the wash liquor. The incorporation of lipase into the composition improves the cleaning performance. Preferred lipases include those produced by Pseudomonas and Chromobacter groups. Preferred lipases are supplied by Novozymes under the tradenames. Lipolase®, Lipolase Ultra®, Lipoprime® and Lipex®. Other suitable lipases are cutinases and esterases.
The composition may also contain other enzymes such as: cellulases, including bacterial or fungal cellulases such as cellulases produced by Humicola insolens, and in particular cellulases supplied by Novo Industries A/A under the tradenames Carezyme®, Endo A®, other suitable cellulases are the EGIII cellulases from Trichoderma longibrachiatum; carbohydrases, including mannanase such as that described in more detail in US 6,060,299, pectate lyase such as that described in more detail in WO 99/27083, cyclomaltodextringlucanotransferase such as that described in more detail in WO 96/33267, xyloglucanase such as that described in more detail in WO 99/02663; bleaching enzymes such as peroxidases, laccases, oxygenases (e.g. catechol 1,2 dioxygenase), lipoxygenase that is described in more detail in WO 95/26393, and non-heme haloperoxidases.
The composition may also contain other enzymes such as: cellulases, including bacterial or fungal cellulases such as cellulases produced by Humicola insolens, and in particular cellulases supplied by Novo Industries A/A under the tradenames Carezyme®, Endo A®, other suitable cellulases are the EGIII cellulases from Trichoderma longibrachiatum; carbohydrases, including mannanase such as that described in more detail in US 6,060,299, pectate lyase such as that described in more detail in WO 99/27083, cyclomaltodextringlucanotransferase such as that described in more detail in WO 96/33267, xyloglucanase such as that described in more detail in WO 99/02663; bleaching enzymes such as peroxidases, laccases, oxygenases (e.g. catechol 1,2 dioxygenase), lipoxygenase that is described in more detail in WO 95/26393, and non-heme haloperoxidases.
SILICATE SALT
The composition herein may further contain silicate salt. The detergent composition may contain less than about 10wt%, from about Owt% to about 5wt%, or less than about 4wt%, or less than about 2wt% silicate salt. It may even be preferred for the detergent about composition to be free from silicate salt. Silicate salts include water-insoluble silicates. Silicate salts include
amorphous silicates and crystalline layered silicates (e.g. SKS-6). A preferred silicate salt is sodium silicate.
POLYMERIC CARBOXYLATES
The composition may contain at least about lwt%, at least about 2wt%, at least about 3wt%, at least about 4wt%, or at least 5wt% polymeric carboxylates. Polymeric carboxylates include: polyacrylates, preferably having a weight average molecular weight of from about 1,000 Da to about 20,000 Da; co-polymers of maleic acid and acrylic acid, preferably having a molar ratio of maleic acid monomers to acrylic acid monomers of from about 1:1 to about 1: 10 and a weight average molecular weight of from about 10,000 Da to about 200,000 Da, or preferably having a molar ratio of maleic acid monomers to acrylic acid monomers of from about 0.3: 1 to about 3:1 and a weight average molecular weight of from about 1,000Da to about 50,000Da.
SOIL DISPERSANT
It may also be preferred for the composition to contain a soil dispersant having the formula: bis((C2H5θ)(C2H4θ)n)(CH3)-N+-CxH2χ-N+-(CH3)-bis((C2H5θ)(C2H4O)n) wherein, n = from 20 to 30, and x = from 3 to 8. Other suitable soil dispersants are sulphonate or sulphated soil dispersants having the formula: sulphonated or sulphated bis((C2H5O)(C2H4θ)n)(CH3)-N+-CxH2x-N+-(CH3)-
Ms(CC2H5O)(C2H4O)^ wherein, n = from 20 to 30, and x = from 3 to 8. Preferably, the composition contains at least about lwt%, or at least about 2wt%, or at least about 3wt% soil dispersants, typically having the above described formulae.
INORGANIC SULPHATE SALT
The composition herein contains from about Owt% to about 10wt%, from about Owt% to about 8wt%, from about Owt% to about 6wt%, or from about Owt% to about lwt% inorganic inorganic sulphate salt. In one embodiment, the composition may be free from deliberately added inorganic sulphate salt, i.e., may be substantially free from inorganic sulphate salt. Sulphate inorganic sulphate salts include sodium sulphate, magnesium sulphate, potassium sulphate, ammonium hydrogen sulphate, ammonium sulphate, and the like. Preferably, the inorganic sulphate salt is sodium sulphate.
BLEACH
The detergent composition contains a peroxide bleach. The peroxide bleach may contain percarbonate and/or perborate, typically from about above Owt%, or from lwt% to about 7wt%, or 5wt%, or to 4wt%, or to 3wt% in total composition.
The percarbonate may be sodium percarbonate and the perborate may be sodium perborate.
BLEACH ACTIVATOR
Preferably, the composition contains a bleach activator. Suitable bleach activators are compounds which when used in conjunction with a hydrogen peroxide source leads to the in situ production of the peracid corresponding to the bleach activator. Various non limiting examples of bleach activators are disclosed in U.S. Patent 4,915,854, issued April 10, 1990 to Mao et al, and U.S. Patent 4,412,934. The nonanoyloxybenzene sulfonate (NOBS) and tetraacetylethylenediamine (TAED) activators are typical, and mixtures thereof can also be used. See also U.S. 4,634,551 for other typical bleaches and activators useful herein. Another suitable bleach activator is decanoyloxybenzenecarboxylic acid (DOBA).
Highly preferred amido-derived bleach activators are those of the formulae:
R1N(RS)C(O)R2C(O)L or R1C(O)N(RS)R2C(O)L wherein as used for these compounds R1 is an alkyl group containing from about 6 to about 12 carbon atoms, R2 is an alkylene containing from 1 to about 6 carbon atoms, RS is H or alkyl, aryl, or alkaryl containing from about 1 to about 10 carbon atoms, and L is any suitable leaving
group. A leaving group is any group that is displaced from the bleach activator as a consequence of the nucleophilic attack on the bleach activator by the hydroperoxide anion. A preferred leaving group is oxybenzenesulfonate.
Preferred examples of bleach activators of the above formulae include (6-octanamido- caproyl)oxybenzenesulfonate, (6-nonanamidocaproyl)oxybenzenesulfonate, (6-decanamido- caproyl)oxybenzenesulfonate, and mixtures thereof as described in U.S. Patent 4,634,551, incorporated herein by reference.
Another class of bleach activators contains the benzoxazin-type activators disclosed by Hodge et al in U.S. Patent 4,966,723, issued October 30, 1990, incorporated herein by reference. A highly preferred activator of the benzoxazin-type is:
Still another class of preferred bleach activators includes the acyl lactam activators, especially acyl caprolactams and acyl valerolactams of the formulae:
wherein as used for these compounds R" is H or an alkyl, aryl, alkoxyaryl, or alkaryl group containing from 1 to about 12 carbon atoms. Highly preferred lactam activators include benzoyl caprolactam, octanoyl caprolactam, 3,5,5-trimethylhexanoyl caprolactam, nonanoyl caprolactam, decanoyl caprolactam, undecenoyl caprolactam, benzoyl valerolactam, octanoyl valerolactam, decanoyl valerolactam, undecenoyl valerolactam, nonanoyl valerolactam, 3,5,5- trimethylhexanoyl valerolactam and mixtures thereof. See also U.S. Patent 4,545,784, issued to
Sanderson, October 8, 1985, incorporated herein by reference, which discloses acyl caprolactams, including benzoyl caprolactam, adsorbed into sodium perborate.
Highly preferred bleach activators are nonanoyloxybenzene sulfonate (NOBS) and/or tetraacetylethylenediamine (TAED) .
It is highly preferred for a large amount of bleach activator relative to the source of hydrogen peroxide to be present in the laundry detergent composition. Preferably, the weight ratio of bleach activator to source of hydrogen peroxide present in the laundry detergent composition is at least 0.5:1, at least 0.6:1, at least 0.7:1, 0.8:1, preferably at least 0.9: 1, or 1.0:1.0, or even 1.2: 1 or higher.
TRANSITION METAL BLEACH CATALYST
The composition may contain a transition metal bleach catalyst. The transition metal bleach catalyst typically contains a transition metal ion, preferably selected from transition metal selected from the group consisting of Mn(II), Mn(III), Mn(IV), Mn(V), Fe(II), Fe(III), Fe(IV), Co(I), Co(II), Co(III), Ni(I), Ni(II), Ni(III), Cu(I), Cu(II), Cu(III), Cr(II), Cr(III), Cr(IV), Cr(V), Cr(VI), V(III), V(IV), V(V), Mo(IV), Mo(V), Mo(VI), W(IV), W(V), W(VI), Pd(II), Ru(II), Ru(III), and Ru(IV), more preferably Mn(II), Mn(III), Mn(IV), Fe(II), Fe(III), Cr(II), Cr(III), Cr(IV), Cr(V), and Cr(VI).
The transition metal bleach catalyst typically contains a ligand, preferably a macropolycyclic ligand, more preferably a cross-bridged macropolycyclic ligand. The transition metal ion is preferably coordinated with the ligand. Preferably, the ligand contains at least four donor atoms, at least two of which are bridgehead donor atoms.
Preferably, the cross-bridged macropolycyclic ligand is coordinated by four or five donor atoms to the same transition metal and contains:
(i) an organic macrocycle ring containing four or more donor atoms selected from N and optionally O and S, at least two of these donor atoms being N (preferably at least 3, more preferably at least 4, of these donor atoms are N), separated from each other by covalent linkages of 2 or 3 non-donor atoms, two to five (preferably three to four, more preferably four) of these donor atoms being coordinated to the same transition metal in the complex;
(ii) a cross-bridging chain which covalently connects at least 2 non-adjacent N donor atoms of the organic macrocycle ring, said covalently connected non-adjacent N donor atoms being bridgehead N donor atoms which are coordinated to the same transition metal in the
complex, and wherein said cross-bridged chain contains from 2 to about 10 atoms (preferably the cross-bridged chain is selected from 2, 3 or 4 non-donor atoms, and 4-6 non-donor atoms with a further, preferably N, donor atom); and
(iii) optionally, one or more non-macropolycyclic ligands, preferably selected from the group consisting of H2O, ROH, NR3, RCN, OH", OOH", RS", RO", RCOO", OCN", SCN", N3 ", CN", F", Cl", Br", I", O2 ", NO3 ", NO2 ", SO4 2", SO3 2", PO4 3", organic phosphates, organic phosphonates, organic sulfates, organic sulfonates, and aromatic N donors such as pyridines, pyrazines, pyrazoles, imidazoles, benzimidazoles, pyrimidines, triazoles and thiazoles with R being H, optionally substituted alkyl, optionally substituted aryl.
A suitable transition metal bleach catalyst contains a complex of a transition metal and a macropolycyclic rigid ligand, preferably a cross-bridged macropolycyclic ligand, wherein:
(1) said transition metal is selected from the group consisting of Mn(II), Mn(III), Mn(IV), Mn(V), Fe(II), Fe(III), Fe(IV), Co(I), Co(II), Co(III), Ni(I), Ni(II), Ni(III), Cu(I), Cu(II), Cu(III), Cr(II), Cr(III), Cr(IV), Cr(V), Cr(VI), V(III), V(IV), V(V), Mo(IV), Mo(V), Mo(VI), W(IV), W(V), W(VI), Pd(II), Ru(II), Ru(III), and Ru(IV);
(2) said macropolycyclic rigid ligand is coordinated by at least four, preferably four or five, donor atoms to the same transition metal and contains:
(i) an organic macrocycle ring containing four or more donor atoms (preferably at least 3, more preferably at least 4, of these donor atoms are N) separated from each other by covalent linkages of at least one, preferably 2 or 3, non-donor atoms, two to five (preferably three to four, more preferably four) of these donor atoms being coordinated to the same transition metal in the complex;
(ii) a linking moiety, preferably a cross-bridging chain, which covalently connects at least 2 (preferably non-adjacent) donor atoms of the organic macrocycle ring, said covalently connected (preferably non-adjacent) donor atoms being bridgehead donor atoms which are coordinated to the same transition metal in the complex, and wherein said linking moiety (preferably a cross-bridged chain) contains from 2 to about 10 atoms (preferably the cross- bridged chain is selected from 2, 3 or 4 non-donor atoms, and 4-6 non-donor atoms with a further donor atom), including for example, a cross-bridge which is the result of a Mannich condensation of ammonia and formaldehyde; and
(iii) optionally, one or more non-macropolycyclic ligands, preferably monodentate ligands, such as those selected from the group consisting of H2O, ROH, NR3, RCN, OH", OOH",
RS", RO", RCOO", OCN", SCN", N3 ", CN", F", Cl", Br", I", O2 ", NO3 ", NO2 ", SO4 2", SO3 2", PO4 3", organic phosphates, organic phosphonates, organic sulfates, organic sulfonates, and aromatic N donors such as pyridines, pyrazines, pyrazoles, imidazoles, benzimidazoles, pyrimidines, triazoles and thiazoles with R being H, optionally substituted alkyl, optionally substituted aryl (specific examples of monodentate ligands including phenolate, acetate or the like).
Suitable cross-bridged macropolycyclic ligands include:
(i) the cross-bridged macropolycyclic ligand of formula (I) having denticity of 4 or 5:
(ii) the cross-bridged macropolycyclic ligand of formula (II) having denticity of 5 or 6:
(iii) the cross-bridged macropolycyclic ligand of formula (III) having denticity of
- each "E" is the moiety (CRn)a-X-(CRn)a', wherein -X- is selected from the group consisting of O, S, NR and P, or a covalent bond, and preferably X is a covalent bond and for each E the sum of a + a' is independently selected from 1 to 5, more preferably 2 and 3;
- each "G" is the moiety (CRnJt,;
- each "R" is independently selected from H, alkyl, alkenyl, alkynyl, aryl, alkylaryl (e.g., benzyl), and heteroaryl, or two or more R are covalently bonded to form an aromatic, heteroaromatic, cycloalkyl, or heterocycloalkyl ring;
- each "D" is a donor atom independently selected from the group consisting of N, O, S, and P, and at least two D atoms are bridgehead donor atoms coordinated to the transition metal (in the preferred embodiments, all donor atoms designated D are donor atoms which coordinate to the transition metal, in contrast with heteroatoms in the structure which are not in D such as those which may be present in E; the non-D heteroatoms can be non-coordinating and indeed are non-coordinating whenever present in the preferred embodiment);
- "B" is a carbon atom or "D" donor atom, or a cycloalkyl or heterocyclic ring;
- each "n" is an integer independently selected from 1 and 2, completing the valence of the carbon atoms to which the R moieties are covalently bonded;
- each "n"' is an integer independently selected from 0 and 1, completing the valence of the D donor atoms to which the R moieties are covalently bonded;
- each "n"" is an integer independently selected from 0, 1, and 2 completing the valence of the B atoms to which the R moieties are covalently bonded;
- each "a" and "a'"is an integer independently selected from 0-5, preferably a + a' equals 2 or 3, wherein the sum of all "a" plus "a"' in the ligand of formula (I) is within the range of from about 6 (preferably 8) to about 12, the sum of all "a" plus "a"' in the ligand of formula (II) is within the range of from about 8 (preferably 10) to about 15, and the sum of all "a" plus "a"' in the ligand of formula (III) is within the range of from about 10 (preferably 12) to about 18;
- each "b" is an integer independently selected from 0-9, preferably 0-5 (wherein when b=0, (CRn)o represents a covalent bond), or in any of the above formulas, one or more of the (CRn)b moieties covalently bonded from any D to the B atom is absent as long as at least two (CRn)b covalently bond two of the D donor atoms to the B atom in the formula, and the sum of all "b" is within the range of from about 1 to about 5.
A suitable cross-bridged macropolycyclic ligand is selected from the group consisting of:
(I), and
(H),
wherein in these formulas:
- each "R" is independently selected from H, alkyl, alkenyl, alkynyl, aryl, alkylaryl (e.g., benzyl) and heteroaryl, or two or more R are covalently bonded to form an aromatic, heteroaromatic, cycloalkyl, or heterocycloalkyl ring;
- each "n" is an integer independently selected from 0, 1 and 2, completing the valence of the carbon atoms to which the R moieties are covalently bonded;
- each "b" is an integer independently selected from 2 and 3; and
- each "a" is an integer independently selected from 2 and 3.
Suitable transition metal bleach catalysts include: Dichloro-5,12-dimethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-4,10-dimethyl-l,4,7,10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Diaquo-5,12-dimethyl- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Manganese(II) Hexafluorophosphate; Aquo-hydroxy-5,12- dimethyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(III) Hexafluorophosphate; Diaquo- 4, 10-dimethyl- 1 ,4,7, 10-tetraazabicyclo[5.5.2]tetradecane Manganese(II) Hexafluorophosphate; Diaquo-5,12-dimethyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(II) Tetrafluoroborate; Diaquo^JO-dimethyl-l^JJO-tetraazabicyclofS.S^Jtetradecane Manganese(II) Tetrafluoroborate; Dichloro-5,12-dimethyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(III);
Hexafluorophosphate; Dichloro-5,12-di-n-butyl-l,5,8,12-tetraaza- bicyclo[6.6.2]hexadecane Manganese(II); Dichloro-5,12-dibenzyl- 1,5,8, 12-tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-S-n-butyl-12-methyl- 1,5,8, 12-tetraaza- bicyclo[6.6.2]hexadecane Manganese(II); Dichloro-5-n-octyl-12-methyl-l,5,8,12-tetraaza- bicyclo[6.6.2]hexadecane Manganese(II);
Dichloro-S-n-butyl-^-methyl- 1,5,8, 12-tetraaza- bicyclo[6.6.2]hexadecane Manganese(II); Dichloro-5 , 12-dimethyl- 1,5,8,12-tetraazabicyclo [6.6.2]hexadecane Iron(II) ; Dichloro-4, 10- dimethyM,4,7,10-tetraazabicyclo[5Λ2]tetradecane Iron(II); Dichloro-5, 12-dimethyl-l,5, 8,12- tetraazabicyclo[6.6.2]hexadecane Copper(II); Dichloro-4, 10-dimethyl- 1,4,7, 10- tetraazabicyclo[5.5.2]tetradecane Copper(II); Dichloro-5, 12-dimethyl- 1, 5, 8,12- tetraazabicyclo[6.6.2]hexadecane Cobalt(II); Dichloro-4, 10-dimethyl- 1,4,7, 10- tetraazabicyclo[5.5.2]tetradecane Cobalt(II); Dichloro 5,12-dimethyl-4-phenyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-4, 10-dimethyl-3-phenyl-l ,4,7, 10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Dichloro-5, 12-dimethyl-4,9-diphenyl- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-4, 10-dimethyl-3,8-diphenyl-l, 4,7, 10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Dichloro-5, 12-dimethyl-2,ll-diphenyl- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-4, 10-dimethyl-4,9-diphenyl-l, 4,7, 10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Dichloro-2,4, 5,9,1 l,12-hexamethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-2,3,5,9,10,12-hexamethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-2,2,4,5,9,9,ll,12-octamethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II) ; Dichloro-2,2,4,5 ,9, 11,11, 12-octamethyl- 1,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-3,3,5,10,10,12-hexamethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-3,5,10,12-tetramethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro-3-butyl-5,10,12-trimethyl-l,5,8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro- 1,4,7, 10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Dichloro- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Iron(II); Dichloro-l,4,7,10-tetraazabicyclo[5.5.2]tetradecane Iron(II); Aquo-chloro-2-(2-hydroxyphenyl)-5,12-dimethyl,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(II); Aquo-chloro-10-(2-hydroxybenzyl)-4,10-dimethyl-l,4,7,10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Chloro-2-(2-hydroxybenzyl)-5-methy 1,5, 8,12- tetraazabicyclo[6.6.2]hexadecane Manganese(II); Chloro-10-(2-hydroxybenzyl)-4-methyl- 1,4,7, 10- tetraazabicyclo[5.5.2]tetradecane Manganese(II); Chloro-5-methyl-12-(2-picolyl)- 1,5,8, 12- tetraazabicyclo[6.6.2]hexadecane Manganese(II) Chloride; Chloro-4-methyl-10-(2-picolyl)- 1,4,7, 10-tetraazabicyclo[5.5.2]tetradecane Manganese(II) Chloride; Dichloro-5 -(2-sulf ato)dodecyl- 12-methyl- 1,5,8, 12-tetraazabicyclo[6.6.2]hexadecane Manganese(III); Aquo-Chloro-5-(2- sulfato)dodecyl-12-methyl- 1,5,8, 12-tetraazabicyclo[6.6.2]hexadecane Manganese(II); Aquo-Chloro-
5-(3-sulfonopropyl)-12-methyl- 1,5,8, 12-tetraazabicyclo[6.6.2]hexadecane Manganese(II); Dichloro- 5-(Trimethylammoniopropyl)dodecyl-12-methyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(III) Chloride; Dichloro-5,12-dimethyl-l,4,7,10,13-pentaazabicyclo[8.5.2]heptadecane Manganese(II); Dichloro-14,20-dimethyl-l,10,14,20-tetraazatriyclo[8.6.6]docosa-3(8),4,6-triene Manganese(II); Dichloro-4, 11 -dimethyl- 1,4,7, ll-tetraazabicyclo[6.5.2]pentadecane Manganese(II); Dichloro-5,12-dimethyl-l,5,8,12-tetraazabicyclo[7.6.2]heptadecane Manganese(II); Dichloro-5,13- dimethyl- 1,5,9, 13-tetraazabicyclo[7.7.2]heptadecane Manganese(II); Dichloro-3,10- bis(butylcarboxy)-5,12-dimethyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(II); Diaquo- 3,10-dicarboxy-5,12-dimethyl-l,5,8,12-tetraazabicyclo[6.6.2]hexadecane Manganese(II); Chloro-
20-methyl-l,9,20,24,25-pentaaza-tetracyclo[7.7.7.13'7.l1 1'15.]pentacosa-3,5,7(24),l 1,13,15(25)- hexaene manganese(II) Hexafluorophosphate; Trifluoromethanesulfono-20-methyl- 1, 9,20,24,25- pentaaza-tetracyclo[7.7.7.l3,7j ll,15.]pentacosa-3, 5,7(24), 11, 13, 15(25)-hexaene Manganese(II) Trifluoromethanesulfonate; Trifluoromethanesulfono-20-methyl-l, 9,20,24 ,25-pentaaza- tetracyclo[7.7.7.13'7.l1 1'15.]pentacosa-3,5,7(24),H,13,15(25)-hexaene Iron(II)
Trifluoromethanesulfonate; Chloro-5,12,17-trimethyl-l,5,8,12,17- pentaazabicyclo[6.6.5]nonadecane Manganese(II) Hexafluorophosphate; Chloro-4,10,15-trimethyl- 1,4,7, 10,15-pentaazabicyclo[5.5.5]heptadecane Manganese(II) Hexafluorophosphate; Chloro- 5,12,17-trimethyl-l,5,8,12,17-pentaazabicyclo[6.6.5]nonadecane Manganese(II) Chloride; Chloro- 4,10,15-trimethyl-l,4,7,10,15-pentaazabicyclo[5.5.5]heptadecane Manganese(II) Chloride; and any mixture thereof.
Other suitable transition metal bleach catalysts are described in U.S. 5,580,485, U.S. 4,430,243; U.S. 4,728,455; U.S. 5,246,621; U.S. 5,244,594; U.S. 5,284,944; U.S. 5,194,416; U.S. 5,246,612; U.S. 5,256,779; U.S. 5,280,117; U.S. 5,274,147; U.S. 5,153,161; U.S. 5,227,084; U.S. 5,114,606; U.S. 5,114,611, EP 549,271 Al; EP 544,490 Al; EP 549,272 Al; and EP 544,440 A2.
A suitable transition metal bleach catalyst is a manganese-based catalyst, for example disclosed in U.S. 5,576,282.
Suitable cobalt bleach catalysts are described, for example, in U.S. 5,597,936 andU.S. 5,595,967. Such cobalt catalysts are readily prepared by known procedures, such as taught for example in U.S. 5,597,936, and U.S. 5,595,967.
A suitable transition metal bleach catalyst is a transition metal complex of ligand such as bispidones described in WO 05/042532 Al.
Transition metal bleach catalysts provide robust cleaning profiles, especially under dilute wash conditions, and especially against beta-carotene, squalene and unsaturated triglyceride soils.
IMINE BLEACH CATALYST
The composition may contain an imine bleach catalyst. Suitable imine bleach catalysts include, but are not limited to: iminium cations and polyions; iminium zwitterions; N-sulphonyl imines; N-phosphonyl imines; N-acyl imines; perfluoroimines; and mixtures thereof.
Suitable iminium cations and polyions include, but are not limited to, N-methyl-3,4- dihydroisoquinolinium tetrafluoroborate, prepared as described in Tetrahedron (1992), 49(2), 423-38 (see, for example, compound 4, p. 433); N-methyl-3,4-dihydroisoquinolinium p-toluene sulphonate, prepared as described in U.S. Pat. 5,360,569 (see, for example, Column 11, Example 1); and N-octyl-3,4-dihydroisoquinolinium p-toluene sulphonate, prepared as described in U.S. Pat. 5,360,568 (see, for example, Column 10, Example 3).
Suitable iminium zwitterions include, but are not limited to, N-(3-sulfopropyl)-3,4- dihydroisoquinolinium, inner salt, prepared as described in U.S. Pat. 5,576,282 (see, for example, Column 31, Example II); N-[2-(sulphooxy)dodecyl]-3,4-dihydroisoquinolinium, inner salt, prepared as described in U.S. Pat. 5,817,614 (see, for example, Column 32, Example V); 2- [3-[(2-ethylhexyl)oxy]-2-(sulphooxy)propyl]-3,4-dihydroisoquinolinium, inner salt, prepared as described in WO05/047264 (see, for example, page 18, Example 8), and 2-[3-[(2- butyloctyl)oxy]-2-(sulphooxy)propyl]-3,4-dihydroisoquinolinium, inner salt.
Suitable N-sulphonyl imine oxygen transfer catalysts include, but are not limited to, 3- methyl-l,2-benzisothiazole 1,1 -dioxide, prepared according to the procedure described in the Journal of Organic Chemistry (1990), 55(4), 1254-61.
Suitable N-phosphonyl imine oxygen transfer catalysts include, but are not limited to, [R- (E)]-N-[(2-chloro-5-nitrophenyl)methylene]-P-phenyl-P-(2,4,6-trimethylphenyl)- phosphinic amide, which can be made according to the procedures described in the Journal of the Chemical Society, Chemical Communications (1994), (22), 2569-70.
Suitable N-acyl imine oxygen transfer catalysts include, but are not limited to, [N(E)J-N- (phenylmethylene)acetamide, which can be made according to the procedures described in Polish Journal of Chemistry (2003), 77(5), 577-590.
Suitable perfluoroimine oxygen transfer catalysts include, but are not limited to, (Z)- 2,2,3,3,4,4,4-heptafluoro-N-(nonafluorobutyl)butanimidoyl fluoride, which can be made according to the procedures described in Tetrahedron Letters (1994), 35(34), 6329-30.
Suitable cyclic sugar ketone oxygen transfer catalysts include, but are not limited to, l,2:4,5-di-O-isopropylidene-D-erythro-2,3-hexodiuro-2,6-pyranose as prepared in U.S. Pat. 6,649,085 (Column 12, Example 1).
Preferably, the imine bleach catalyst contains an iminium and/or carbonyl functional group and is typically capable of forming an oxaziridinium and/or dioxirane functional group upon acceptance of an oxygen atom, especially upon acceptance of an oxygen atom from a peroxyacid and/or salt thereof. Preferably, the imine bleach catalyst contains an oxaziridinium functional group and/or is capable of forming an oxaziridinium functional group upon acceptance of an oxygen atom, especially upon acceptance of an oxygen atom from a peroxyacid and/or salt thereof. Preferably, the imine bleach catalyst contains a cyclic iminium functional group, preferably wherein the cyclic moiety has a ring size of from five to eight atoms (including the nitrogen atom), preferably six atoms. Preferably, the imine bleach catalyst contains an aryliminium functional group, preferably a bi-cyclic aryliminium functional group, preferably a 3,4-dihydroisoquinolinium functional group. Typically, the imine functional group is a quaternary imine functional group and is typically capable of forming a quaternary oxaziridinium functional group upon acceptance of an oxygen atom, especially upon acceptance of an oxygen atom from a peroxyacid and/or salt thereof.
Preferably, the imine bleach catalyst has a chemical structure corresponding to the following chemical formula
wherein: n and m are independently from 0 to 4, preferably n and m are both 0; each R is independently selected from a substituted or unsubstituted radical selected from the group consisting of hydrogen, alkyl, cycloalkyl, aryl, fused aryl, heterocyclic ring, fused heterocyclic
ring, nitro, halo, cyano, sulphonato, alkoxy, keto, carboxylic, and carboalkoxy radicals; and any two vicinal R1 substituents may combine to form a fused aryl, fused carbocyclic or fused heterocyclic ring; each R2 is independently selected from a substituted or unsubstituted radical independently selected from the group consisting of hydrogen, hydroxy, alkyl, cycloalkyl, alkaryl, aryl, aralkyl, alkylenes, heterocyclic ring, alkoxys, arylcarbonyl groups, carboxyalkyl groups and amide groups; any R2 may be joined together with any other of R2 to form part of a common ring; any geminal R2 may combine to form a carbonyl; and any two R2 may combine to form a substituted or unsubstituted fused unsaturated moiety; R3 is a Ci to C20 substituted or unsubstituted alkyl; R4 is hydrogen or the moiety Q1-A, wherein: Q is a branched or unbranched alkylene, t = 0 or 1 and A is an anionic group selected from the group consisting of OS(V, SO3 ", CO2 ", OCO2 ", OPO3 2", OPO3H" and OPO2 "; R5 is hydrogen or the moiety
[(CR9R10)y-O]k-R8, wherein: each Y is independently selected from the group consisting of O, S,
N-H, or N-R ; and each R is independently selected from the group consisting of alkyl, aryl and heteroaryl, said moieties being substituted or unsubstituted, and whether substituted or unsubsituted said moieties having less than 21 carbons; each G is independently selected from the group consisting of CO, SO2, SO, PO and PO2; R9 and R10 are independently selected from the group consisting of H and Ci-C4 alkyl; R11 and R12 are independently selected from the group consisting of H and alkyl, or when taken together may join to form a carbonyl; b = O or 1; c can = O or 1, but c must = O if b = O; y is an integer from 1 to 6; k is an integer from O to 20; R6 is H, or an alkyl, aryl or heteroaryl moiety; said moieties being substituted or unsubstituted; and X, if present, is a suitable charge balancing counterion, preferably X is present when R4 is hydrogen, suitable X, include but are not limited to: chloride, bromide, sulphate, methosulphate, sulphonate, p-toluenesulphonate, borontetraflouride and phosphate.
In one embodiment of the present invention, the imine bleach catalyst has a structure corresponding to general formula below:
wherein R » 13 is a branched alkyl group containing from three to 24 carbon atoms (including the branching carbon atoms) or a linear alkyl group containing from one to 24 carbon atoms;
preferably R13 is a branched alkyl group containing from eight to 18 carbon atoms or linear alkyl group containing from eight to eighteen carbon atoms; preferably R13 is selected from the group consisting of 2-ethylhexyl, 2-propylheptyl, 2-butyloctyl, 2-pentylnonyl, 2-hexyldecyl, n- dodecyl, n-tetradecyl, n-hexadecyl, n-octadecyl, iso-nonyl, iso-decyl, iso-tridecyl and iso- pentadecyl; preferably R13 is selected from the group consisting of 2-butyloctyl, 2-pentylnonyl, 2-hexyldecyl, iso-tridecyl and iso-pentadecyl.
In another embodiment of the present invention, the imine bleach catalyst has a structure corresponding to general formula below or mixtures thereof.
wherein: G is selected from -O-, -CH2O-, -(CH2)2-, and -CH2-. R1 is selected from H or Ci-C4 alkyl. Suitable Ci-C4 alkyl moieties include, but are not limited to methyl, ethyl, iso-propyl, and tert-butyl. Each R2 is independently selected from C4-Cs alkyl, benzyl, 2-methylbenzyl, 3- methylbenzyl, 4-methylbenzyl, 4-ethylbenzyl, 4-iso-propylbenzyl and 4-tert-butylbenzyl. Suitable C4-Cs alkyl moieties include, but are not limited to n-butyl, n-pentyl, cyclopentyl, n- hexyl, cyclohexyl, cyclohexylmethyl, n-heptyl and octyl.
In one aspect of the invention G is selected from -O- and -CH2-. R1 is selected from H, methyl, ethyl, iso-propyl, and tert-butyl. Each R2 is independently selected from C4-C6 alkyl, benzyl, 2-methylbenzyl, 3-methylbenzyl, and 4-methylbenzyl.
In another aspect of the invention G is -CH2-, R1 is H and each R2 is independently selected from n-butyl, n-pentyl, n-hexyl, benzyl, 2-methylbenzyl, 3-methylbenzyl, and 4- methylbenzyl.
CHELANT
The composition may contain a chelant. Suitable chelants include diethylene triamine pentaacetate, diethylene triamine penta(methyl phosphonic acid), ethylene diamine-N'N'- disuccinic acid, ethylene diamine tetraacetate, ethylene diamine tetra(methylene phosphonic acid) and hydroxyethane di(methylene phosphonic acid). A preferred chelant is ethylene
diamine-N'N'-disuccinic acid (EDDS) and/or hydroxyethane diphosphonic acid (HEDP). Preferably the ethylene diamine-N'N'-disuccinic acid is in S'S' enantiomeric form.
OTHER DETERGENT INGREDIENTS
The composition may contain a detergent ingredient selected from: suds suppressing systems such as silicone based suds suppressors; brighteners; photobleach; filler salts; fabric- softening agents such as clay, silicone and/or quaternary ammonium compounds; flocculants such as polyethylene oxide; dye transfer inhibitors such as polyvinylpyrrolidone, poly 4- vinylpyridine N-oxide and/or co-polymer of vinylpyrrolidone and vinylimidazole; fabric integrity components such as hydrophobically modified cellulose and oligomers produced by the condensation of imidazole and epichlorhydrin; soil dispersants and soil anti-redeposition aids such as polycarboxylates, alkoxylated polyamines and ethoxylated ethyleneimine polymers; and anti-redeposition components such as carboxymethyl cellulose and polyesters.
EXAMPLES
30 g of the following free-flowing particulate laundry detergent compositions were used to wash 3.0 kg fabric in a Miele 3622 front-loading automatic washing machine (13L wash liquor volume, short wash cycle, Ih, 25 mines), 300C wash temperature).
The dimensions and values disclosed herein are not to be understood as being strictly limited to the exact numerical values recited. Instead, unless otherwise specified, each such dimension is intended to mean both the recited value and a functionally equivalent range surrounding that value. For example, a dimension disclosed as "40 mm" is intended to mean "about 40 mm."
Every document cited herein, including any cross referenced or related patent or application, is hereby incorporated herein by reference in its entirety unless expressly excluded or otherwise limited. The citation of any document is not an admission that it is prior art with respect to any invention disclosed or claimed herein or that it alone, or in any combination with any other reference or references, teaches, suggests or discloses any such invention. Further, to the extent that any meaning or definition of a term in this document conflicts with any meaning or definition of the same term in a document incorporated by reference, the meaning or definition assigned to that term in this document shall govern.
While particular embodiments of the present invention have been illustrated and described, it would be obvious to those skilled in the art that various other changes and modifications can be made without departing from the spirit and scope of the invention. It is therefore intended to cover in the appended claims all such changes and modifications that are within the scope of this invention.
Claims
1. A solid laundry detergent composition comprising:
(i) from about lwt% to about 60wt% detersive surfactant,
(ii) from about 0wt% to about 4wt% zeolite builder,
(iii) from about 0wt% to about 4wt% phosphate builder,
(iv) from about above 0wt% to about 7wt% peroxide bleach selected from the group consisting of percarbonate, perborate and a mixture thereof, and
(v) balance additional detergent ingredients.
2. A composition of Claim 1 , wherein the composition comprises anionic surfactant.
3. A composition of Claim 2, comprising from about 4wt% to about 40wt% anionic surfactant.
4. A composition of Claim 1, wherein the composition comprises non-ionic surfactant.
5. A composition of Claim 1, wherein the composition comprises cationic surfactant.
6. A composition of Claim 1 , wherein the composition comprises enzyme.
7. A composition of Claim 6, wherein the enzyme is selected from the group consisting of cellulase, protease, lipase, amylase, and a mixture thereof.
8. A composition of Claim 1, wherein the composition is substantially free from zeolite builder.
9. A composition of Claim 1, wherein the composition is substantially free from phosphate builder.
10. A composition of Claim 1, wherein the composition is a free-flowing particulate laundry detergent composition.
11. A composition of Claim 1 , further comprising a bleach activator.
12. A composition of Claim 1, further comprising sodium percarbonate.
13. A composition of Claim 1, wherein the composition comprises from about lwt% to about 4wt% peroxide bleach.
14. A composition of Claim 1, wherein the composition comprises a transition metal bleach catalyst.
15. A composition of Claim 1, wherein the composition comprises an imine bleach catalyst.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US22416709P | 2009-07-09 | 2009-07-09 | |
| US61/224,167 | 2009-07-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011005623A1 true WO2011005623A1 (en) | 2011-01-13 |
Family
ID=42728296
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/040506 WO2011005623A1 (en) | 2009-07-09 | 2010-06-30 | Laundry detergent composition comprising low level of bleach |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2011005623A1 (en) |
Cited By (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011134809A1 (en) | 2010-04-26 | 2011-11-03 | Novozymes A/S | Enzyme granules |
| WO2012101149A1 (en) | 2011-01-26 | 2012-08-02 | Novozymes A/S | Storage-stable enzyme granules |
| WO2012175401A2 (en) | 2011-06-20 | 2012-12-27 | Novozymes A/S | Particulate composition |
| WO2012175708A2 (en) | 2011-06-24 | 2012-12-27 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2013001087A2 (en) | 2011-06-30 | 2013-01-03 | Novozymes A/S | Method for screening alpha-amylases |
| WO2013007594A1 (en) | 2011-07-12 | 2013-01-17 | Novozymes A/S | Storage-stable enzyme granules |
| WO2013024021A1 (en) | 2011-08-15 | 2013-02-21 | Novozymes A/S | Polypeptides having cellulase activity and polynucleotides encoding same |
| WO2013041689A1 (en) | 2011-09-22 | 2013-03-28 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2013076269A1 (en) | 2011-11-25 | 2013-05-30 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2013092635A1 (en) | 2011-12-20 | 2013-06-27 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2013110766A1 (en) | 2012-01-26 | 2013-08-01 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2013120948A1 (en) | 2012-02-17 | 2013-08-22 | Novozymes A/S | Subtilisin variants and polynucleotides encoding same |
| WO2013131964A1 (en) | 2012-03-07 | 2013-09-12 | Novozymes A/S | Detergent composition and substitution of optical brighteners in detergent compositions |
| WO2013167581A1 (en) | 2012-05-07 | 2013-11-14 | Novozymes A/S | Polypeptides having xanthan degrading activity and polynucleotides encoding same |
| WO2013189972A2 (en) | 2012-06-20 | 2013-12-27 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2014096259A1 (en) | 2012-12-21 | 2014-06-26 | Novozymes A/S | Polypeptides having protease activiy and polynucleotides encoding same |
| WO2014183921A1 (en) | 2013-05-17 | 2014-11-20 | Novozymes A/S | Polypeptides having alpha amylase activity |
| WO2014207227A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2014207224A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2015001017A2 (en) | 2013-07-04 | 2015-01-08 | Novozymes A/S | Polypeptides having anti-redeposition effect and polynucleotides encoding same |
| EP2832853A1 (en) | 2013-07-29 | 2015-02-04 | Henkel AG&Co. KGAA | Detergent composition comprising protease variants |
| WO2015049370A1 (en) | 2013-10-03 | 2015-04-09 | Novozymes A/S | Detergent composition and use of detergent composition |
| WO2015134737A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015134729A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of non-cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015150457A1 (en) | 2014-04-01 | 2015-10-08 | Novozymes A/S | Polypeptides having alpha amylase activity |
| WO2015189371A1 (en) | 2014-06-12 | 2015-12-17 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2016079305A1 (en) | 2014-11-20 | 2016-05-26 | Novozymes A/S | Alicyclobacillus variants and polynucleotides encoding same |
| EP3106508A1 (en) | 2015-06-18 | 2016-12-21 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| US9539190B2 (en) | 2010-05-07 | 2017-01-10 | Conopco, Inc. | Skin conditioning compositions containing 12-hydroxystearic acid |
| WO2017064253A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2017064269A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptide variants |
| WO2017207762A1 (en) | 2016-06-03 | 2017-12-07 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2018011277A1 (en) | 2016-07-13 | 2018-01-18 | Novozymes A/S | Bacillus cibi dnase variants |
| EP3309249A1 (en) | 2013-07-29 | 2018-04-18 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3321360A2 (en) | 2013-01-03 | 2018-05-16 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| EP3453757A1 (en) | 2013-12-20 | 2019-03-13 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2019081721A1 (en) | 2017-10-27 | 2019-05-02 | Novozymes A/S | Dnase variants |
| WO2019084349A1 (en) | 2017-10-27 | 2019-05-02 | The Procter & Gamble Company | Detergent compositions comprising polypeptide variants |
| WO2019201793A1 (en) | 2018-04-17 | 2019-10-24 | Novozymes A/S | Polypeptides comprising carbohydrate binding activity in detergent compositions and their use in reducing wrinkles in textile or fabric. |
| EP3608403A2 (en) | 2014-12-15 | 2020-02-12 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| EP3611260A1 (en) | 2013-07-29 | 2020-02-19 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3690037A1 (en) | 2014-12-04 | 2020-08-05 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2020188095A1 (en) | 2019-03-21 | 2020-09-24 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2020207944A1 (en) | 2019-04-10 | 2020-10-15 | Novozymes A/S | Polypeptide variants |
| EP3739029A1 (en) | 2014-07-04 | 2020-11-18 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3786269A1 (en) | 2013-06-06 | 2021-03-03 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2021037895A1 (en) | 2019-08-27 | 2021-03-04 | Novozymes A/S | Detergent composition |
| WO2021053127A1 (en) | 2019-09-19 | 2021-03-25 | Novozymes A/S | Detergent composition |
| WO2021064068A1 (en) | 2019-10-03 | 2021-04-08 | Novozymes A/S | Polypeptides comprising at least two carbohydrate binding domains |
| EP3872175A1 (en) | 2015-06-18 | 2021-09-01 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3878960A1 (en) | 2014-07-04 | 2021-09-15 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3892708A1 (en) | 2020-04-06 | 2021-10-13 | Henkel AG & Co. KGaA | Cleaning compositions comprising dispersin variants |
| WO2022074037A2 (en) | 2020-10-07 | 2022-04-14 | Novozymes A/S | Alpha-amylase variants |
| WO2022171780A2 (en) | 2021-02-12 | 2022-08-18 | Novozymes A/S | Alpha-amylase variants |
| WO2022268885A1 (en) | 2021-06-23 | 2022-12-29 | Novozymes A/S | Alpha-amylase polypeptides |
Citations (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1154344A (en) * | 1955-07-13 | 1958-04-04 | Henkel & Cie Gmbh | Washing and bleaching agents |
| US3630921A (en) * | 1968-12-03 | 1971-12-28 | Henkel & Cie Gmbh | Scouring agents with a bleaching and disinfecting action |
| US4228042A (en) | 1978-06-26 | 1980-10-14 | The Procter & Gamble Company | Biodegradable cationic surface-active agents containing ester or amide and polyalkoxy group |
| US4239660A (en) | 1978-12-13 | 1980-12-16 | The Procter & Gamble Company | Detergent composition comprising a hydrolyzable cationic surfactant and specific alkalinity source |
| US4260529A (en) | 1978-06-26 | 1981-04-07 | The Procter & Gamble Company | Detergent composition consisting essentially of biodegradable nonionic surfactant and cationic surfactant containing ester or amide |
| US4412934A (en) | 1982-06-30 | 1983-11-01 | The Procter & Gamble Company | Bleaching compositions |
| US4430243A (en) | 1981-08-08 | 1984-02-07 | The Procter & Gamble Company | Bleach catalyst compositions and use thereof in laundry bleaching and detergent compositions |
| US4483779A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions comprising polyglycoside and polyethoxylate surfactants and anionic fluorescer |
| US4483780A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions containing polyglycoside and polyethoxylate detergent surfactants |
| EP0130756A1 (en) | 1983-06-24 | 1985-01-09 | Genencor International, Inc. | Procaryotic carbonyl hydrolases, methods, DNA, vectors and transformed hosts for producing them, and detergent compositions containing them |
| US4545784A (en) | 1983-04-14 | 1985-10-08 | Interox Chemicals Limited | Particulate sodium perborate monohydrate containing adsorbed activator |
| US4565647A (en) | 1982-04-26 | 1986-01-21 | The Procter & Gamble Company | Foaming surfactant compositions |
| US4634551A (en) | 1985-06-03 | 1987-01-06 | Procter & Gamble Company | Bleaching compounds and compositions comprising fatty peroxyacids salts thereof and precursors therefor having amide moieties in the fatty chain |
| US4728455A (en) | 1986-03-07 | 1988-03-01 | Lever Brothers Company | Detergent bleach compositions, bleaching agents and bleach activators |
| US4915854A (en) | 1986-11-14 | 1990-04-10 | The Procter & Gamble Company | Ion-pair complex conditioning agent and compositions containing same |
| US4966723A (en) | 1988-02-11 | 1990-10-30 | Bp Chemicals Limited | Bleach activators in detergent compositions |
| WO1991006637A1 (en) | 1989-10-31 | 1991-05-16 | Genencor International, Inc. | Subtilisin mutants |
| WO1992006162A1 (en) | 1990-09-28 | 1992-04-16 | The Procter & Gamble Company | Detergent containing alkyl sulfate and polyhydroxy fatty acid amide surfactants |
| US5114606A (en) | 1990-02-19 | 1992-05-19 | Lever Brothers Company, Division Of Conopco, Inc. | Bleaching composition comprising as a bleaching catalyst a complex of manganese with a non-carboxylate polyhydroxy ligand |
| US5114611A (en) | 1989-04-13 | 1992-05-19 | Lever Brothers Company, Divison Of Conopco, Inc. | Bleach activation |
| US5153161A (en) | 1991-11-26 | 1992-10-06 | Lever Brothers Company, Division Of Conopco, Inc. | Synthesis of manganese oxidation catalyst |
| US5194416A (en) | 1991-11-26 | 1993-03-16 | Lever Brothers Company, Division Of Conopco, Inc. | Manganese catalyst for activating hydrogen peroxide bleaching |
| EP0544440A2 (en) | 1991-11-20 | 1993-06-02 | Unilever Plc | Bleach catalyst composition, manufacture and use thereof in detergent and/or bleach compositions |
| EP0544490A1 (en) | 1991-11-26 | 1993-06-02 | Unilever Plc | Detergent bleach compositions |
| EP0549271A1 (en) | 1991-12-20 | 1993-06-30 | Unilever Plc | Bleach activation |
| EP0549272A1 (en) | 1991-12-20 | 1993-06-30 | Unilever Plc | Bleach activation |
| US5227084A (en) | 1991-04-17 | 1993-07-13 | Lever Brothers Company, Division Of Conopco, Inc. | Concentrated detergent powder compositions |
| US5244594A (en) | 1990-05-21 | 1993-09-14 | Lever Brothers Company, Division Of Conopco, Inc. | Bleach activation multinuclear manganese-based coordination complexes |
| US5246612A (en) | 1991-08-23 | 1993-09-21 | Lever Brothers Company, Division Of Conopco, Inc. | Machine dishwashing composition containing peroxygen bleach, manganese complex and enzymes |
| WO1993019038A1 (en) | 1992-03-26 | 1993-09-30 | The Procter & Gamble Company | Process for reducing the levels of fatty acid contaminants in polyhydroxy fatty acid amide surfactants |
| WO1993019146A1 (en) | 1992-03-16 | 1993-09-30 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| US5256779A (en) | 1992-06-18 | 1993-10-26 | Lever Brothers Company, Division Of Conopco, Inc. | Synthesis of manganese oxidation catalyst |
| US5274147A (en) | 1991-07-11 | 1993-12-28 | Lever Brothers Company, Division Of Conopco, Inc. | Process for preparing manganese complexes |
| US5280117A (en) | 1992-09-09 | 1994-01-18 | Lever Brothers Company, A Division Of Conopco, Inc. | Process for the preparation of manganese bleach catalyst |
| WO1994002597A1 (en) | 1992-07-23 | 1994-02-03 | Novo Nordisk A/S | MUTANT α-AMYLASE, DETERGENT, DISH WASHING AGENT, AND LIQUEFACTION AGENT |
| US5284944A (en) | 1992-06-30 | 1994-02-08 | Lever Brothers Company, Division Of Conopco, Inc. | Improved synthesis of 1,4,7-triazacyclononane |
| WO1994009099A1 (en) | 1992-10-13 | 1994-04-28 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| US5332528A (en) | 1990-09-28 | 1994-07-26 | The Procter & Gamble Company | Polyhydroxy fatty acid amides in soil release agent-containing detergent compositions |
| US5360568A (en) | 1993-11-12 | 1994-11-01 | Lever Brothers Company, Division Of Conopco, Inc. | Imine quaternary salts as bleach catalysts |
| US5360569A (en) | 1993-11-12 | 1994-11-01 | Lever Brothers Company, Division Of Conopco, Inc. | Activation of bleach precursors with catalytic imine quaternary salts |
| WO1995010591A1 (en) | 1993-10-14 | 1995-04-20 | The Procter & Gamble Company | Protease-containing cleaning compositions |
| WO1995026393A1 (en) | 1994-03-29 | 1995-10-05 | The Procter & Gamble Company | Detergent composition comprising lipoxidase enzymes |
| WO1995028468A1 (en) * | 1994-04-13 | 1995-10-26 | The Procter & Gamble Company | Detergent compositions |
| WO1996023873A1 (en) | 1995-02-03 | 1996-08-08 | Novo Nordisk A/S | Amylase variants |
| WO1996033267A1 (en) | 1995-04-21 | 1996-10-24 | Novo Nordisk A/S | Cyclomaltodextrin glucanotransferase variants |
| US5576282A (en) | 1995-09-11 | 1996-11-19 | The Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| US5580485A (en) | 1994-06-13 | 1996-12-03 | Lever Brothers Company, Division Of Conopco, Inc. | Bleach activation |
| US5595967A (en) | 1995-02-03 | 1997-01-21 | The Procter & Gamble Company | Detergent compositions comprising multiperacid-forming bleach activators |
| US5597936A (en) | 1995-06-16 | 1997-01-28 | The Procter & Gamble Company | Method for manufacturing cobalt catalysts |
| WO1998035006A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Liquid cleaning composition |
| WO1998035002A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Cleaning compositions |
| WO1998035004A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Solid detergent compositions |
| WO1998035003A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Detergent compound |
| WO1998035005A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | A cleaning composition |
| US5817614A (en) | 1996-08-29 | 1998-10-06 | Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| WO1999002663A1 (en) | 1997-07-07 | 1999-01-21 | Novo Nordisk A/S | Alkaline xyloglucanase |
| WO1999005243A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Detergent compositions containing mixtures of crystallinity-disrupted surfactants |
| WO1999005084A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Process for making alkylbenzenesulfonate surfactants from alcohols and products thereof |
| WO1999005242A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkylbenzenesulfonate surfactants |
| WO1999005244A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkyl aryl sulfonate surfactants |
| WO1999005082A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved processes for making alkylbenzenesulfonate surfactants and products thereof |
| WO1999005241A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Cleaning products comprising improved alkylarylsulfonate surfactants prepared via vinylidene olefins and processes for preparation thereof |
| WO1999007656A2 (en) | 1997-08-08 | 1999-02-18 | The Procter & Gamble Company | Improved processes for making surfactants via adsorptive separation and products thereof |
| WO1999020726A1 (en) | 1997-10-23 | 1999-04-29 | The Procter & Gamble Company | Bleaching compositions comprising multiply-substituted protease variants |
| WO1999027083A1 (en) | 1997-11-24 | 1999-06-03 | Novo Nordisk A/S | PECTIN DEGRADING ENZYMES FROM $i(BACILLUS LICHENIFORMIS) |
| US6004922A (en) | 1996-05-03 | 1999-12-21 | The Procter & Gamble Company | Laundry detergent compositions comprising cationic surfactants and modified polyamine soil dispersents |
| US6020303A (en) | 1996-04-16 | 2000-02-01 | The Procter & Gamble Company | Mid-chain branched surfactants |
| US6022844A (en) | 1996-03-05 | 2000-02-08 | The Procter & Gamble Company | Cationic detergent compounds |
| WO2000023548A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| WO2000023549A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| US6060443A (en) | 1996-04-16 | 2000-05-09 | The Procter & Gamble Company | Mid-chain branched alkyl sulfate surfactants |
| US6060299A (en) | 1998-06-10 | 2000-05-09 | Novo Nordisk A/S | Enzyme exhibiting mannase activity, cleaning compositions, and methods of use |
| US6093856A (en) | 1996-11-26 | 2000-07-25 | The Procter & Gamble Company | Polyoxyalkylene surfactants |
| WO2000047708A1 (en) | 1999-02-10 | 2000-08-17 | The Procter & Gamble Company | Low density particulate solids useful in laundry detergents |
| WO2000061718A1 (en) * | 1999-04-13 | 2000-10-19 | The Procter & Gamble Company | Light reflecting particles |
| US6136769A (en) | 1996-05-17 | 2000-10-24 | The Procter & Gamble Company | Alkoxylated cationic detergency ingredients |
| US6150322A (en) | 1998-08-12 | 2000-11-21 | Shell Oil Company | Highly branched primary alcohol compositions and biodegradable detergents made therefrom |
| US6221825B1 (en) | 1996-12-31 | 2001-04-24 | The Procter & Gamble Company | Thickened, highly aqueous liquid detergent compositions |
| WO2001042408A2 (en) | 1999-12-08 | 2001-06-14 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| WO2001083661A1 (en) * | 2000-04-28 | 2001-11-08 | The Procter & Gamble Company | Pouched composition |
| US20020032142A1 (en) * | 2000-06-30 | 2002-03-14 | The Procter & Gamble Company | Detergent compositions comprising a cyclodextrin glucanotransferase enzyme and a detergent ingredient |
| US6482994B2 (en) | 1997-08-02 | 2002-11-19 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| US6486116B1 (en) * | 1998-07-10 | 2002-11-26 | Gerald Thomas Hinton | Detergent |
| US6566114B1 (en) * | 1998-06-10 | 2003-05-20 | Novozymes, A/S | Mannanases |
| US6649085B2 (en) | 2000-11-25 | 2003-11-18 | Clariant Gmbh | Cyclic sugar ketones as catalysts for peroxygen compounds |
| WO2005042532A1 (en) | 2003-10-31 | 2005-05-12 | Unilever Plc | Bispidon-derivated ligands and complex for catalytically bleaching a substrate |
| WO2005047264A1 (en) | 2003-11-06 | 2005-05-26 | The Procter & Gamble Company | Process for producing dihydroisoquinoline zwitterions |
-
2010
- 2010-06-30 WO PCT/US2010/040506 patent/WO2011005623A1/en active Application Filing
Patent Citations (90)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1154344A (en) * | 1955-07-13 | 1958-04-04 | Henkel & Cie Gmbh | Washing and bleaching agents |
| US3630921A (en) * | 1968-12-03 | 1971-12-28 | Henkel & Cie Gmbh | Scouring agents with a bleaching and disinfecting action |
| US4228042A (en) | 1978-06-26 | 1980-10-14 | The Procter & Gamble Company | Biodegradable cationic surface-active agents containing ester or amide and polyalkoxy group |
| US4260529A (en) | 1978-06-26 | 1981-04-07 | The Procter & Gamble Company | Detergent composition consisting essentially of biodegradable nonionic surfactant and cationic surfactant containing ester or amide |
| US4239660A (en) | 1978-12-13 | 1980-12-16 | The Procter & Gamble Company | Detergent composition comprising a hydrolyzable cationic surfactant and specific alkalinity source |
| US4430243A (en) | 1981-08-08 | 1984-02-07 | The Procter & Gamble Company | Bleach catalyst compositions and use thereof in laundry bleaching and detergent compositions |
| US4565647B1 (en) | 1982-04-26 | 1994-04-05 | Procter & Gamble | Foaming surfactant compositions |
| US4483779A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions comprising polyglycoside and polyethoxylate surfactants and anionic fluorescer |
| US4483780A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions containing polyglycoside and polyethoxylate detergent surfactants |
| US4565647A (en) | 1982-04-26 | 1986-01-21 | The Procter & Gamble Company | Foaming surfactant compositions |
| US4412934A (en) | 1982-06-30 | 1983-11-01 | The Procter & Gamble Company | Bleaching compositions |
| US4545784A (en) | 1983-04-14 | 1985-10-08 | Interox Chemicals Limited | Particulate sodium perborate monohydrate containing adsorbed activator |
| EP0130756A1 (en) | 1983-06-24 | 1985-01-09 | Genencor International, Inc. | Procaryotic carbonyl hydrolases, methods, DNA, vectors and transformed hosts for producing them, and detergent compositions containing them |
| US4634551A (en) | 1985-06-03 | 1987-01-06 | Procter & Gamble Company | Bleaching compounds and compositions comprising fatty peroxyacids salts thereof and precursors therefor having amide moieties in the fatty chain |
| US4728455A (en) | 1986-03-07 | 1988-03-01 | Lever Brothers Company | Detergent bleach compositions, bleaching agents and bleach activators |
| US4915854A (en) | 1986-11-14 | 1990-04-10 | The Procter & Gamble Company | Ion-pair complex conditioning agent and compositions containing same |
| US4966723A (en) | 1988-02-11 | 1990-10-30 | Bp Chemicals Limited | Bleach activators in detergent compositions |
| US5114611A (en) | 1989-04-13 | 1992-05-19 | Lever Brothers Company, Divison Of Conopco, Inc. | Bleach activation |
| WO1991006637A1 (en) | 1989-10-31 | 1991-05-16 | Genencor International, Inc. | Subtilisin mutants |
| US5114606A (en) | 1990-02-19 | 1992-05-19 | Lever Brothers Company, Division Of Conopco, Inc. | Bleaching composition comprising as a bleaching catalyst a complex of manganese with a non-carboxylate polyhydroxy ligand |
| US5246621A (en) | 1990-05-21 | 1993-09-21 | Lever Brothers Company, Division Of Conopco, Inc. | Bleach activation by manganese-based coordination complexes |
| US5244594A (en) | 1990-05-21 | 1993-09-14 | Lever Brothers Company, Division Of Conopco, Inc. | Bleach activation multinuclear manganese-based coordination complexes |
| WO1992006162A1 (en) | 1990-09-28 | 1992-04-16 | The Procter & Gamble Company | Detergent containing alkyl sulfate and polyhydroxy fatty acid amide surfactants |
| US5332528A (en) | 1990-09-28 | 1994-07-26 | The Procter & Gamble Company | Polyhydroxy fatty acid amides in soil release agent-containing detergent compositions |
| US5227084A (en) | 1991-04-17 | 1993-07-13 | Lever Brothers Company, Division Of Conopco, Inc. | Concentrated detergent powder compositions |
| US5274147A (en) | 1991-07-11 | 1993-12-28 | Lever Brothers Company, Division Of Conopco, Inc. | Process for preparing manganese complexes |
| US5246612A (en) | 1991-08-23 | 1993-09-21 | Lever Brothers Company, Division Of Conopco, Inc. | Machine dishwashing composition containing peroxygen bleach, manganese complex and enzymes |
| EP0544440A2 (en) | 1991-11-20 | 1993-06-02 | Unilever Plc | Bleach catalyst composition, manufacture and use thereof in detergent and/or bleach compositions |
| US5153161A (en) | 1991-11-26 | 1992-10-06 | Lever Brothers Company, Division Of Conopco, Inc. | Synthesis of manganese oxidation catalyst |
| EP0544490A1 (en) | 1991-11-26 | 1993-06-02 | Unilever Plc | Detergent bleach compositions |
| US5194416A (en) | 1991-11-26 | 1993-03-16 | Lever Brothers Company, Division Of Conopco, Inc. | Manganese catalyst for activating hydrogen peroxide bleaching |
| EP0549271A1 (en) | 1991-12-20 | 1993-06-30 | Unilever Plc | Bleach activation |
| EP0549272A1 (en) | 1991-12-20 | 1993-06-30 | Unilever Plc | Bleach activation |
| WO1993019146A1 (en) | 1992-03-16 | 1993-09-30 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| WO1993019038A1 (en) | 1992-03-26 | 1993-09-30 | The Procter & Gamble Company | Process for reducing the levels of fatty acid contaminants in polyhydroxy fatty acid amide surfactants |
| US5256779A (en) | 1992-06-18 | 1993-10-26 | Lever Brothers Company, Division Of Conopco, Inc. | Synthesis of manganese oxidation catalyst |
| US5284944A (en) | 1992-06-30 | 1994-02-08 | Lever Brothers Company, Division Of Conopco, Inc. | Improved synthesis of 1,4,7-triazacyclononane |
| WO1994002597A1 (en) | 1992-07-23 | 1994-02-03 | Novo Nordisk A/S | MUTANT α-AMYLASE, DETERGENT, DISH WASHING AGENT, AND LIQUEFACTION AGENT |
| US5280117A (en) | 1992-09-09 | 1994-01-18 | Lever Brothers Company, A Division Of Conopco, Inc. | Process for the preparation of manganese bleach catalyst |
| WO1994009099A1 (en) | 1992-10-13 | 1994-04-28 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| WO1995010591A1 (en) | 1993-10-14 | 1995-04-20 | The Procter & Gamble Company | Protease-containing cleaning compositions |
| US5360568A (en) | 1993-11-12 | 1994-11-01 | Lever Brothers Company, Division Of Conopco, Inc. | Imine quaternary salts as bleach catalysts |
| US5360569A (en) | 1993-11-12 | 1994-11-01 | Lever Brothers Company, Division Of Conopco, Inc. | Activation of bleach precursors with catalytic imine quaternary salts |
| WO1995026393A1 (en) | 1994-03-29 | 1995-10-05 | The Procter & Gamble Company | Detergent composition comprising lipoxidase enzymes |
| WO1995028468A1 (en) * | 1994-04-13 | 1995-10-26 | The Procter & Gamble Company | Detergent compositions |
| US5580485A (en) | 1994-06-13 | 1996-12-03 | Lever Brothers Company, Division Of Conopco, Inc. | Bleach activation |
| WO1996023873A1 (en) | 1995-02-03 | 1996-08-08 | Novo Nordisk A/S | Amylase variants |
| US5595967A (en) | 1995-02-03 | 1997-01-21 | The Procter & Gamble Company | Detergent compositions comprising multiperacid-forming bleach activators |
| WO1996033267A1 (en) | 1995-04-21 | 1996-10-24 | Novo Nordisk A/S | Cyclomaltodextrin glucanotransferase variants |
| US5597936A (en) | 1995-06-16 | 1997-01-28 | The Procter & Gamble Company | Method for manufacturing cobalt catalysts |
| US5576282A (en) | 1995-09-11 | 1996-11-19 | The Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| US6022844A (en) | 1996-03-05 | 2000-02-08 | The Procter & Gamble Company | Cationic detergent compounds |
| US6060443A (en) | 1996-04-16 | 2000-05-09 | The Procter & Gamble Company | Mid-chain branched alkyl sulfate surfactants |
| US6020303A (en) | 1996-04-16 | 2000-02-01 | The Procter & Gamble Company | Mid-chain branched surfactants |
| US6004922A (en) | 1996-05-03 | 1999-12-21 | The Procter & Gamble Company | Laundry detergent compositions comprising cationic surfactants and modified polyamine soil dispersents |
| US6136769A (en) | 1996-05-17 | 2000-10-24 | The Procter & Gamble Company | Alkoxylated cationic detergency ingredients |
| US5817614A (en) | 1996-08-29 | 1998-10-06 | Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| US6153577A (en) | 1996-11-26 | 2000-11-28 | The Procter & Gamble Company | Polyoxyalkylene surfactants |
| US6093856A (en) | 1996-11-26 | 2000-07-25 | The Procter & Gamble Company | Polyoxyalkylene surfactants |
| US6221825B1 (en) | 1996-12-31 | 2001-04-24 | The Procter & Gamble Company | Thickened, highly aqueous liquid detergent compositions |
| WO1998035005A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | A cleaning composition |
| WO1998035006A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Liquid cleaning composition |
| WO1998035002A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Cleaning compositions |
| WO1998035004A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Solid detergent compositions |
| WO1998035003A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Detergent compound |
| WO1999002663A1 (en) | 1997-07-07 | 1999-01-21 | Novo Nordisk A/S | Alkaline xyloglucanase |
| WO1999005244A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkyl aryl sulfonate surfactants |
| WO1999005241A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Cleaning products comprising improved alkylarylsulfonate surfactants prepared via vinylidene olefins and processes for preparation thereof |
| WO1999005082A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved processes for making alkylbenzenesulfonate surfactants and products thereof |
| WO1999005242A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkylbenzenesulfonate surfactants |
| WO1999005084A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Process for making alkylbenzenesulfonate surfactants from alcohols and products thereof |
| WO1999005243A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Detergent compositions containing mixtures of crystallinity-disrupted surfactants |
| US6482994B2 (en) | 1997-08-02 | 2002-11-19 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| WO1999007656A2 (en) | 1997-08-08 | 1999-02-18 | The Procter & Gamble Company | Improved processes for making surfactants via adsorptive separation and products thereof |
| WO1999020726A1 (en) | 1997-10-23 | 1999-04-29 | The Procter & Gamble Company | Bleaching compositions comprising multiply-substituted protease variants |
| WO1999027083A1 (en) | 1997-11-24 | 1999-06-03 | Novo Nordisk A/S | PECTIN DEGRADING ENZYMES FROM $i(BACILLUS LICHENIFORMIS) |
| US6060299A (en) | 1998-06-10 | 2000-05-09 | Novo Nordisk A/S | Enzyme exhibiting mannase activity, cleaning compositions, and methods of use |
| US6566114B1 (en) * | 1998-06-10 | 2003-05-20 | Novozymes, A/S | Mannanases |
| US6486116B1 (en) * | 1998-07-10 | 2002-11-26 | Gerald Thomas Hinton | Detergent |
| US6150322A (en) | 1998-08-12 | 2000-11-21 | Shell Oil Company | Highly branched primary alcohol compositions and biodegradable detergents made therefrom |
| WO2000023549A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| WO2000023548A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| WO2000047708A1 (en) | 1999-02-10 | 2000-08-17 | The Procter & Gamble Company | Low density particulate solids useful in laundry detergents |
| WO2000061718A1 (en) * | 1999-04-13 | 2000-10-19 | The Procter & Gamble Company | Light reflecting particles |
| WO2001042408A2 (en) | 1999-12-08 | 2001-06-14 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| WO2001083661A1 (en) * | 2000-04-28 | 2001-11-08 | The Procter & Gamble Company | Pouched composition |
| US20020032142A1 (en) * | 2000-06-30 | 2002-03-14 | The Procter & Gamble Company | Detergent compositions comprising a cyclodextrin glucanotransferase enzyme and a detergent ingredient |
| US6649085B2 (en) | 2000-11-25 | 2003-11-18 | Clariant Gmbh | Cyclic sugar ketones as catalysts for peroxygen compounds |
| WO2005042532A1 (en) | 2003-10-31 | 2005-05-12 | Unilever Plc | Bispidon-derivated ligands and complex for catalytically bleaching a substrate |
| WO2005047264A1 (en) | 2003-11-06 | 2005-05-26 | The Procter & Gamble Company | Process for producing dihydroisoquinoline zwitterions |
Non-Patent Citations (5)
| Title |
|---|
| "Journal of the Chemical Society", CHEMICAL COMMUNICATIONS, 1994, pages 2569 - 70 |
| POLISH JOURNAL OF CHEMISTRY, vol. 77, no. 5, 2003, pages 577 - 590 |
| TETRAHEDRON LETTERS, vol. 35, no. 34, 1994, pages 6329 - 30 |
| TETRAHEDRON, vol. 49, no. 2, 1992, pages 423 - 38 |
| THE JOURNAL OF ORGANIC CHEMISTRY, vol. 55, no. 4, 1990, pages 1254 - 61 |
Cited By (66)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2840134A1 (en) | 2010-04-26 | 2015-02-25 | Novozymes A/S | Enzyme granules |
| WO2011134809A1 (en) | 2010-04-26 | 2011-11-03 | Novozymes A/S | Enzyme granules |
| US9539190B2 (en) | 2010-05-07 | 2017-01-10 | Conopco, Inc. | Skin conditioning compositions containing 12-hydroxystearic acid |
| WO2012101149A1 (en) | 2011-01-26 | 2012-08-02 | Novozymes A/S | Storage-stable enzyme granules |
| WO2012175401A2 (en) | 2011-06-20 | 2012-12-27 | Novozymes A/S | Particulate composition |
| WO2012175708A2 (en) | 2011-06-24 | 2012-12-27 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| EP4026901A2 (en) | 2011-06-30 | 2022-07-13 | Novozymes A/S | Method for screening alpha-amylases |
| EP3543333A2 (en) | 2011-06-30 | 2019-09-25 | Novozymes A/S | Method for screening alpha-amylases |
| WO2013001087A2 (en) | 2011-06-30 | 2013-01-03 | Novozymes A/S | Method for screening alpha-amylases |
| WO2013007594A1 (en) | 2011-07-12 | 2013-01-17 | Novozymes A/S | Storage-stable enzyme granules |
| WO2013024021A1 (en) | 2011-08-15 | 2013-02-21 | Novozymes A/S | Polypeptides having cellulase activity and polynucleotides encoding same |
| WO2013041689A1 (en) | 2011-09-22 | 2013-03-28 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2013076269A1 (en) | 2011-11-25 | 2013-05-30 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2013092635A1 (en) | 2011-12-20 | 2013-06-27 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2013110766A1 (en) | 2012-01-26 | 2013-08-01 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2013120948A1 (en) | 2012-02-17 | 2013-08-22 | Novozymes A/S | Subtilisin variants and polynucleotides encoding same |
| WO2013131964A1 (en) | 2012-03-07 | 2013-09-12 | Novozymes A/S | Detergent composition and substitution of optical brighteners in detergent compositions |
| WO2013167581A1 (en) | 2012-05-07 | 2013-11-14 | Novozymes A/S | Polypeptides having xanthan degrading activity and polynucleotides encoding same |
| WO2013189972A2 (en) | 2012-06-20 | 2013-12-27 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2014096259A1 (en) | 2012-12-21 | 2014-06-26 | Novozymes A/S | Polypeptides having protease activiy and polynucleotides encoding same |
| EP3321360A2 (en) | 2013-01-03 | 2018-05-16 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2014183921A1 (en) | 2013-05-17 | 2014-11-20 | Novozymes A/S | Polypeptides having alpha amylase activity |
| EP3786269A1 (en) | 2013-06-06 | 2021-03-03 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2014207227A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2014207224A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2015001017A2 (en) | 2013-07-04 | 2015-01-08 | Novozymes A/S | Polypeptides having anti-redeposition effect and polynucleotides encoding same |
| EP2832853A1 (en) | 2013-07-29 | 2015-02-04 | Henkel AG&Co. KGAA | Detergent composition comprising protease variants |
| EP3309249A1 (en) | 2013-07-29 | 2018-04-18 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3611260A1 (en) | 2013-07-29 | 2020-02-19 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3613853A1 (en) | 2013-07-29 | 2020-02-26 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3339436A1 (en) | 2013-07-29 | 2018-06-27 | Henkel AG & Co. KGaA | Detergent composition comprising protease variants |
| WO2015049370A1 (en) | 2013-10-03 | 2015-04-09 | Novozymes A/S | Detergent composition and use of detergent composition |
| EP3453757A1 (en) | 2013-12-20 | 2019-03-13 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2015134737A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015134729A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of non-cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015150457A1 (en) | 2014-04-01 | 2015-10-08 | Novozymes A/S | Polypeptides having alpha amylase activity |
| WO2015189371A1 (en) | 2014-06-12 | 2015-12-17 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| EP3739029A1 (en) | 2014-07-04 | 2020-11-18 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3878960A1 (en) | 2014-07-04 | 2021-09-15 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2016079305A1 (en) | 2014-11-20 | 2016-05-26 | Novozymes A/S | Alicyclobacillus variants and polynucleotides encoding same |
| EP3690037A1 (en) | 2014-12-04 | 2020-08-05 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| US10760036B2 (en) | 2014-12-15 | 2020-09-01 | Henkel Ag & Co. Kgaa | Detergent composition comprising subtilase variants |
| EP3608403A2 (en) | 2014-12-15 | 2020-02-12 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| EP3106508A1 (en) | 2015-06-18 | 2016-12-21 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| EP4071244A1 (en) | 2015-06-18 | 2022-10-12 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3872175A1 (en) | 2015-06-18 | 2021-09-01 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2017064253A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| EP4324919A2 (en) | 2015-10-14 | 2024-02-21 | Novozymes A/S | Polypeptide variants |
| WO2017064269A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptide variants |
| WO2017207762A1 (en) | 2016-06-03 | 2017-12-07 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3950941A2 (en) | 2016-07-13 | 2022-02-09 | Novozymes A/S | Dnase polypeptide variants |
| WO2018011276A1 (en) | 2016-07-13 | 2018-01-18 | The Procter & Gamble Company | Bacillus cibi dnase variants and uses thereof |
| WO2018011277A1 (en) | 2016-07-13 | 2018-01-18 | Novozymes A/S | Bacillus cibi dnase variants |
| WO2019081724A1 (en) | 2017-10-27 | 2019-05-02 | Novozymes A/S | Dnase variants |
| WO2019081721A1 (en) | 2017-10-27 | 2019-05-02 | Novozymes A/S | Dnase variants |
| WO2019084349A1 (en) | 2017-10-27 | 2019-05-02 | The Procter & Gamble Company | Detergent compositions comprising polypeptide variants |
| WO2019201793A1 (en) | 2018-04-17 | 2019-10-24 | Novozymes A/S | Polypeptides comprising carbohydrate binding activity in detergent compositions and their use in reducing wrinkles in textile or fabric. |
| WO2020188095A1 (en) | 2019-03-21 | 2020-09-24 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2020207944A1 (en) | 2019-04-10 | 2020-10-15 | Novozymes A/S | Polypeptide variants |
| WO2021037895A1 (en) | 2019-08-27 | 2021-03-04 | Novozymes A/S | Detergent composition |
| WO2021053127A1 (en) | 2019-09-19 | 2021-03-25 | Novozymes A/S | Detergent composition |
| WO2021064068A1 (en) | 2019-10-03 | 2021-04-08 | Novozymes A/S | Polypeptides comprising at least two carbohydrate binding domains |
| EP3892708A1 (en) | 2020-04-06 | 2021-10-13 | Henkel AG & Co. KGaA | Cleaning compositions comprising dispersin variants |
| WO2022074037A2 (en) | 2020-10-07 | 2022-04-14 | Novozymes A/S | Alpha-amylase variants |
| WO2022171780A2 (en) | 2021-02-12 | 2022-08-18 | Novozymes A/S | Alpha-amylase variants |
| WO2022268885A1 (en) | 2021-06-23 | 2022-12-29 | Novozymes A/S | Alpha-amylase polypeptides |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2011005623A1 (en) | Laundry detergent composition comprising low level of bleach | |
| US20110005007A1 (en) | Method of Laundering Fabric Using a Compacted Laundry Detergent Composition | |
| AU662577B2 (en) | Bleach activation | |
| US20110010869A1 (en) | Method of Laundering Fabric Using a Compacted Laundry Detergent Composition | |
| EP2292725B2 (en) | Method of laundering fabrics at low temperature | |
| EP1811014B1 (en) | A composition comprising a pre-formed peroxyacid and a bleach catalyst | |
| US8541354B2 (en) | Catalytic laundry detergent composition comprising relatively low levels of water-soluble electrolyte | |
| US20110005006A1 (en) | Method of Laundering Fabric Using a Compacted Laundry Detergent Composition | |
| US8211848B2 (en) | Catalytic laundry detergent composition comprising relatively low levels of water-soluble electrolyte | |
| EP0754218A1 (en) | Bleach compositions comprising metal-containing bleach catalysts and antioxidants | |
| EP2451919A1 (en) | Method of laundering fabric using a liquid laundry detergent composition | |
| US8586521B2 (en) | Method of laundering fabrics at low temperature | |
| AU2003205759B2 (en) | Liquid cleaning compositions and their use | |
| WO2011005917A1 (en) | Method of laundering fabric using a liquid laundry detergent composition | |
| WO2011005911A1 (en) | Method of laundering fabric using a compacted liquid laundry detergent composition | |
| WO2011133372A1 (en) | Detergent composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10734599 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 10734599 Country of ref document: EP Kind code of ref document: A1 |